
Concept explainers
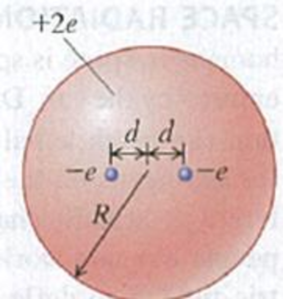
Thomson’s Model of the Atom, Continued. Using Thomson’s (outdated) model of (he atom described in Problem 22.50, consider an atom consisting of two electrons, each of charge −e, embedded in a sphere of charge +2e and radius R. In equilibrium, each electron is a distance d from the center of the atom (Fig. P22.51). Find the distanced in terms of the other properties of the atom.
Figure P22.51
Learn your wayIncludes step-by-step video

Chapter 22 Solutions
University Physics with Modern Physics (14th Edition)
Additional Science Textbook Solutions
Physics: Principles with Applications
Physics for Scientists and Engineers: A Strategic Approach with Modern Physics (4th Edition)
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Lecture- Tutorials for Introductory Astronomy
- The HCl molecule has a dipole moment of about 3.4x10-30 C·m. The two atoms areseparated by about 1.0x10-10 m.a. What is the net charge on each atom? Is this equal to an integer multiple of e? Ifnot, explain.b. What maximum torque would this dipole experience in a 2.5x104 N/C electricfield?c. How much energy would be needed to rotate one molecule 45° from itsequilibrium (lowest potential energy) position?arrow_forwardSolar wind is a flow of heavily charged ions emanating from the Sun’s corona. Spectroscopic analysis suggests it contains a density of approximately 5.0 electrons per cubic centimeter (cm3). Given that the elementary charge ? ≈ 1.6 × 10−19 ? and using the combined average of your measured values of Q 0.0375nC, determine the charge density of a cubic meter (m3) of solar wind sample as a function of Qarrow_forwardThe net charge of an object can be found be summing the individual charges. Since charge is quantized, all net charges are just an integer multiple of the elementary charge. q =n e where q is the net charge and n is an integer. For example, sodium can be a +1 ion and chlorine can be a -1 ion. What does this mean concerning their charge and structure?arrow_forward
- Suppose a capacitor consists of two coaxial thin cylindrical conductors. The inner cylinder of radius ra has a charge of +Q, while the outer cylinder of radius rp has charge -Q. The electric field E at a radial distance r from the central axis is given by the function: E = ae-T/ao + B/r + bo where alpha (a), beta (8), ao and bo are constants. Find an expression for its capacitance. First, let us derive the potential difference Vah between the two conductors. The potential difference is related to the electric field by: Vab = Edr = - Edr Calculating the antiderivative or indefinite integral, Vab = (-aage-r/a0 + B + bo By definition, the capacitance Cis related to the charge and potential difference by: C = Evaluating with the upper and lower limits of integration for Vab, then simplifying: C= Q/( (e-rb/ao - eTalao) + B In( ) + bo ( ))arrow_forwardProblem 12: The classic Millikan oil drop experiment was the first to obtain an accurate measurement of the charge on an electron. In it, oil drops were suspended against the gravitational force by a vertical electric field. microscope ©theexpertta.com Given that the oil drop is 0.85 µum in radius and has a density of 925 kg/m³, find the weight of the drop in N. W = sin() cos() tan() 7 8 9 HOME cotan() asin() acos() sinh() 4 5 6 atan() acotan() 1 2 3 cosh() tanh() cotanh() + - END ODegrees O Radians VO BACKSPACE DEL CLEAR Submit Hint Feedback I give up! If the drop has a single excess electron, find the electric field strength needed to balance its weight.arrow_forwardAn electron is fired with a v = 10° m/s into the space between two charged plates in the direction shown in the figure below. Determine the trajectory and coordinates of the electron so that the electron hits one of its plates. The charge and mass of the electron are e = 1,6x10-19C and m = 9,1x10-31 Kg. The electric field intensity in the space is 1 KN/C and the plate distance is 0.5 cm. + +++++++++ +++++ v = 10°m/sarrow_forward
- Suppose a capacitor consists of two coaxial thin cylindrical conductors. The inner cylinder of radius ra has a charge of +Q, while the outer cylinder of radius rp has charge -Q. The electric field E at a radial distance r from the central axis is given by the function: E = aer/ao + B/r + bo %3D where alpha (a), beta (B), ao and bo are constants. Find an expression for its capacitance. First, let us derive the potential difference Vab between the two conductors. The potential difference is related to the electric field by: Vab = | S"Edr= - [ *Edr Calculating the antiderivative or indefinite integral, Vab = (-aage-r/ao + B + bo By definition, the capacitance C is related to the charge and potential difference by: C = Evaluating with the upper and lower limits of integration for Vab, then simplifying: C = Q/( (e-rb/ao - eralao) + B In( ) + bo ( ))arrow_forwardr -e Physical constants (A) What is the radius of the orbit with n = 4? (B) What is the speed of the orbit with n = 4? The old Bohr model of the hydrogen atom was based on... (1) the assumption that the electron travels on a circle h = 6.626 x 10-34 Js; -28 (A) (in m) Bohr Model mv² r mvr = n and obeys Newton's second law, and (2) the hypothsis that angular momentum is quantized. For the Bohr model, Ke² = 2.307 x 107 Jm; 31 m9.11 x 10 kg. (B) (in m/s) Ke² 12. (1) (2) OA: 2.034x10-10 OB: 2.706x10-10OC: 3.598x10-10 OD: 4.786x10-10 OE: 6.365x10-10 OF: 8.466x10-10 OG: 1.126x109 OH: 1.497x109 OA: 8.533x104 OB: 1.237x105 OC: 1.794x105 OD: 2.601x105 OE: 3.772x105 OF: 5.470x105 OG: 7.931x105 OH: 1.150x106arrow_forwardAn carly model of the atom, proposed by Rutherford after his discovery of the atomic nucleus, had a positive point charge +Ze (the nucleus) at the center of a sphere of radius R with uniformly distributed negative charge -Ze. Z is the atomic number, the number of protons in the nucleus and the number of clectrons in the negative sphere. (a) Show that the clectric ficld inside this atom is Ze Ein = 1 r2 R3 (b) What is E at the surface of the atom? Is this the expected value? Explain. (c) A uranium atom has Z 92 and R = 0.10 nm. What is the clectric ficld strength %3D at r =arrow_forward
- 2.7. A water molecule is a permanent dipole with a dipole moment of 6.186 × 10-30 Cm. Treat the axis of the dipole as the z axis and calculate the torque on a the molecule if it is in an 8500 N/C electric field aligned in the xz plane at an angle of 42° relative to the x axis.arrow_forwardFind the total charge contained in the 2 [cm] length of the cylindrical electron beam that is showed at the instant represented in the Figure, which is between z = 2 [cm] and z = 4 [cm], and has a radius of 1 [cm]. At that instant, the bulk density charge is assumed to be: p(r, z) = -5-10-6-e-10³rz X Zarrow_forward2. . 4.6325 x 1015 protons and 4.6325 x 1015 clectrons; the charge on this object is Coulombs. Solution and Answer:arrow_forward
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning

