
Concept explainers
Devise a synthesis of each labeled compound using
a. 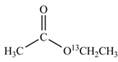 b.
b. 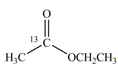 c.
c. 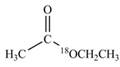 d.
d. 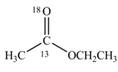
(a)
Interpretation:
The synthesis of given compound by the use of
Concept introduction:
The replacement or substitution of one functional group with another functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons is known as a nucleophile. In nucleophilc acyl substitution reaction, a nucleophile takes the position of a leaving group.
Answer to Problem 22.75P
The synthesis of given compound by the use of
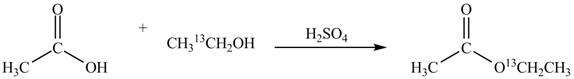
Explanation of Solution
An ester is formed by the reaction of carboxylic acid with alcohol. This reaction is known as Fischer Esterification. During this reaction, bond formation between carbon and oxygen takes place and water removes as a byproduct. In the synthesis of given compound, acetic acid is used as the source of unlabeled carbon atom and
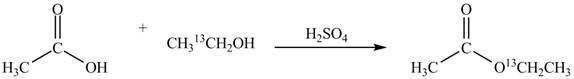
Figure 1
The synthesis of given compound by the use of
(b)
Interpretation:
The synthesis of given compound by the use of
Concept introduction:
The replacement or substitution of one functional group with another functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons is known as a nucleophile. In nucleophilc acyl substitution reaction, a nucleophile takes the position of a leaving group.
Answer to Problem 22.75P
The synthesis of given compound by the use of
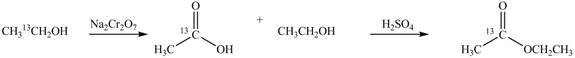
Explanation of Solution
An ester is formed by the reaction of carboxylic acid with alcohol. This reaction is known as Fischer Esterification. During this reaction, bond formation between carbon and oxygen takes place and water removes as a byproduct. In the synthesis of given compound,
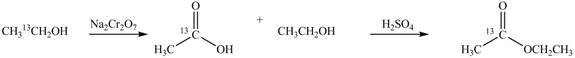
Figure 2
The synthesis of given compound by the use of
(c)
Interpretation:
The synthesis of given compound by the use of
Concept introduction:
The replacement or substitution of one functional group with another functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons is known as a nucleophile. In nucleophilc acyl substitution reaction, a nucleophile takes the position of a leaving group.
Answer to Problem 22.75P
The synthesis of given compound by the use of
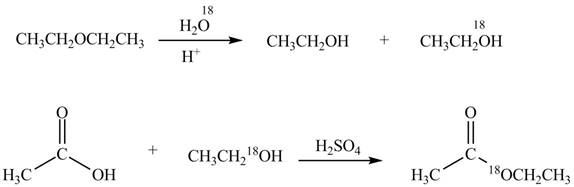
Explanation of Solution
An ester is formed by the reaction of carboxylic acid with alcohol. This reaction is known as Fischer Esterification. During this reaction, bond formation between carbon and oxygen takes place and water removes as a byproduct. In the synthesis of given compound, diethylether is first hydrolyzed to give

Figure 3
The synthesis of given compound by the use of
(d)
Interpretation:
The synthesis of given compound by the use of
Concept introduction:
The replacement or substitution of one functional group with another functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons is known as a nucleophile. In nucleophilc acyl substitution reaction, a nucleophile takes the position of a leaving group.
Answer to Problem 22.75P
The synthesis of given compound by the use of
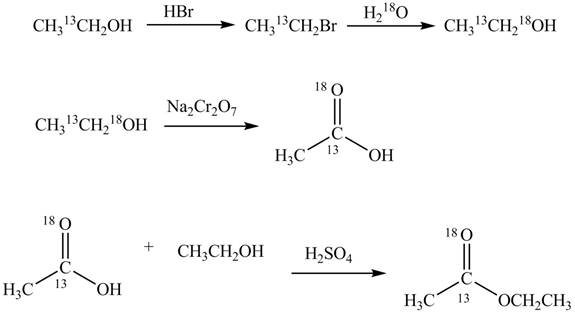
Explanation of Solution
An ester is formed by the reaction of carboxylic acid with alcohol. This reaction is known as Fischer Esterification. During this reaction, bond formation between carbon and oxygen takes place and water removes as a byproduct. In the synthesis of given compound,
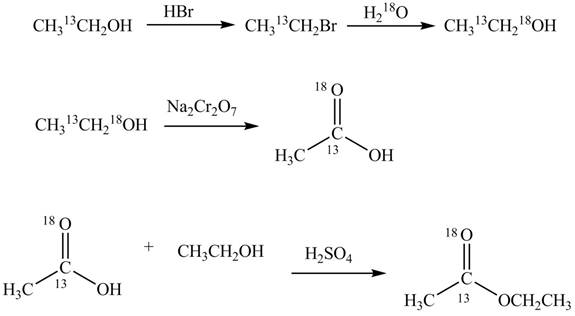
Figure 4
The synthesis of given compound by the use of
Want to see more full solutions like this?
Chapter 22 Solutions
Organic Chemistry-Package(Custom)
- Devise a synthesis of each compound from benzene. You may use alcohols with one or two carbons and any inorganic reagents. a. qe Br H Br b. C. N OHarrow_forwardIdentify the major organic product(s) of the following reaction. x A. B. 1) KOH D. ه (2 مسلسل موسلی yasarrow_forwardSynthesize each compound from acetylene. You may use any other organic or inorganic reagents. C. CH,CH,CH,CH, CHO d. CH3CH, CHỖ CHarrow_forward
- Select the carbonyl compound which can form only a single enol via tautomerization. A. B. H C. D.arrow_forward16.69 Use the reactions in this chapter along with those learned in Chapters 11 and 12 to synthesize each compaund. You may use benzene, acetylene (HC=CH), ethanol, ethylene oxide, and any inorganic reagents. a. b. CI он C ON d. NO2arrow_forward2. How many different ß-hydroxyaldehydes and ß-hydroxyketones, including constitutional isomers and stereoisomers, are formed upon treatment of a mixture of acetone and benzaldehyde with base? a. b. 2 c. 3 d. 4arrow_forward
- Devise a synthesis of each compound using 1-bromobutane (CH3CH2CH2CH2Br) as the only organic starting material. You may use any other inorganic reagents.arrow_forwardDevise a synthesis of each compound from phenol (C6H5OH) and any other organic or inorganic reagents.arrow_forwardWhat is the major organic product obtained from the following reaction? A. B. CI لله مللی حمله C. D. فيarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





