
Concept explainers
Interpretation:
The chirality centers in aldotetrose and ketopentose are to be calculated and the stereoisomers for each general case are to be determined.
Concept Introduction:
Carbohydrates are categorized mainly as monosaccharides, disaccharides, and polysaccharides. Monosaccharides are single sugar units, mainly glucose and fructose, disaccharides are two sugar units, such as sucrose, and polysaccharides are more than two sugar units, such as starch and cellulose.
Monosaccharides containing 3-carbon atoms are called triose, 4-carbon atoms called tetrose, 5-carbon atoms called pentose, and so on.
In chiral molecules, carbon atom having four nonidentical substituent groups is called the chirality center of that molecule. Chirality center may also be called stereocenter, which signifies any point in the molecule where the interchanging of any two groups may lead to stereoisomers. The carbon of a carbohydrate can be considered as chiral when the carbon has all four different substituents attached to it.
The stereoisomers are calculated as follows:
Here,
Answer to Problem 1PP
Solution:
a) Two
b) Two
c) Four
Explanation of Solution
a) The aldotetrose
A monosaccharide containing four carbon atoms is called a tetrose. An aldotetrose is a monosaccharide that contains
The structure of aldotetrose is as follows:
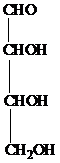
The carbon atom attached to four different groups is chiral carbon. The chiral center in ketopentose is marked by * as follows:
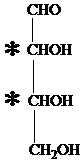
Hence, an aldotetrose has two chirality centers.
b) The ketopentose
A monosaccharide containing five carbon atoms is called a pentose. A pentose containing a keto group is called a ketopentose.
The structure of ketopentose is as follows:
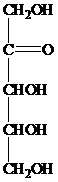
The carbon atom attached to four different groups is chiral carbon. The chiral center in ketopentose is marked by * as follows:
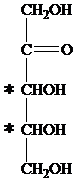
Hence, a ketopentose has two chirality centers.
c) The number of stereoisomers that will be expected from each general structure
Stereoisomers of a molecule have the same molecular formula, but different arrangement of atoms in space. Stereoisomers are different from enantiomers as they are not mirror images of each other, while enantiomers are mirror images of one another.
The compounds aldotetrose and ketopentose have two sets of enantiomers. The number of stereoisomers is calculated as:
Substitute 2 for
Hence, they will have four stereoisomers for each general structure.
Want to see more full solutions like this?
Chapter 22 Solutions
EBK ORGANIC CHEMISTRY
- (a) Which of the d-aldopentoses will give optically active aldaric acids on oxidation with HNO3 ?(b) Which of the d-aldotetroses will give optically active aldaric acids on oxidation with HNO3 ?(c) Sugar X is known to be a d-aldohexose. On oxidation with HNO3, X gives an optically inactive aldaric acid. WhenX is degraded to an aldopentose, oxidation of the aldopentose gives an optically active aldaric acid. Determine thestructure of X.(d) Even though sugar X gives an optically inactive aldaric acid, the pentose formed by degradation gives an opticallyactive aldaric acid. Does this finding contradict the principle that optically inactive reagents cannot form opticallyactive products?(e) Show what product results if the aldopentose formed from degradation of X is further degraded to an aldotetrose.Does HNO3 oxidize this aldotetrose to an optically active aldaric acid?arrow_forwardCoibacin B (shown below) is a natural product that exhibits potent anti-inflammatory activity and potential activity in the treatment of leishmaniasis, a disease caused by certain parasites (Org. Lett. 2012, 14, 3878-3881): (a) Assign the configuration (R or S) of each chirality center (labeled A to C) in coibacin B. (b) Identify the number of possible stereoisomers for this compound, assuming that the geometry of the alkenes are fixed. Choices are given below and write the CAPITAL LETTER of your choice. A. 2 В. 4 С. 8 D. 16 ANSWERS: (a) A. В. C. (b)arrow_forwardCompound A is a D-aldopentose that can be oxidized to an optically inactive aldaric acid B. On Kiliani-Fischer chain extension, A is converted into C and D; C can be oxidized to an optically active aldaric acid E, but D is oxidized to an optically inactive aldaric acid F. What is the structure of compound F? • Use the wedge/hash bond tools to indicate stereochemistry where it exists. You do not have to explicitly draw H atoms. If a group is achiral, do not use wedged or hashed bonds on it. • Show stereochemistry in a meso compound. • Do not include lone pairs in your answer. They will not be considered in the grading.arrow_forward
- Draw the following monosaccharides, using chair conformations for the pyranoses andHaworth projections for the furanoses.(a) a-d-mannopyranose (C2 epimer of glucose)(b) b-d-galactopyranose (C4 epimer of glucose)(c) b-d-allopyranose (C3 epimer of glucose)(d) a-d-arabinofuranose(e) b-d-ribofuranose (C2 epimer of arabinose)arrow_forwardIdentify the hemiacetal function in lactose and show mechanistically how the stereochemistry at that carbon can easily invert, especially in the presence of acids and bases.arrow_forwardHow many chiral centers are in B-d-glucopyranose and a-D-galactopyranose? How many stereoisomers of these two aldohexoses can theoretically be drawn?arrow_forward
- A key step in the synthesis of the narcotic analgesic meperidine (trade name Demerol) is the conversion of phenylacetonitrile to X. (a) What is the structure of X? (b) What reactions convert X to meperidine?arrow_forwardThe cyclic hemiacetal is more stable than the open-chain form, so very little of the open-chain form is present atequilibrium. Will an aqueous solution of glucose reduce Tollens reagent and give a positive Tollens test? Explain.arrow_forwardTreatment of -D-glucose with methanol in the presence of an acid catalyst converts it into a mixture of two compounds called methyl glucosides (Section 25.3A). In these representations, the six-membered rings are drawn as planar hexagons. (a) Propose a mechanism for this conversion and account for the fact that only the OH on carbon 1 is transformed into an OCH3 group. (b) Draw the more stable chair conformation for each product. (c) Which of the two products has the chair conformation of greater stability? Explain.arrow_forward
- Predict the products formed when the following sugars react with excess acetic anhydrideand pyridine.(a) a-d-glucopyranosearrow_forward31. Which of the following statements about cholesterol is not correct? CH. HO Cholesterol 16 (a) Cholesterol is a steroid that contains a tetracyclic ring system. (b) Cholesterol is a steroid that contains 8 chiral carbons and can form 28 or 256 stereoisomers. (c) Each atom or group attached to a ring-junction carbon (i.e., carbons a-e) is in a trans or axial position. Because of this the tetracyclic ring system is mostly flat. (d) Cholesterol is used to synthesized vitamin D, bile acids, sex hormones, and adrenocorticoid hormones. (e) Cholesterol is not found in the cell membranes of animals.arrow_forward(d) Draw the structure of the expected product when monosaccharide B undergo mutarotation upon dissolving in water in the presence of Tollens reagent (AGNO3, NHẠOH). он OH O. OH OH OH monosaccharide Barrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
