
(a)
Draw the PV diagram of the closed cycle.
(a)
Answer to Problem 34P
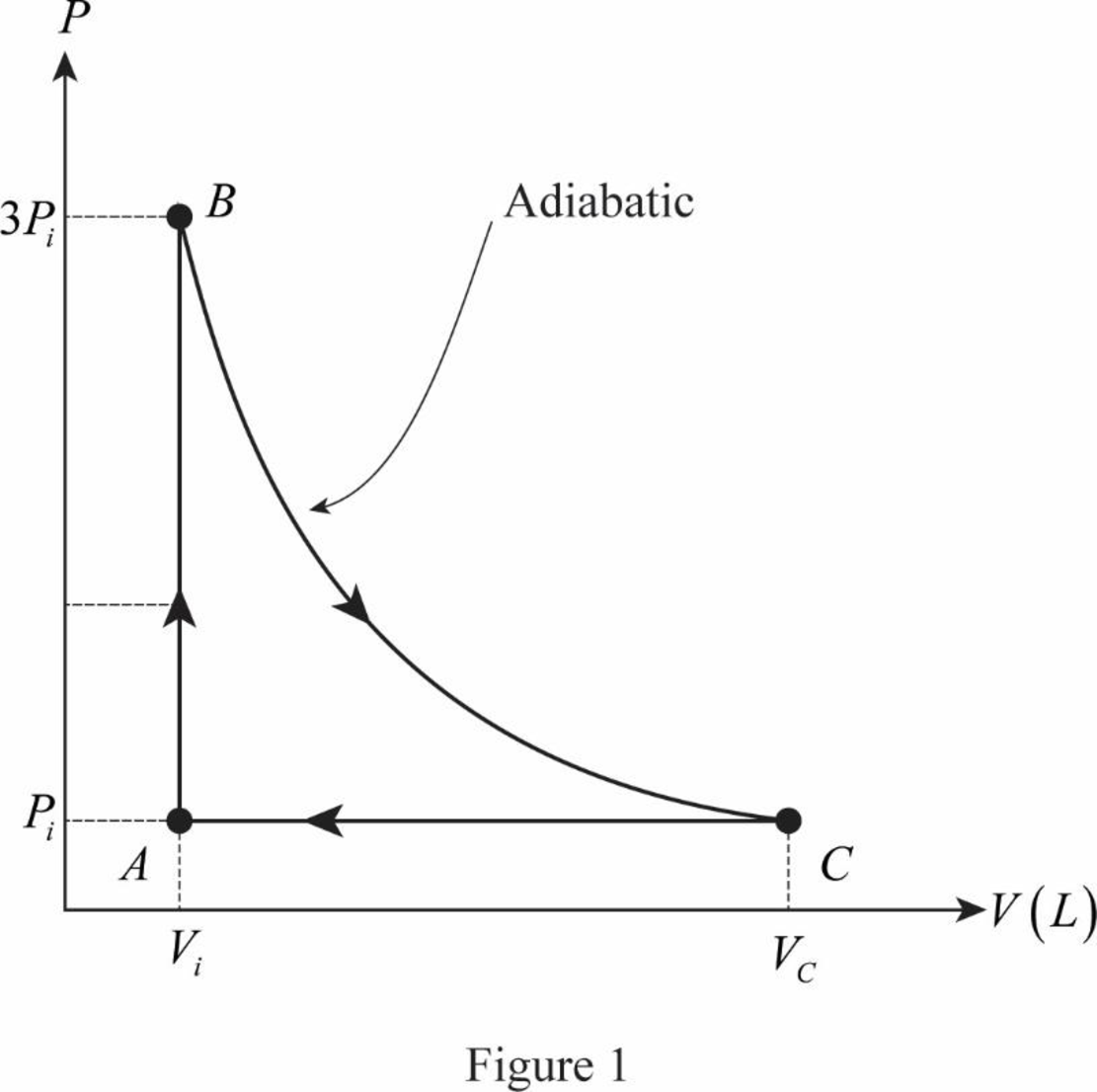
The PV diagram for the closed cycle is drawn.
Explanation of Solution
An adiabatic process occurs without transfer of heat or mass of substance between a
An Isobaric process is a thermodynamic process in which the pressure stays constant. The heat transferred to the system does work, but also changes the internal energy of the system.
Conclusion:
The figure 1 shows the PV diagram for the ideal gas expands adiabatically to its original pressure and compressed isobarically to its original volume.
(b)
The volume of the gas at the end of the adiabatic expansion.
(b)
Answer to Problem 34P
The volume of the gas at the end of the adiabatic expansion is
Explanation of Solution
Write the expression from PV diagram at the point B and C.
Here,
Conclusion:
Rewrite the above expression from the PV diagram to find volume.
Here,
Therefore, the volume of the gas at the end of the adiabatic expansion is
(c)
The temperature of the gas in an adiabatic expansion.
(c)
Answer to Problem 34P
The temperature of the gas in an adiabatic expansion is
Explanation of Solution
Write the expression from ideal
Here,
Write the expression from the PV diagram
Here,
Conclusion:
Compare equation (II) and (III) to find
Therefore, the temperature of the gas in an adiabatic expansion is
(d)
The temperature at the end of the cycle.
(d)
Answer to Problem 34P
The temperature at the end of the cycle is
Explanation of Solution
Write the expression for temperature for whole cycle.
Here,
Conclusion:
Therefore, the temperature at the end of the cycle is
(e)
The net work done on the gas.
(e)
Answer to Problem 34P
The net work done on the gas is
Explanation of Solution
Write the expression for energy transfer between the points AB.
Here,
Replace
Write the expression for energy transfer for adiabatic process between the points BC.
Write the expression from ideal gas law.
Here,
Replace
Write the expression for energy transfer between the points CA.
Here,
Replace
Conclusion:
Find the energy transfer for whole cycle.
Substitute the equation (V), (VI) and (VII) in above equation.
Rewrite the above equation.
Find the internal energy for whole cycle.
Here,
Substitute equation (VIII) in the above equation.
Therefore, the net work done on the gas is
Want to see more full solutions like this?
Chapter 21 Solutions
Physics for Scientists and Engineers With Modern Physics
- At point A in a Carnot cycle, 2.34 mol of a monatomic ideal gas has a pressure of 1 4000 kPa, a volume of 10.0 L, and a temperature of 720 K. The gas expands isothermally to point B and then expands adiabatically to point C, where its volume is 24.0 L. An isothermal compression brings it to point D, where its volume is 15.0 L. An adiabatic process returns the gas to point A. (a) Determine all the unknown pressures, volumes, and temperatures as you f ill in the following table: (b) Find the energy added by heat, the work done by the engine, and the change in internal energy for each of the steps A B, B C, C D, and D A (c) Calculate the efficiency Wnet/|Qk|. (d) Show that the efficiency is equal to 1 - TC/TA, the Carnot efficiency.arrow_forwardA copper rod of cross-sectional area 5.0 cm2 and length 5.0 m conducts heat from a heat reservoir at 373 K to one at 273 K. What is the time rate of change of the universe's entropy for this process?arrow_forwardA 1.00-mol sample of an ideal monatomic gas is taken through the cycle shown in Figure P21.37. The process A B is a reversible isothermal expansion. Calculate (a) the net work done by the gas, (b) the energy added to the gas by heat, (c) the energy exhausted from the gas by heat, and (d) the efficiency of the cycle. (e) Explain how the efficiency compares with that of a Carnot engine operating between the same temperature extremes. Figure P21.37arrow_forward
- Of the following, which is not a statement of the second law of thermodynamics? (a) No heat engine operating in a cycle can absorb energy from a reservoir and use it entirely to do work, (b) No real engine operating between two energy reservoirs can be more efficient than a Carnot engine operating between the same two reservoirs, (c) When a system undergoes a change in state, the change in the internal energy of the system is the sum of the energy transferred to the system by heat and the work done on the system, (d) The entropy of the Universe increases in all natural processes, (e) Energy will not spontaneously transfer by heat from a cold object to a hot object.arrow_forwardAs shown below, calculate the work done by the gas in the quasi-static processes represented by the paths (a) AB; (b) ADB; (c) ACB; and (d) ADCB. `arrow_forwardFind the work done in the quasi-static processes shown below. The states are given as (p, V) values for the points in the PV plane: 1 (3 atm, 4 L), 2 (3 atm, 6 L), 3 (5 atm, 4 L), 4 (2 atm, 6 L), 5 (4 atm, 2 L), 6 (5 atm, 5 L) and 7 (2 atm, 5 L).arrow_forward
- During the power stroke in a four-stroke automobile engine, the piston is forced down as the mixture of combustion products and air undergoes an adiabatic expansion. Assume (1) the engine is running at 2 500 cycles/min; (2) the gauge pressure immediately before the expansion is 20.0 atm; (3) the volumes of the mixture immediately before and after the expansion are 50.0 cm3 and 400 cm3, respectively (Fig. P21.31); (4) the time interval for the expansion is one-fourth that of the total cycle; and (5) the mixture behaves like an ideal gas with specific heat ratio 1.40. Find the average power generated during the power stroke.arrow_forwardA Carnot engine employs 1.5 mol of nitrogen gas as a working substance, which is considered as an ideal diatomic gas with =7.5 at the working temperatures of the engine. The Carnot cycle goes in the cycle ABCDA with AB being an isothermal expansion. The volume at points A and C of the cycle are 5.0103 m3 and 0.15 L, respectively. The engine operates between two thermal baths of temperature 500 K 300 K. (a) Find the values of volume at B and D. (b) How much heat is absorbed by the gas in the AB isothermal expansion? (c) How much work is done by the gas in the AB isothermal expansion? (d) How much heat is given up by the gas in the CD isothermal expansion? (e) How much work is done by the gas in the CD isothermal compression? (f) How much work is done by the gas in the BC adiabatic expansion? (g) How much work is done by the gas in the DA adiabatic compression? (h) Find the value of efficiency of the engine based on the net and heat input. Compare this value to the efficiency of a Carnot engine based on the temperatures of the baths.arrow_forward
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning




