
Concept explainers
(a)
Interpretation: To determine the electron carrier NADH is associated with which of the protein complexes (I, II, III, and IV) of the ETC.
Concept introduction: Electron transport chain is a sequence of biochemical reactions in which electrons and hydrogen atoms from the citric acid cycle are transferred to various intermediate carriers and finally reacts with molecular oxygen to form a water molecule. There are four complexes associated with the electron transport chain that is present in the inner mitochondrial membrane. The four complexes that help in the electron transfer in the electron transport chain are:
Complex I:
Complex II:
Complex III:
Complex IV:
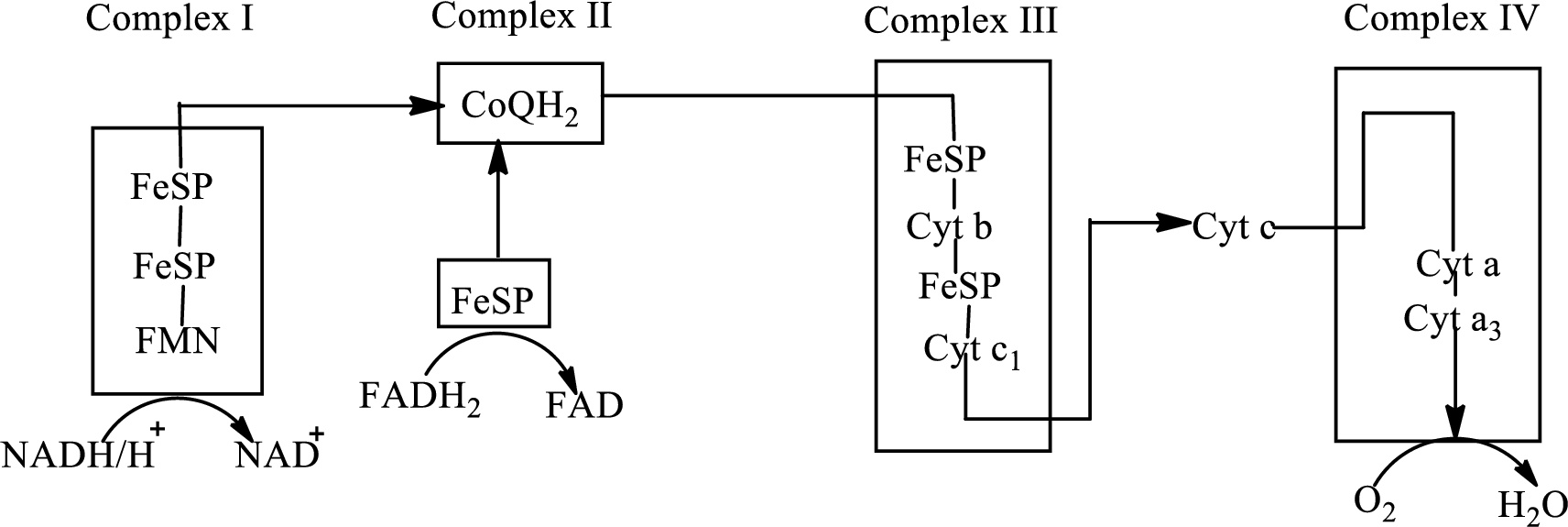
An overview of the electron transport chain is as follows:
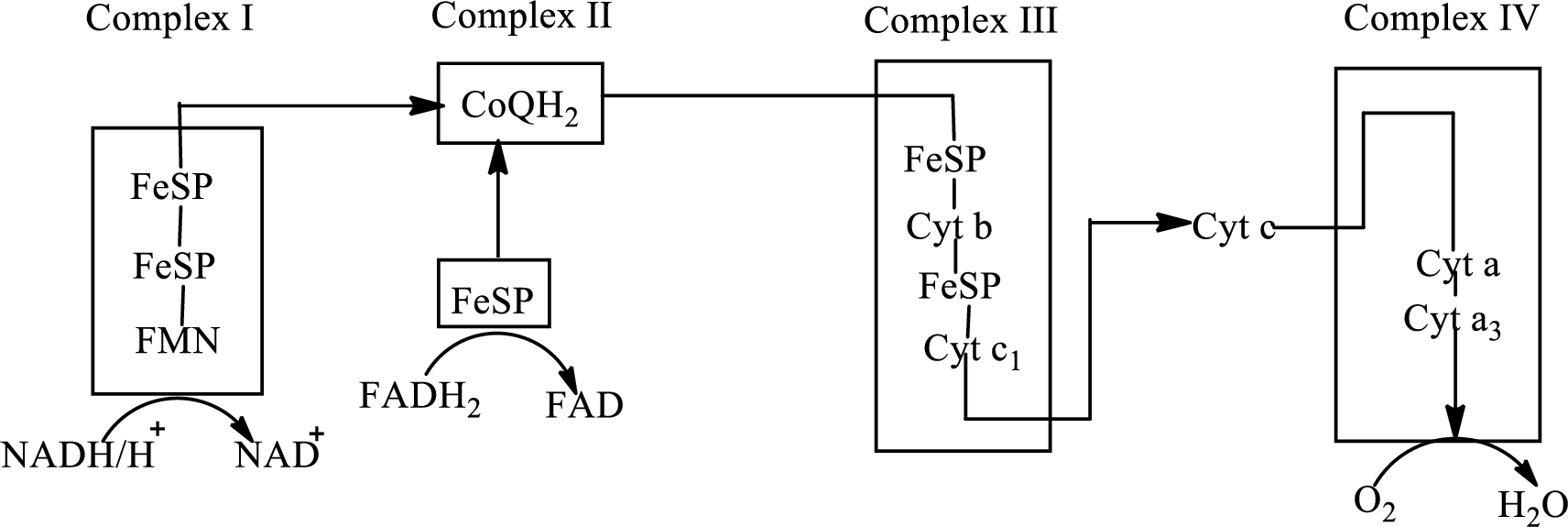
(a)
Answer to Problem 23.91EP
NADH is associated with the complex I of the electron transport chain. Electrons from the citric acid cycle are transported to complex I through NADH molecule.
Explanation of Solution
Complex I consists of more than 40 structural subunits. Its structure has B-vitamin-containing flavin mononucleotide
(b)
Interpretation: To determine the electron carrier
Concept introduction: Electron transport chain is a sequence of biochemical reactions in which electrons and hydrogen atoms from the citric acid cycle are transferred to various intermediate carriers and finally reacts with molecular oxygen to form a water molecule. There are four complexes associated with the electron transport chain that is present in the inner mitochondrial membrane. The four complexes that help in the electron transfer in the electron transport chain are:
Complex I:
Complex II:
Complex III:
Complex IV:
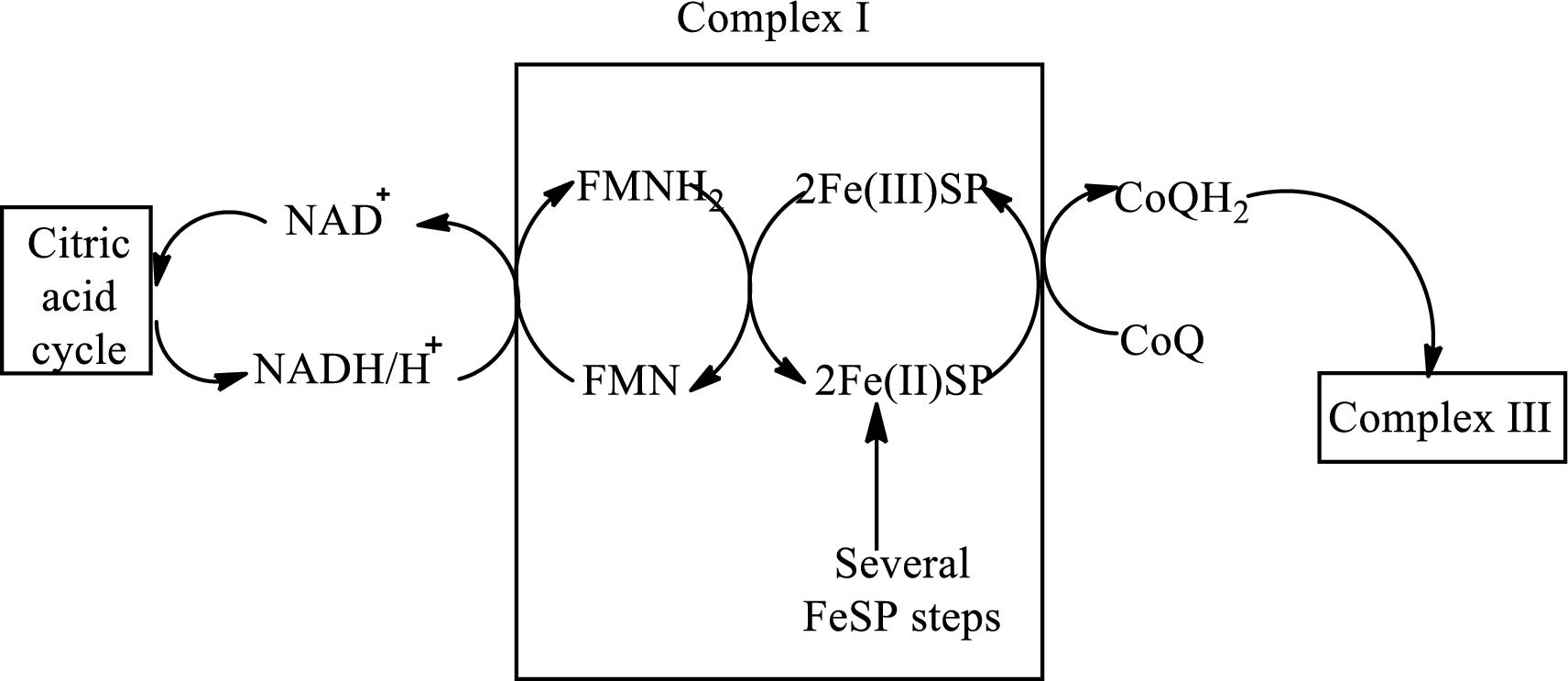
An overview of the electron transport chain is as follows:
(b)
Answer to Problem 23.91EP
Explanation of Solution
NADH and FADH2from complex I and II transfers electrons to
In complex III,
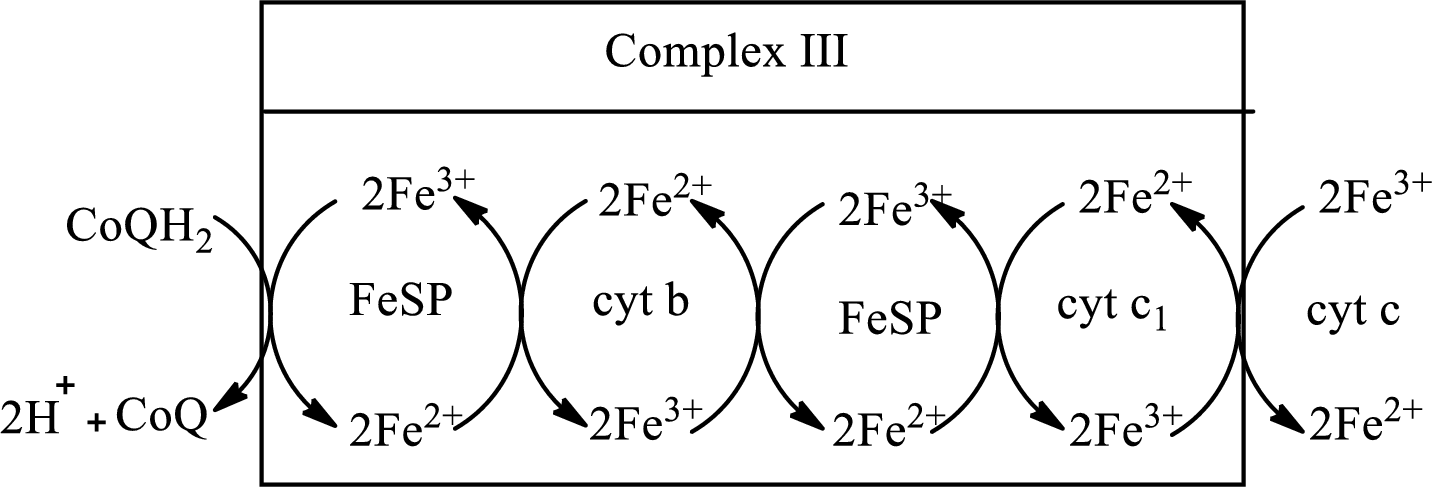
(c)
Interpretation: To determine the electron carrier
Concept introduction: Electron transport chain is a sequence of biochemical reactions in which electrons and hydrogen atoms from the citric acid cycle are transferred to various intermediate carriers and finally reacts with molecular oxygen to form a water molecule. There are four complexes associated with the electron transport chain that is present in the inner mitochondrial membrane. The four complexes that help in the electron transfer in the electron transport chain are:
Complex I:
Complex II:
Complex III:
Complex IV:
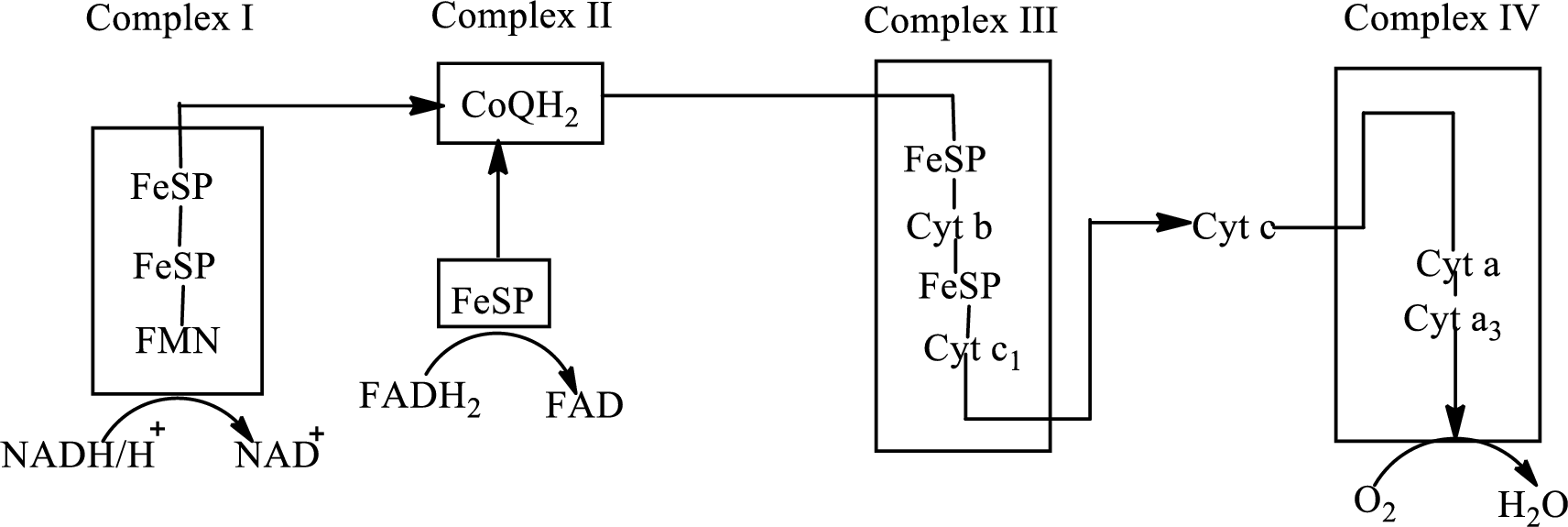
An overview of the electron transport chain is as follows:
(c)
Answer to Problem 23.91EP
Explanation of Solution
Complex III consists of 11 structural subunits. Its structural subunits contain iron-sulfur proteins and various cytochromes.
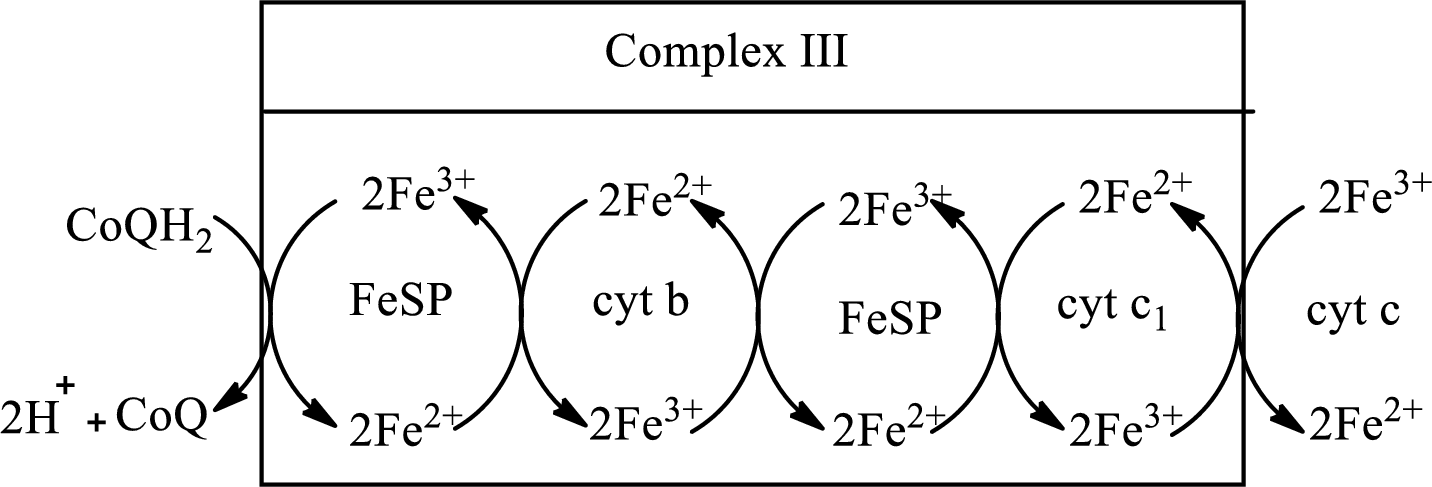
Initially,
(d)
Interpretation: To determine that the electron carrier
Concept introduction: Electron transport chain is a sequence of biochemical reactions in which electrons and hydrogen atoms from the citric acid cycle are transferred to various intermediate carriers and finally reacts with molecular oxygen to form a water molecule. There are four complexes associated with the electron transport chain that is present in the inner mitochondrial membrane. The four complexes that help in the electron transfer in the electron transport chain are:
Complex I:
Complex II:
Complex III:
Complex IV:
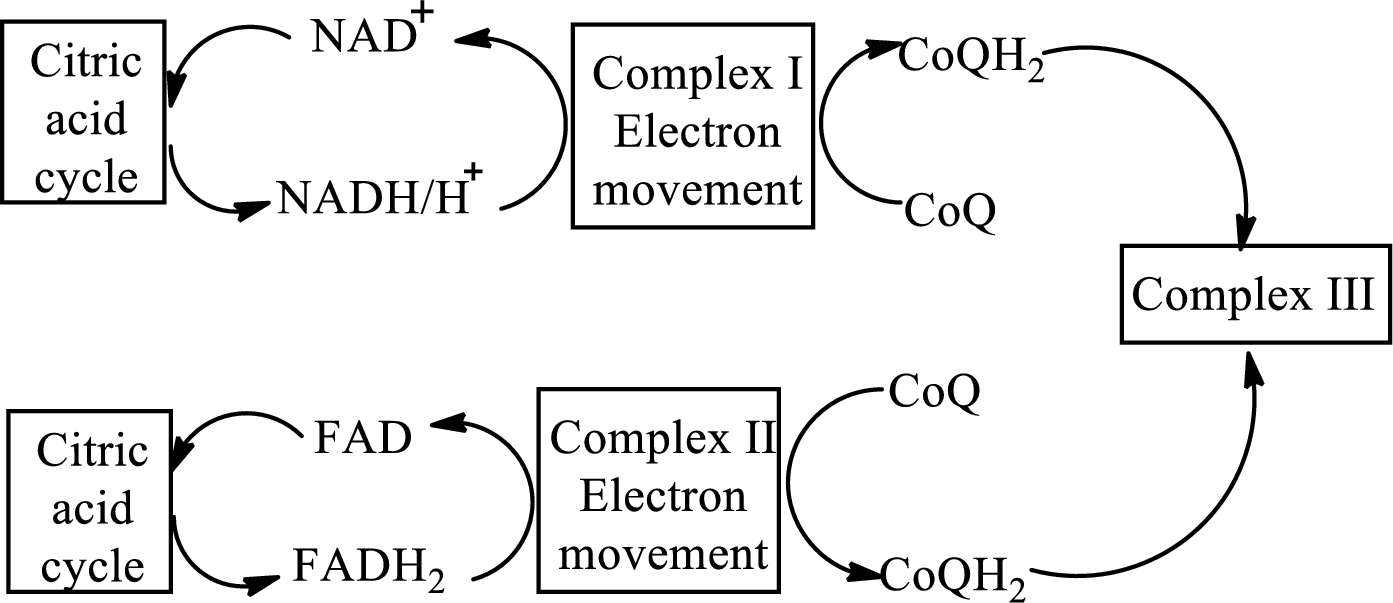
An overview of the electron transport chain is as follows:
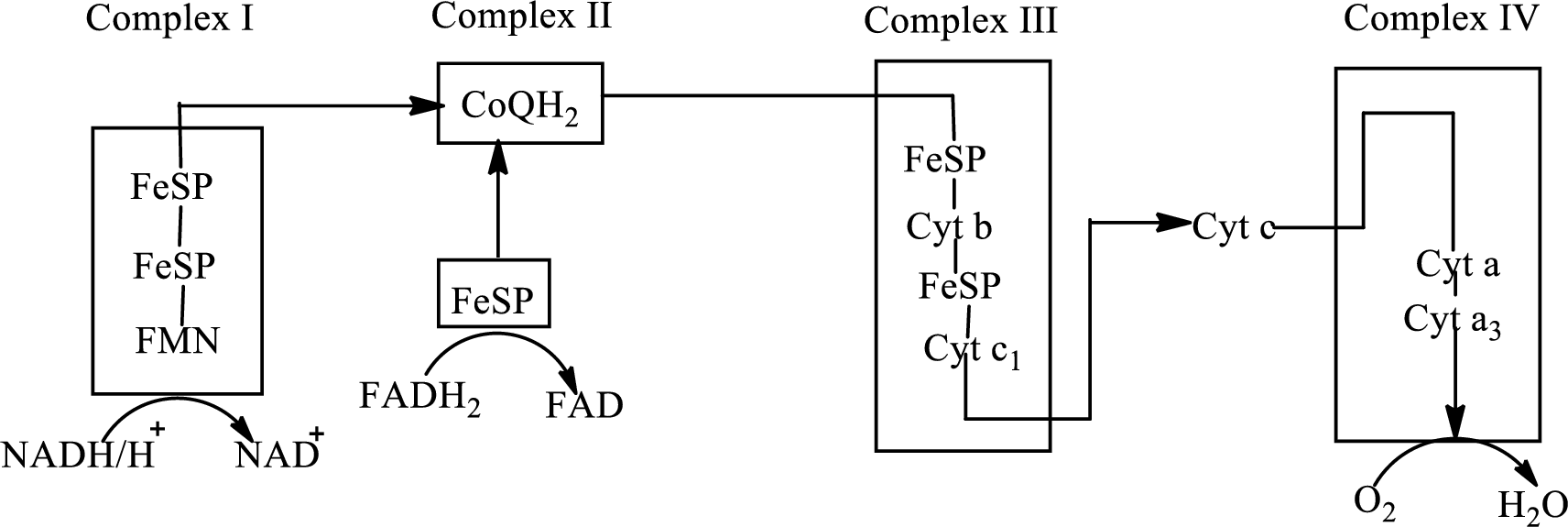
(d)
Answer to Problem 23.91EP
Explanation of Solution
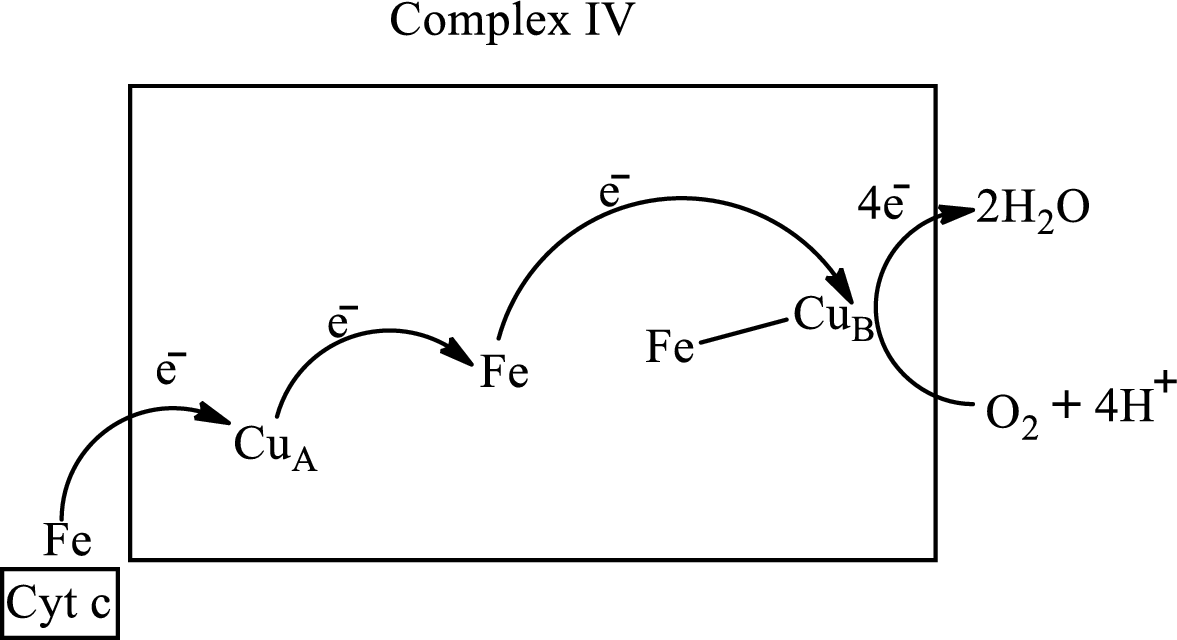
Complex IV contains 13 structural subunits. It has two cytochrome units that have copper along with iron. The copper also participates in the
The diagrammatic representation of the electron transfer in complex IV in the electron transport chain is:
Want to see more full solutions like this?
Chapter 23 Solutions
Study Guide with Selected Solutions for Stoker's General, Organic, and Biological Chemistry, 7th
- Define the following terms: a. Fe–S clusters b. electron transport c. CoQ d. ubisemiquinone e. UQH2arrow_forwardAll are single-electron carriers EXCEPT: a. UQH2. b. Cyt bL. c. Cyt bH. d. Rieske protein Fe-S clusters. e. Cyt c1. 2.The energy input for the synthesis of glycogen occurs during which of the following enzyme catalysed steps? a. phosphoglucomutase b. UDP-glucose pyrophTosphorylase c. glycogen synthase d. amylo-(1,4®1,6)-transglycosylase e. none of the above require energy input ______________________________________________________________________________(arrow_forwardin one of the reactions in the electron transport chain, complex I transfer electrons to coenzyme Q. which molecule is reduced and which is oxidized in this reaction? a. complex I is reduced b/c it loses electrons, and Q is oxidized b/c it gains electrons b. complex I is oxidized b/c it loses electrons, and Q is reduced b/c it gains electrons c. complex I is reduced b/c it gains electrons, and Q is oxidized b/c it loses electrons d. complex 1 and Q are both oxidized b/c elections are moved from one molecule to the otherarrow_forward
- Mitochondria are extracted from yeast and placed in a test tube at pH 7.0, where a series of experiments are performed. Draw the predicted graph of total 02 consumed (dashed lines) and total ATP concentration (solid lines) for the following experiment. Label the axes and clearly indicate on your graph where each new addition takes place. a. Add succinate b. ADP and Pi c. Raise the pH in the test tube to pH 9.0 d. Lower the pH in the test tube to pH 5.0 e. Add cyanide (blocks all activity of complex IV) f. Add carbonyl cyanide m-chloro phenyl hydrazine (CCCP; a mitochondrial uncoupling agent)arrow_forwardWhich of the following statements concerning ATP is true? a. The free energy value for the hydrolysis of ATP is nearly the same for ADP. b. The free energy value for the hydrolysis of ATP is greater than that for ADP. c. ATP hydrolysis is more likely at pH 5 than at pH 7. d. One mole of glycerate-1,3-bisphosphate can phosphorylate one mole of AMP to yield ATP.arrow_forwardClassify each of the following molecules as (1) an oxidizing agent, (2) a reducing agent, or (3) neither an oxidizing agent nor a reducing agent. a. FAD b. ADP c. Coenzyme A d. NAD+arrow_forward
- List the following substances in order of increasing tendency to accept e-: a. pyruvate, b. CoQ, c. O2 d. FADarrow_forwardWhich of the following statements is a lie? Select one: a.The transfer of energy in living systems use electrons to allow the cell to transfer energy in large amounts. b.Electrons play an important role in redox reactions. This is true because it is the electrons that undergo the redox reactions in living systems. c.For nicotinamide adenine (NAD), its oxidized form is NAD+ while its reduced form is NADH. This is true because NAD+ can accept electrons from an organic molecule.arrow_forwardFe-S center is an oxidoreductive coenzyme present in the electron transport enzyme complex. Which of the following complex(es) do(es) NOT contain this coenzyme? Select one: A. Complex I B. Complex IV C. Complex II D. Complex IIIarrow_forward
- What role does superoxide dismutase play in ameliorating the effects of reactive oxygen species? Options: A. It catalyzes the conversion of superoxide to hydrogen peroxide and O2. B. It generates superoxide by reacting ubisemiquinone with O2. C. It synthesizes superoxide by transferring a free electron on to O2. D. It directly transforms superoxide into water.arrow_forwardWhich of the following organisms cannot perform oxidative phosphorylation? Select one: a. E. coli b. tulips c. white rhinos d. mushrooms e. none of the above Clear my choicearrow_forwardYou create an artificial membrane system, containing a mixture of electron donors and other chemical components. Place the components listed below in the correct functional order (the order they should be for normal function). For each list, circle the actual final electron acceptor for the combination listed-meaning if the electrons cannot be passed along the full list of molecules, where do they stop? a. NADH, Q, Complexes I, Il and III, O2 b. FAD, cytochrome C, Q, Complexes I, II and IV, O2 c. NADH, Q, cytochrome C, Complexes I, IIl and IV, O2 d. Same as "c", but Q activity is blocked e. FADH2, Q, cytochrome C, Complexes II, IIl and IV, O2arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





