
Concept explainers
Interpretation:
To locate the two nonmetals form more than four bonds on the periodic table
Concept introduction:
A table in which the chemical elements are arranged in order by their
Nonmetals have a low melting point and boiling points. The nonmetals are dividing into two parts. They are reactive nonmetals and noble gases. The nonmetals present in the periodic table: hydrogen, carbon, nitrogen, oxygen, phosphorous, sulfur, selenium, and the noble gases.
Interpretation:
To locate the nonmetals form maximum four bonds on the periodic table
Concept introduction:
Nonmetals have a low melting point and boiling points. The nonmetals are dividing into two parts. They are reactive nonmetals and noble gases. The nonmetals present in the periodic table: hydrogen, carbon, nitrogen, oxygen, phosphorous, sulfur, selenium, and the noble gases. A table in which the chemical elements are arranged in order by their atomic number, an electronic configuration is said to be a periodic table. In general, the elements in a vertical column are said to be groups and rows are said to be periods. Elements in the same groups generally possess similar chemical properties.
Interpretation:
To locate the two nonmetals form oxides that are gases at
Concept introduction:
Nonmetals have a low melting point and boiling points. The nonmetals are dividing into two parts. They are reactive nonmetals and noble gases. The nonmetals present in the periodic table: hydrogen, carbon, nitrogen, oxygen, phosphorous, sulfur, selenium, and the noble gases. A table in which the chemical elements are arranged in order by their atomic number, an electronic configuration is said to be a periodic table. In general, the elements in a vertical column are said to be groups and rows are said to be periods. Elements in the same groups generally possess similar chemical properties.
Interpretation:
To locate the nonmetal that forms an oxide that is solid 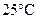
Concept introduction:
Nonmetals have a low melting point and boiling points. The nonmetals are dividing into two parts. They are reactive nonmetals and noble gases. The nonmetals present in the periodic table: hydrogen, carbon, nitrogen, oxygen, phosphorous, sulfur, selenium, and the noble gases. A table in which the chemical elements are arranged in order by their atomic number, an electronic configuration is said to be a periodic table. In general, the elements in a vertical column are said to be groups and rows are said to be periods. Elements in the same groups generally possess similar chemical properties.
Want to see the full answer?
Check out a sample textbook solution
Chapter 22 Solutions
Chemistry (7th Edition)
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





