
(a)
Interpretation:
The reaction of ammonia with nitrous acid is to be completed. Also, the major product formed in the corresponding reaction is to be stated.
Concept introduction:
Answer to Problem 23.65AP
The reaction of ammonia with nitrous acid is shown below.
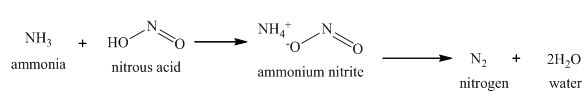
The major product formed is the nitrogen gas.
Explanation of Solution
When ammonia reacts with nitrous acid, diazotization occurs with the formation of ammonium nitrite intermediate which dissociates and liberates nitrogen gas along with the formation of water.
The complete reaction is shown below.

Figure 1
The reaction of ammonia with nitrous acid is shown in Figure 1.
(b)
Interpretation:
The reaction between
Concept introduction:
The conversion of amines into
Answer to Problem 23.65AP
The complete reaction between
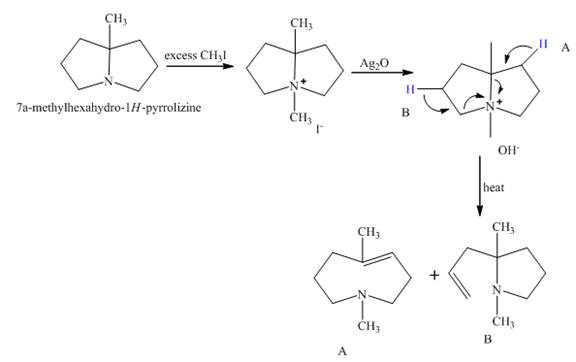
The major product will be compound
Explanation of Solution
Hofmann elimination reaction takes place when

Figure 2
The complete reaction between
(c)
Interpretation:
The reaction between
Concept introduction:
The conversion of amines into alkenes can be achieved by Hoffmann elimination reaction. Amines have poor leaving groups. They react with excess of alkyl halides to form ammonium salts. These ammonium salts undergo
Answer to Problem 23.65AP
The reaction between
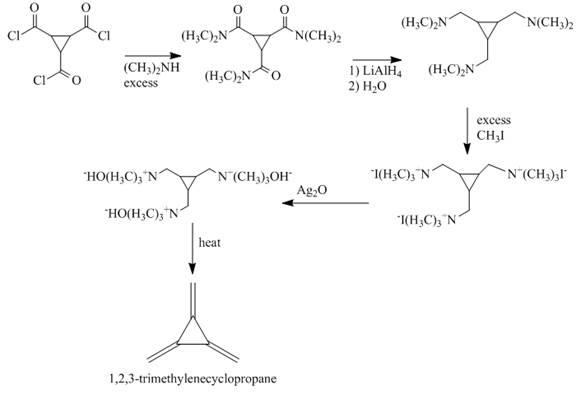
The major product formed is
Explanation of Solution
When

Figure 3
The reaction between
(d)
Interpretation:
The reaction of phenol with hydrochloric acid and sodium nitrite is to be completed. Also, the major product of the corresponding reaction is to be stated.
Concept introduction:
Electrophilic aromatic substitution reaction involves the substitution of an electrophile on an aromatic ring. The
Answer to Problem 23.65AP
The reaction of phenol with hydrochloric acid and sodium nitrite is shown below.
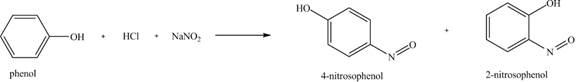
The major product formed is
Explanation of Solution
The compounds

Figure 4
The reaction of phenol with hydrochloric acid and sodium nitrite is shown in Figure 4.
(e)
Interpretation:
The reaction between
Concept introduction:
Amines are the organic compounds that are formed by replacement of hydrogen from ammonia with a substituent. It may be alkyl or aryl group. The formation of diazonium salt from aromatic amines takes place using sodium nitrite and hydrohalide at low temperatures. This process is known as diazotization. Aryl diazonium salts undergo a variety of specific substitution reactions in which the nucleophilic Z group replaces (a very good leaving group) to form corresponding products.
Answer to Problem 23.65AP
The reaction between
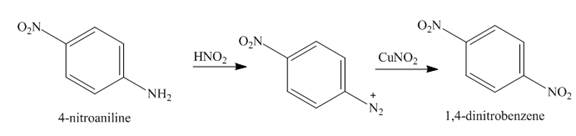
The major product in the corresponding reaction is
Explanation of Solution
When

Figure 5
The reaction between
(f)
Interpretation:
The reaction between butylamine with oxirane is to be completed. The major product of the corresponding reaction is to be stated.
Concept introduction:
Nucleophilic substitution reaction is the reaction in which a nucleophile attacks the electrophilic center and a substituted product is formed. It takes place by the generation of an electrophilic intermediate.
Answer to Problem 23.65AP
The reaction between butylamine with oxirane is shown below.
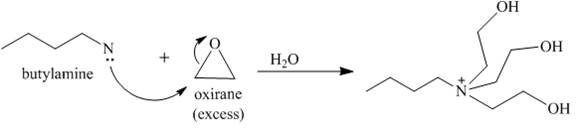
Explanation of Solution
Oxirane is a symmetrical molecule. Therefore, the nucleophile can attack at any carbon. Nucleophilic substitution reaction takes place when butyl amine reacts with oxirane. The lone pair of electrons attacks the carbon atom of the oxirane which opens the ring. Since, the oxirane is in excess, so, the amine will react with oxirane till it’s all the hydrogen groups are replaced by the oxirane to yield a quaternary ammonium derivative as shown below.

Figure 6
The reaction between butylamine with oxirane is show in Figure 6.
(g)
Interpretation:
The reaction of
Concept introduction:
Nucleophilic substitution reaction is the reaction in which a nucleophile attacks the electrophilic center and a substituted product is formed. It takes place by the generation of an electrophilic intermediate.
Answer to Problem 23.65AP
The reaction of
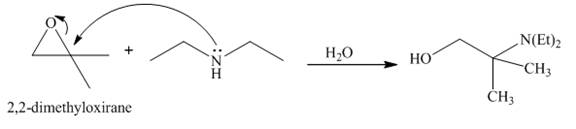
Explanation of Solution
The reaction between

Figure 7
The reaction between
(h)
Interpretation:
The reaction between phthalimide and
Concept introduction:
Gabriel synthesis is a reaction in which a primary amine is formed when a phthalimide anion is alkylated and then undergoes hydrolysis. It is the best method to prepare primary aliphatic amines. Aromatic amines cannot be prepared by this reaction.
Answer to Problem 23.65AP
The reaction between phthalimide and
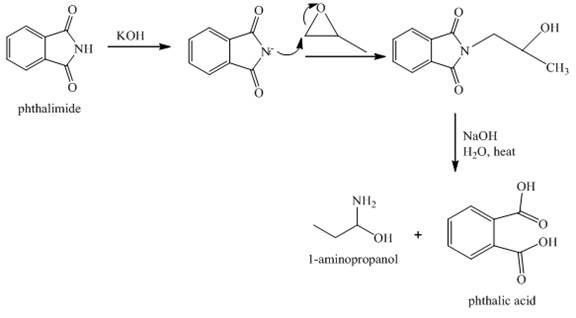
Explanation of Solution
Gabriel phthalimide reaction occurs when phthalimide reacts with

Figure 8
The reaction between phthalimide and
(i)
Interpretation:
The reaction between
Concept introduction:
Nucleophilic substitution reaction is the reaction in which a nucleophile attacks the electrophilic center and a substituted product is formed. It takes place by the generation of an electrophilic intermediate.
Answer to Problem 23.65AP
The nucleophilic substitution reaction between
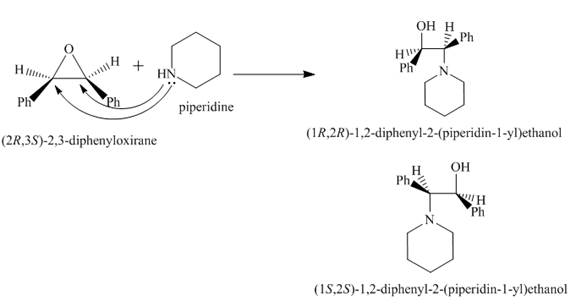
Two enantiomeric products will be formed in equal proportions because the substrate is symmetrical, so, the attack of nucleophile is feasible equally at both the carbon sites in substituted oxirane.
Explanation of Solution
Nucleophilic substitution reaction takes place when

Figure 9
The reaction between
(j)
Interpretation:
The reaction between
Concept introduction:
Nucleophilic substitution reaction is the reaction in which a nucleophile attacks the electrophilic center and a substituted product is formed. It takes place by the generation of an electrophilic intermediate.
Answer to Problem 23.65AP
The reaction between
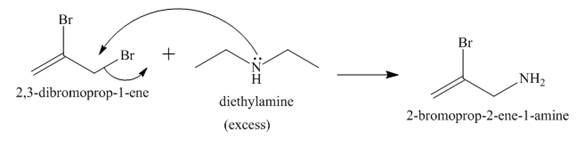
Explanation of Solution
The nucleophilic substitution reaction takes place when diethyl amine reacts with

Figure 10
The reaction between
(k)
Interpretation:
The reaction of nitrobenzene with
Concept introduction:
Catalytic hydrogenation is a reduction process of addition of hydrogen atoms in an alkene or an
Answer to Problem 23.65AP
The reaction of nitrobenzene with
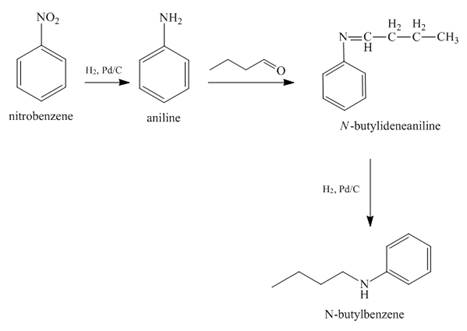
Explanation of Solution
When nitrobenzene is catalytically hydrogenated with
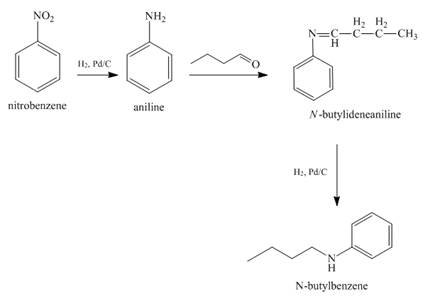
Figure 11
The reaction of nitrobenzene with
Want to see more full solutions like this?
Chapter 23 Solutions
EBK ORGANIC CHEMISTRY
- The saccharide shown here is present in some plant-derived foods. (a) Indicate the anomeric carbon atom(s) in this structure by drawing a circle around the atom(s) or by drawing an arrow pointing to the atom(s). (b) Would this saccharide give a positive result with Benedict’s reagent? Why or why not? (c) Would this saccharide give a positive result with Barfoed’s reagent? Why or why not? (d) Would this saccharide give a positive result with Seliwanoff’s reagent? Why or why not? (e) In a separate set of experiments, the saccharide solution was treated with a reagent that breaks glycosidic bonds. After this treatment, would any of the three assays give different results? Be sure to indicate which assay results would be different and give a reason.arrow_forwardGive the structure of compound A.arrow_forward(a) Why are alkyl halides insoluble in water? (b) Why is Butan-l-ol optically inactive but Butan-2-ol is optically active? (c) Although chlorine is an electron withdrawing group, yet it is ortho-, Para- directing in electrophilic aromatic substitution reaction. Why?arrow_forward
- (A) Provide the major organic product for the reaction below (B) Would the product be optically active of optically in active?arrow_forwardCompound F may be synthesised by the method attached Draw the isomer of compound B and explain which one would be the major product and why.arrow_forwardAccount for the following:(i) Aniline does not give Friedel-Crafts reaction.(ii) Ethylamine is soluble in water whereas aniline is not.(iii) pKb of methylamine is less than that of aniline.arrow_forward
- Provide the appropriate reagents or product in the following examples..arrow_forwardHow would you account for the following :(a) Electrophilic susbstitution in case of aromatic amines takes place more readily than benzene.(b) Ethanamide is a weaker base than ethanamine.arrow_forward(i) Explain why a high reaction temperature favours elimination reactions, instead of substitution reactions. (ii) Explain why polar aprotic solvents favour Sn2 reaction but not favour SN1 reaction.arrow_forward
- Rank the basicity of the following sets of compounds. And give explanation.arrow_forwardthe following reaction scheme leads to the formation of compound C. give the structure of the final product C and of the intermediate products A and B and justify, using the mechanism, the formation of the product A. Give the serereochemistry of the final product obtainedarrow_forwardSafrole, which is isolated from sassafras (Problem 21.33), can be converted to the illegal stimulant MDMA (3,4-methylenedioxymethamphetamine, "Ecstasy") by a variety of methods. (a) Devise a synthesis that begins with safrole and uses a nucleophilic substitution reaction to introduce the amine. (b) Devise a synthesis that begins with safrole and uses reductive amination to introduce the amine. NHCH9 MDMA safrolearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





