
Concept explainers
(a)
Interpretation:
The synthesis of a given compound from
Concept introduction:
The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 23.70P
The formation of the given product from
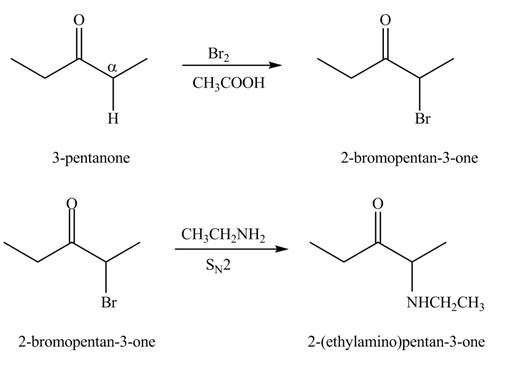
Explanation of Solution
A chemical equation for the given synthesis is shown below.
Figure 1
The formation of a given product by using
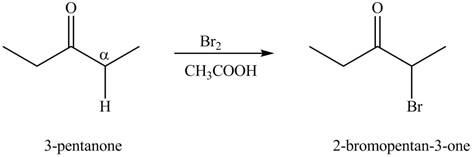
Figure 2
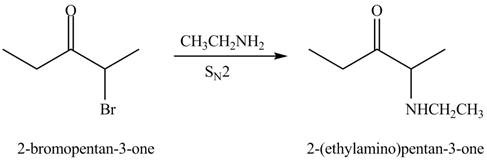
Figure 3
The first step of the reaction involves the bromination of
The formation of the given product from
(b)
Interpretation:
The synthesis of a given compound from
Concept introduction:
The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 23.70P
The synthesis of a given compound from
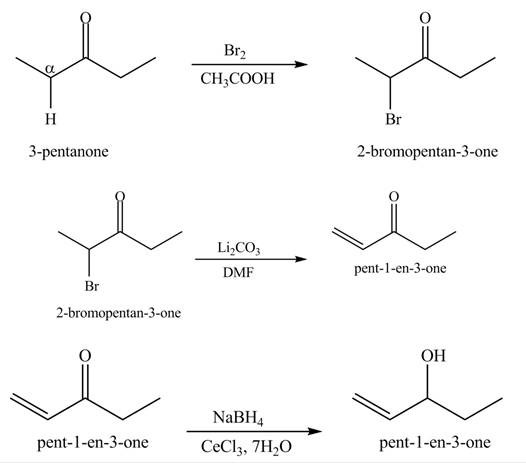
Explanation of Solution
A chemical equation for the given synthesis is shown below.

Figure 4
The formation of the given product by using
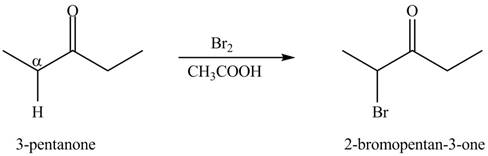
Figure 5
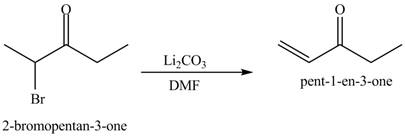
Figure 6
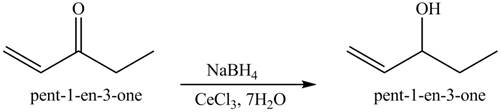
Figure 7
The first step of the reaction involves the bromination of
The synthesis of a given compound from
(c)
Interpretation:
The synthesis of a given compound from
Concept introduction:
The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 23.70P
The synthesis of a given compound from
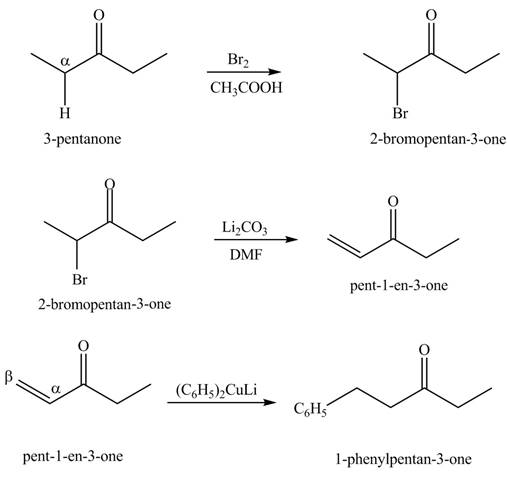
Explanation of Solution
A chemical equation for the given synthesis is shown below.
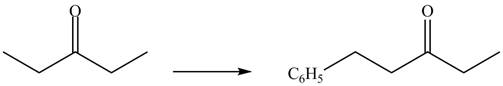
Figure 8
The formation of a given product by using
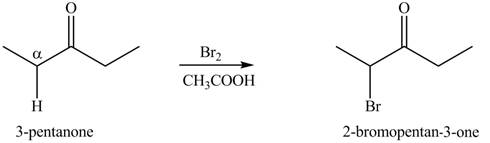
Figure 9
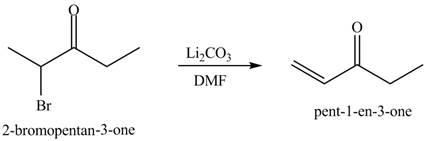
Figure 10
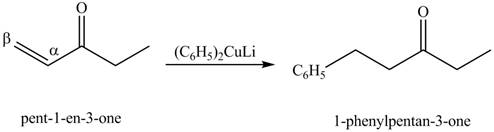
Figure 11
The first step of the reaction involves the bromination of
The synthesis of a given compound from
(d)
Interpretation:
The synthesis of a given compound from
Concept introduction:
The synthesis of the products relies upon the type of reactants and reagents that are used during the reactions. The energy of a target molecule should be low because it increases the stability of a molecule that results in the formation of a molecule with high yield. The reagents perform numerous functions in reactions like proton abstraction, oxidation, reduction, catalysis, and dehydrogenation.
Answer to Problem 23.70P
The synthesis of a given compound from
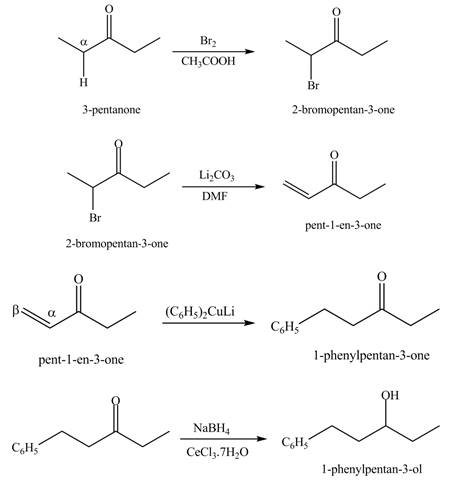
Explanation of Solution
A chemical equation for the given synthesis is shown below.
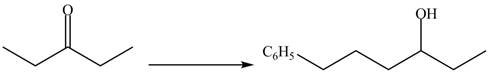
Figure 12
The formation of a given product by using
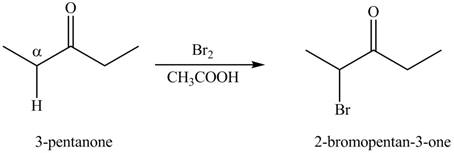
Figure 13
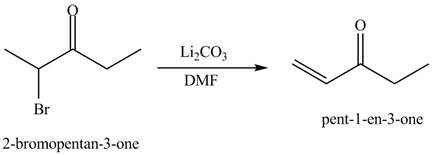
Figure 14
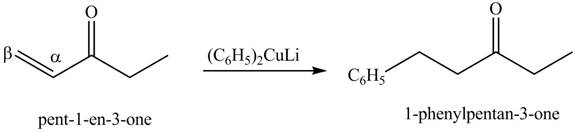
Figure 15
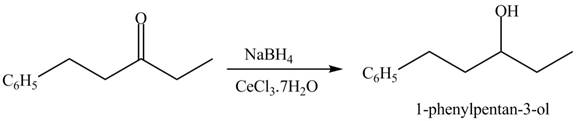
Figure 16
The first step of the reaction involves the bromination of
The synthesis of a given compound from
Want to see more full solutions like this?
Chapter 23 Solutions
Organic Chemistry
- Give the IUPAC name for each alcohol но сн CH,CHCCH,CH,CH3 (CH)½CHCH,CHCH,CH3 b. a. CH,CH,CH,OH HO. с. CH,CH,CH,CH, Draw the products formed when each alcohol is dehydrated with H2SO4. Use the Zaitsev rule to predict the major product when a mixture forms. OH он b. -CHCH,CH3 а. Он с. CH3CHCH2CH,CH;CH3arrow_forwardcdn.fbsbx.com I. What products are formed when each alcohol is oxidized with K,Cr,0,? a. CH,CH,CH,CH,CH,OH OH OH H3C CH3 b. C. II, Give the structure corresponding to each IUPAC name:arrow_forwardDraw the organic products formed when cyclopentene is treated with each reagent. With some reagents, no reaction occurs. a.H2 + Pd-C b.H2 + Lindlar catalyst c.Na, NH3 d.CH3CO3H e.[1] CH3CO3H; [2] H2O, HO− f.[1]OsO4 + NMO; [2] NaHSO3, H2O g.KMnO4, H2O, HO− h.[1] LiAlH4; [2] H2O i. [1] O3; [2] CH3SCH3 j.(CH3)3COOH, Ti[OCH(CH3)2]4, (−)-DET k.mCPBA l.Product in (k); then [1] LiAlH4; [2] H2Oarrow_forward
- Synthesize each compound from cyclohexanol using any other organic or inorganic compounds. a. ہیں۔ بھی مله C. OH d.arrow_forwardSynthesize each compound from benzene. a. SO₂H 19일 g b. 00 이 Harrow_forwardSynthesize each compound from benzene, organic halides with < 5 C's, and any other organic or inorganic reagents.arrow_forward
- Draw the products formed when phenol(C6H5OH) is treated with each reagent. Give an explanation. d. (CH3CH2)2CHCOCl, AlCl3 j. product in (d), then NH2NH2, – OHarrow_forwardDraw out each compound to clearly show what groups are bonded to the carbonyl carbon. Label each compound as a ketone or aldehyde. a. CH 3CH 2CHO b. CH 3CH 2COCH 3 c. (CH 3) 3CCOCH 3 d. (CH 3CH 2) 2CHCHOarrow_forward3. What alcohol is formed when each alkene is treated with H2O in the presence of H2SO4 (as the catalyst)? а. СНЗСН-СНСНЗ b. CH3CH2CH=CH2 С. CH3 CH3arrow_forward
- Give the IUPAC name for each alkyne. a. CH,CH,CH(CH,)C=CCH,CH, d. HC=C-CH(CH2CH3)CH,CH,CH3 g. CH3 e. CH;CH2-C-C=CH CH,CH,CH, b. (CH2),CHC CCH(CH3)2 c. (CH;CH2),CHC=CCH(CH,CH3)CH(CH)CH,CH3 f. CH;CH,C=CCH,C=CCH3 h.arrow_forwardConvert benzene into each compound. You may also use any inorganic reagents and organic alcohols having four or fewer carbons. One step of the synthesis must use a Grignard reagent.arrow_forwardDevise a synthesis of each alkene using a Wittig reaction to form the double bond. You may use benzene and organic alcohols having four or fewer carbons as starting materials and any required reagents. a. CH,CH2CH2CH=CHCH3 b. GgH;CH=CHCH2CH¿CH3 c. (CHa2C=CHCH(CH)2arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





