
Concept explainers
The fatty acid composition of three triacylglycerols (A, B, and C) is reported below. Predict which one has the highest melting point. Which one do you expect to be liquid (oil) at room temperature? Explain.
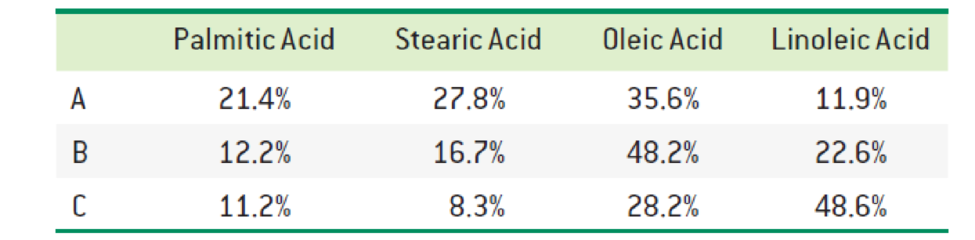
Interpretation:
The triacylglycerols with highest melting point and fatty acid expected to be liquid at room temperature has to be identified.
Concept Introduction:
Triacylglycerol:
It is also known as triglyceride, an ester composed of glycerol and long chains of acyl fatty acids.
Explanation of Solution
The fatty acid composition A has the highest melting point and B and C are expected to be at low melting point.
Since the composition A contains
The triacylglycerols with highest melting point and fatty acid expected to be liquid at room temperature was identified.
Want to see more full solutions like this?
Chapter 23 Solutions
FUND.OF GEN CHEM CHAP 1-13 W/ACCESS
- Predict the order of the melting points of triglycerides containing fatty acids, as follows: From lowest to highest melting point Drag and drop your selection from the following list to complete the answer: linolenic, lauric, palmitic palmitic, palmitic, stearic palmitoleic, linoleic, oleicarrow_forwardRank the following in order of increasing solubility in water (assuming that all are made with the same fatty acids): triglycerides, diglycerides, and monoglycerides. Explain your answer.arrow_forwardBriefly describe the structure of a triglyceride. What are the differences between saturated, unsaturated, monounsaturated, and polyunsaturated fats? no handwritten answers please.arrow_forward
- Rank the following in order of increasing solubility in water (assuming that all are made with the same fatty acids): (a) triglycerides, (b) diglycerides, and (c) monoglycerides. Explain your answer in 1-2 sentences.arrow_forwardSamples of three different triglycerides, A, B, and C, were tested to determine the melting point of each one. The results of the tests are shown in the graph. The length of the fatty acids in A, B, and C is the same. Which of the three triglycerides is likely to have the fewest double bonds in the fatty acid side chains? triglyceride C There is no way of knowing based on the information available triglyceride A triglyceride Barrow_forwardWhich of the following is NOT TRUE about unsaturated fatty acids?arrow_forward
- Samples of three different triglycerides, A, B, and C, were tested to determine the melting point of each one. The results of the tests are shown in the graph. The length of the fatty acids in A, B, and C is the same. Which of the three triglycerides is likely to have the most saturated fatty acid side chains? triglyceride C triglyceride B triglyceride A There is no way of knowing based on the information availablearrow_forwardGive the lipid number for the following fatty acid:arrow_forwardWhich of the following fatty acids is/are most likely responsible for the liquid state of oils? COOH A A B c^ C COOH ✓ COOHarrow_forward
- Construct and name the structure of a monoglyceride Two diglycerides (positions 1,2 and 1,3) and two triglycerides (one mixed and one simple) using the following fatty acids. Myristic Oleic Laurico ice = oil G Fatty acid Fatty acid Fatty acidarrow_forwardDraw the skeletal structure of the products formed when the given triacylglycerol is hydrolyzed with water in the presence of sodium hydroxide. You may draw the structures in any arrangement that you like, so long as they aren't touching.arrow_forwardDraw structures of glyceryl tripalmitate and glyceryl trioleate. Which would you expect to have a higher melting point?arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





