
A sample of a monatomic ideal gas occupies 5.00 L at atmospheric pressure and 300 K (point A in Fig. P21.65). It is warmed at constant volume to 3.00 atm (point B). Then it is allowed to expand isothermally to 1.00 atm (point C) and at last compressed isobarically to its original state, (a) Find the number of moles in the sample.
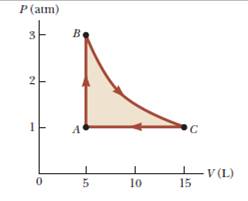
Find (b) the temperature at point B, (c) the temperature at point C, and (d) the volume at point C. (e) Now consider the processes A → B, B→ C, and C → A. Describe how to carry out each process experimentally, (f) Find Q, W, and ΔEint for each of the processes, (g) For the whole cycle A→ B→ C→ A, find Q, W, and ΔEint.
(a)
The number of moles in the sample.
Answer to Problem 21.65AP
The number of moles in the sample is
Explanation of Solution
Given info: The volume of the monatomic ideal gas is
The number of moles in the ideal gas equation is,
Here,
The value of the ideal gas constant is
Substitute
Conclusion:
Therefore, the number of moles in the sample is
(b)
The temperature at point
Answer to Problem 21.65AP
The temperature at point
Explanation of Solution
Given info: The volume of the monatomic ideal gas is
In the process from point
The expression for the process from point
Here,
Substitute
Conclusion:
Therefore, the temperature at point
(c)
The temperature at point
Answer to Problem 21.65AP
The temperature at point
Explanation of Solution
Given info: The volume of the monatomic ideal gas is
In the process from point
So, the temperature at point
Conclusion:
Therefore, the temperature at point
(d)
The volume at point
Answer to Problem 21.65AP
The volume at point
Explanation of Solution
Given info: The volume of the monatomic ideal gas is
In the process from point
The expression for the process from point
Here,
Substitute
Conclusion:
Therefore, the volume at point
(e)
The experimental methods to carry out the process
Answer to Problem 21.65AP
The experimental method to carry out the process
Explanation of Solution
Given info: The volume of the monatomic ideal gas is
In the process from point
The volume does not change. The temperature varies from
In the process from point
The temperature does not change. The pressure varies from
In the process from point
The pressure does not change. The temperature varies from
Conclusion:
Therefore, the experimental method to carry out the process
(f)
The heat
Answer to Problem 21.65AP
The heat
Explanation of Solution
Given info: The volume of the monatomic ideal gas is
For the process from point
The volume of gas does not change.
The work done is,
The change in internal energy is equal to the heat.
The expression for the change in internal energy is,
Here,
Substitute
Substitute
Thus, change in internal energy in process from point
For the process from point
The temperature does not change.
The change in internal energy is,
The expression of the work done is,
Substitute
Thus the change in internal energy in process from point
For the process from point
The formula of work done is,
Substitute
Thus, the work done for the point
The formula for the change in kinetic energy is,
Substitute
The heat obtain in this process is,
Conclusion:
Therefore, the heat
(g)
The heat
Answer to Problem 21.65AP
For the whole cycle
Explanation of Solution
Given info: The volume of the monatomic ideal gas is
The expression for the heat in complete cycle is,
Substitute
Thus, the heat in cycle is
The expression for the work done in complete cycle is,
Substitute
Thus, the total work done is
As the process is cyclic, the change in internal energy will be zero.
Conclusion:
Therefore, For the whole cycle
Want to see more full solutions like this?
Chapter 21 Solutions
Physics for Scientists and Engineers, Volume 1
- A sample of a monatomic ideal gas occupies 5.00 L at atmospheric pressure and 300 K (point A in Fig. P17.68). It is warmed at constant volume to 3.00 atm (point B). Then it is allowed to expand isothermally to 1.00 atm (point C) and at last compressed isobarically to its original state. (a) Find the number of moles in the sample. Find (b) the temperature at point B, (c) the temperature at point C, and (d) the volume at point C. (e) Now consider the processes A B, B C, and C A. Describe how to carry out each process experimentally. (f) Find Q, W, and Eint for each of the processes. (g) For the whole cycle A B C A, find Q, W, and Eint. Figure P17.68arrow_forwardOne process for decaffeinating coffee uses carbon dioxide ( M=44.0 g/mol) at a molar density of about 14,0 mol/m3 and a temperature of about 60 . (a) Is CO2 a solid, liquid, gas, or supercritical fluid under those conditions? (b) The van der Waals constants for carbon dioxide are a=0.3658 Pa m6/mol2 and b=4.286105 m3/mol. Using the van der Waals equation, estimate pressure of CO2 at that temperature and density. `arrow_forwardOn a hot summer day, the density of air at atmospheric pressure at 35.0C is 1.1455 kg/m3. a. What is the number of moles contained in 1.00 m3 of an ideal gas at this temperature and pressure? b. Avogadros number of air molecules has a mass of 2.85 102 kg. What is the mass of 1.00 m3 of air? c. Does the value calculated in part (b) agree with the stated density of air at this temperature?arrow_forward
- Cylinder A contains oxygen (O2) gas, and cylinder B contains nitrogen (N2) gas. If the molecules in the two cylinders have the same rms speeds, which of the following statements is false? (a) The two gases haw different temperatures. (b) The temperature of cylinder B is less than the temperature of cylinder A. (c) The temperature of cylinder B is greater than the temperature of cylinder A. (d) The average kinetic energy of the nitrogen molecules is less than the average kinetic energy of the oxygen molecules.arrow_forwardConsider a gas filling two connected chambers that are separated by a removable barrier (Fig. P20.68). The gas molecules on the left (red) are initially at a higher temperature than the ones on the right (blue). When the barrier between the two chambers is removed, the molecules begin to mix and move from one chamber to the other. a. Describe what happens to the temperature in the left chamber and in the right chamber as time goes on, once the barrier is open. Discuss in terms of the mixing of the molecules from each gas. b. Describe what happens to the most probable speed and average speed in the left chamber and in the right chamber as time goes on, once the barrier is open. Do they increase or decrease by the same factor? Explain. FIGURE P20.68 Problems 68 and 69.arrow_forwardHow many moles are there in (a) 0.0500 g of N2 gas (M = 28.0 g/mol)? (b) 10.0 g of CO2 gas (M = 44.0 g/mol)? (c) How many molecules are present in each case?arrow_forward
- An ideal gas initially at 300 K undergoes an isobaric expansion at 2.50 kPa. If the volume increases from 1.00 m3 to 3.00 m3 and 12.5 kJ is transferred to the gas by heat, what are (a) the change in its internal energy and (b) its final temperature?arrow_forwardA 2.00-mol sample of a diatomic ideal gas expands slowly and adiabatically from a pressure of 5.00 atm and a volume of 12.0 L to a final volume of 30.0 L. (a) What is the final pressure of the gas? (b) What are the initial and final temperatures? Find (c) Q, (d) Eint, and (e) W for the gas during this process.arrow_forwardA hollow container is filled with an ideal gas. The container is designed to maintain a constant pressure by allowing gas to enter or leave as needed. At all times the gas & container are in thermal equilibrium. Initially the temperature is 2350 K. Then the temperature decreases to 500 K and the volume decreases to 0.81 times the initial volume. Determine the coefficient of volume expansion for the container and the ratio of the final number of moles to the initial number of moles. Bcontainer nf ni = = K-1arrow_forward
- (a) A rigid tank contains 1.50 moles of carbon dioxide, which can be treated as an ideal gas, at a pressure of 22.7 atm. While the tank and gas maintain a constant volume and temperature, a number of moles are removed from the tank, reducing the pressure to 5.10 atm. How many moles are removed? mol (b) What If? In a separate experiment beginning from the same initial conditions, including a temperature T, of 25.0°C, half the number of moles found in part (a) are withdrawn while the temperature is allowed to vary and the pressure undergoes the same change from 22.7 atm to 5.10 atm. What is the final temperature (in °C) of the gas? °Carrow_forward1.50 moles of a monatomic ideal gas goes isothermally from state 1 to state 2. P1 = 3.5×105 Pa, V1 = 71 m3, and P2 = 5.5×105 Pa. What is the volume in state 2, in m3?arrow_forwardAn ideal gas has initial volume of 0.550 m³ and pressure of 9.65 x 104 Pa. (a) If the initial temperature is 281 K, find the number of moles of gas in the system. mol (b) If the gas is heated at constant volume to 388 K, what is the final pressure? Paarrow_forward
 Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning



