
To find if the absolute value of the freezing point of chlorine greater than or less than the absolute value of the freezing point of nitrogen.
Answer to Problem 54IP
The absolute value of the freezing point of chlorine is less than the absolute value of the freezing point of nitrogen.
Explanation of Solution
Given information:
The table shows the freezing point of various elements:
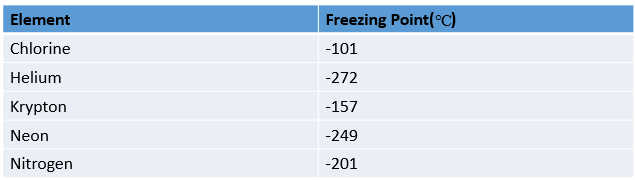
The absolute value of the freezing point of chlorine is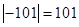
The absolute value of the freezing point of nitrogen is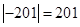
Show the absolute value of the freezing point of chlorine and nitrogen using a number line:
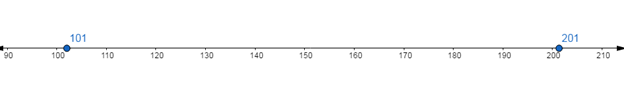
Since, 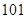 is to the left of
is to the left of
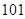 is less than
is less than
Hence, the absolute value of the freezing point of chlorine is less than the absolute value of the freezing point of nitrogen.
Chapter 2 Solutions
Glencoe Math Accelerated, Student Edition
Additional Math Textbook Solutions
Calculus and Its Applications (11th Edition)
University Calculus: Early Transcendentals (3rd Edition)
Single Variable Calculus: Early Transcendentals (2nd Edition) - Standalone book
Calculus: Early Transcendentals (3rd Edition)
Calculus: Early Transcendentals (2nd Edition)
 Calculus: Early TranscendentalsCalculusISBN:9781285741550Author:James StewartPublisher:Cengage Learning
Calculus: Early TranscendentalsCalculusISBN:9781285741550Author:James StewartPublisher:Cengage Learning Thomas' Calculus (14th Edition)CalculusISBN:9780134438986Author:Joel R. Hass, Christopher E. Heil, Maurice D. WeirPublisher:PEARSON
Thomas' Calculus (14th Edition)CalculusISBN:9780134438986Author:Joel R. Hass, Christopher E. Heil, Maurice D. WeirPublisher:PEARSON Calculus: Early Transcendentals (3rd Edition)CalculusISBN:9780134763644Author:William L. Briggs, Lyle Cochran, Bernard Gillett, Eric SchulzPublisher:PEARSON
Calculus: Early Transcendentals (3rd Edition)CalculusISBN:9780134763644Author:William L. Briggs, Lyle Cochran, Bernard Gillett, Eric SchulzPublisher:PEARSON Calculus: Early TranscendentalsCalculusISBN:9781319050740Author:Jon Rogawski, Colin Adams, Robert FranzosaPublisher:W. H. Freeman
Calculus: Early TranscendentalsCalculusISBN:9781319050740Author:Jon Rogawski, Colin Adams, Robert FranzosaPublisher:W. H. Freeman
 Calculus: Early Transcendental FunctionsCalculusISBN:9781337552516Author:Ron Larson, Bruce H. EdwardsPublisher:Cengage Learning
Calculus: Early Transcendental FunctionsCalculusISBN:9781337552516Author:Ron Larson, Bruce H. EdwardsPublisher:Cengage Learning





