
Introduction To General, Organic, And Biochemistry
12th Edition
ISBN: 9781337571357
Author: Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21, Problem 51P
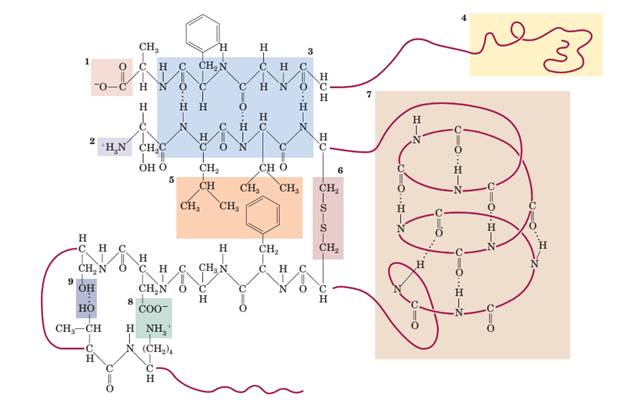
22-63 Identify the primary, secondary, and tertiary structures in the numbered boxes:
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
We know that trivalent cations (Cr3+, Mn3+, V3+) with a large difference between octahedral and tetrahedral EECC, form exclusively normal spinels. Bivalent cations (Ni2+ and Cu2+) with high EECC, form inverse spinels. Is this statement correct?
(b) Draw the product A that would be formed through the indicated sequence of steps from the given starting material.
MeO
(1) Br₂, hv
(2) NaOEt,
EtOH, A
(3) BH3:THF
(4) H₂O2, HO-
B
H₂C.
CH₂
OH
Edit
Small changes in
secondary; tertiary
primary; secondary
primary; tertiary
tertiary; secondary
protein structure may lead to big changes in
protein structures.
Chapter 21 Solutions
Introduction To General, Organic, And Biochemistry
Ch. 21.1 - Prob. 21.1QCCh. 21.2 - Prob. 21.2QCCh. 21.3 - What is the overall charge (+,0, or ) for the...Ch. 21.4 - Problem 22-1 Show how to form the dipeptide...Ch. 21.5 - Prob. 21.5QCCh. 21.7 - Prob. 21.6QCCh. 21.8 - Prob. 21.7QCCh. 21.8 - Prob. 21.8QCCh. 21.10 - Prob. 21.9QCCh. 21 - 22-5 What are the functions of (a) ovalbumin and...
Ch. 21 - 22-6 The members of which class of proteins are...Ch. 21 - 22-7 What is the function of an immunoglobulin?Ch. 21 - Prob. 4PCh. 21 - 22-9 What is the difference in structure between...Ch. 21 - 22-10 Classify the following amino acids as...Ch. 21 - 22-11 Which amino acid has the highest percentage...Ch. 21 - Prob. 8PCh. 21 - Prob. 9PCh. 21 - Prob. 10PCh. 21 - Prob. 11PCh. 21 - Prob. 12PCh. 21 - 22-17 What are the similarities and differences in...Ch. 21 - 22-18 Draw the structures of L- and D-valine.Ch. 21 - Prob. 15PCh. 21 - 22-20 Show how alanine, in solution at its...Ch. 21 - 22-21 Explain why an amino acid cannot exist in an...Ch. 21 - 22-22 Draw the structure of valine at pH 1 and at...Ch. 21 - Prob. 19PCh. 21 - 22-24 Draw the most predominant form of histidine...Ch. 21 - 22-25 Draw the most predominant form of lysine at...Ch. 21 - Prob. 22PCh. 21 - 22-27 Show by chemical equations how alanine and...Ch. 21 - 22-28 A tetrapeptide is abbreviated as DPKH. Which...Ch. 21 - 22-29 Draw the structure of a tripeptide made of...Ch. 21 - 22-30 (a) Use the three-letter abbreviations to...Ch. 21 - 22-31 A polypeptide chain is made of alternating...Ch. 21 - Prob. 28PCh. 21 - 22-33 Which of the three functional groups on...Ch. 21 - Prob. 30PCh. 21 - 22-35 Why is histidine considered a basic amino...Ch. 21 - Prob. 32PCh. 21 - Prob. 33PCh. 21 - 22-38 Why does proline not absorb light at 280 nm?Ch. 21 - Prob. 35PCh. 21 - Prob. 36PCh. 21 - Prob. 37PCh. 21 - 22-42 (a) How many atoms of the peptide bond lie...Ch. 21 - 22-43 (a) Draw the structural formula of the...Ch. 21 - 22-44 How can a protein act as a buffer?Ch. 21 - 22-45 Proteins are least soluble at their...Ch. 21 - 22-46 How many different tripeptides can be made...Ch. 21 - 22-47 How many different tetrapeptides can be made...Ch. 21 - 22-48 How many amino acid residues in the A chain...Ch. 21 - 22-49 Based on your knowledge of the chemical...Ch. 21 - Prob. 46PCh. 21 - 22-59 What is the effect of salt bridges on the...Ch. 21 - Prob. 48PCh. 21 - 22-61 Polyglutamic acid (a polypeptide chain made...Ch. 21 - 22-62 Distinguish between intermolecular and...Ch. 21 - 22-63 Identify the primary, secondary, and...Ch. 21 - 22-64 If both cysteine residues on the B chain of...Ch. 21 - 22-65 (a) What is the difference in the quaternary...Ch. 21 - Prob. 54PCh. 21 - Prob. 55PCh. 21 - Prob. 56PCh. 21 - Prob. 57PCh. 21 - Prob. 58PCh. 21 - 22-71 Which amino acid side chain is most...Ch. 21 - 22-72 What does the reducing agent do in...Ch. 21 - 22-73 Silver nitrate is sometimes put into the...Ch. 21 - 22-74 Why do nurses and physicians use 70% alcohol...Ch. 21 - Prob. 63PCh. 21 - (Chemical Connections 21B ) AGE products become...Ch. 21 - Prob. 65PCh. 21 - Prob. 66PCh. 21 - Prob. 67PCh. 21 - Prob. 68PCh. 21 - Prob. 69PCh. 21 - 22-84 How many different dipeptides can be made...Ch. 21 - 22-85 Denaturation is usually associated with...Ch. 21 - Prob. 72PCh. 21 - Prob. 73PCh. 21 - Prob. 74PCh. 21 - 22-89 What kind of noncovalent interaction occurs...Ch. 21 - Prob. 76PCh. 21 - 22-91 Which amino acid does not rotate the plane...Ch. 21 - 22-92 Write the expected products of the acid...Ch. 21 - 22-93 What charges are on aspartic acid at pH 2.0?Ch. 21 - Prob. 80PCh. 21 - Prob. 81PCh. 21 - Prob. 82PCh. 21 - 22-97 Gelatin is derived from collagen by...Ch. 21 - Prob. 84PCh. 21 - Prob. 85PCh. 21 - Prob. 86PCh. 21 - 22-101 Using what you know about protein...Ch. 21 - 22-104 Why is collagen not a very good source of...Ch. 21 - Prob. 89P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 6) Select the major product of the following reaction. 1) Cl , AlCl3 ? ستى b) 2) H₂O or of o OH €arrow_forwardShow work. Don't give Ai generated solutionarrow_forwardσυ 3. Determine the rate law equation for a chemical re Mild The following is a chemical reaction: Fron law, 2A+2B C+D+E Run The reaction is found to be first order with respect to A and second order with respect to B. Write the rate law equation for the reaction. (include K, but you can't find the value). 1 How would doubling the concentration of reactant A affect the reaction rate? How would doubling the concentration of reactant B affect the reaction rate? 2 3 K Using yoarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

World of Chemistry
Chemistry
ISBN:9780618562763
Author:Steven S. Zumdahl
Publisher:Houghton Mifflin College Div

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY