
Concept explainers
What Wittig reagent and carbonyl compound are needed to prepare each
a.
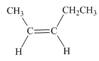 c.
c. ![]() d.
d. 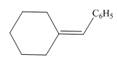
(a)
Interpretation: The Wittig reagent and carbonyl compound that are needed to prepare the given alkene are to be predicted, and the preferred route is to be identified, if any.
Concept introduction: One should follow the two steps retro synthesis to find out the starting materials that are needed to prepare the given alkene. The first step is cleaving of
Answer to Problem 21.57P
The Wittig reagent and carbonyl compound that are needed to prepare the given alkene are
Explanation of Solution
One should follow the two steps retrosynthesis to find out the Wittig reagent and carbonyl compound that are needed to prepare the given alkene. The first step is cleaving of
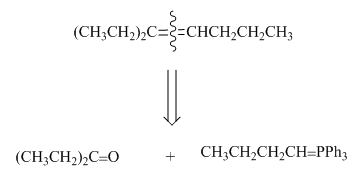
Figure 1
The second step is selection of preferred pathway. The Wittig reaction involves two pathways to synthesize unsymmetrical alkene and only one pathway to synthesize symmetrical alkene.
While selection one should consider that pathway in which Wittig reagent is derived from an unhindered alkyl halide, like
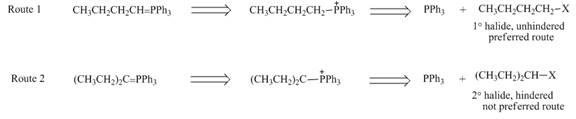
Figure 2
The first step for the preparation of Wittig reagent, formation of phosphonium salts, takes place through the
Thus, the Wittig reagent and carbonyl compound that are needed to prepare the given alkene are
The Wittig reagent and carbonyl compound that are needed to prepare the given alkene are
(b)
Interpretation: The Wittig reagent and carbonyl compound that are needed to prepare the given alkene are to be predicted, and the preferred route is to be identified, if any.
Concept introduction: One should follow the two steps retro synthesis to find out the starting materials that are needed to prepare the given alkene. The first step is cleaving of
Answer to Problem 21.57P
The Wittig reagent and carbonyl compound that are needed to prepare the given alkene are
Explanation of Solution
One should follow the two steps retrosynthesis to find out the Wittig reagent and carbonyl compound that are needed to prepare the given alkene. The first step is cleaving of
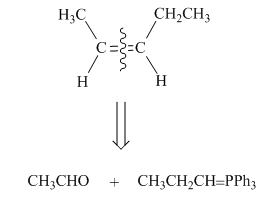
Figure 3
The second step is selection of preferred pathway. The Wittig reaction involves two pathways to synthesize unsymmetrical alkene and only one pathway to synthesize symmetrical alkene.
While selection one should consider that pathway in which Wittig reagent is derived from an unhindered alkyl halide, like
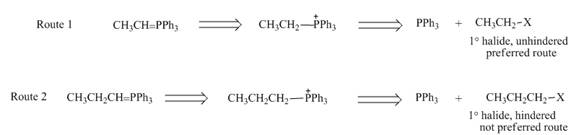
Figure 4
The first step for the preparation of Wittig reagent, formation of phosphonium salts, takes place through the
Thus, the Wittig reagent and carbonyl compound that are needed to prepare the given alkene are
The Wittig reagent and carbonyl compound that are needed to prepare the given alkene are
(c)
Interpretation: The Wittig reagent and carbonyl compound that are needed to prepare the given alkene are to be predicted, and the preferred route is to be identified, if any.
Concept introduction: One should follow the two steps retrosynthesis to find out the starting materials that are needed to prepare the given alkene. The first step is cleaving of
Answer to Problem 21.57P
The Wittig reagent and carbonyl compound that are needed to prepare the given alkene are shown in Figure 6. The preferred pathway is Route 1.
Explanation of Solution
One should follow the two steps retrosynthesis to find out the Wittig reagent and carbonyl compound that are needed to prepare the given alkene. The first step is cleaving of
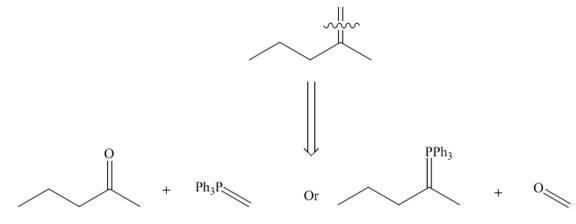 Figure 5
Figure 5
The second step is selection of preferred pathway. The Wittig reaction involves two pathways to synthesize unsymmetrical alkene and only one pathway to synthesize symmetrical alkene.
While selection one should consider that pathway in which Wittig reagent is derived from an unhindered alkyl halide, like
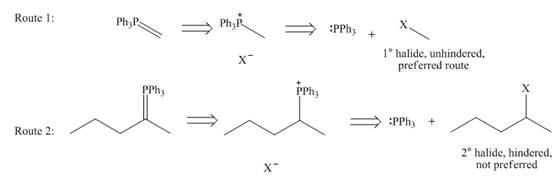
Figure 6
The first step for the preparation of Wittig reagent, formation of phosphonium salts, takes place through the
Thus, the Wittig reagent and carbonyl compound that are needed to prepare the given alkene are,
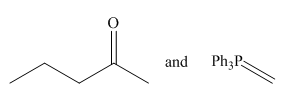
Figure 7
The Wittig reagent and carbonyl compound that are needed to prepare the given alkene are shown in Figure 6. The preferred pathway is Route 1.
(d)
Interpretation: The Wittig reagent and carbonyl compound that are needed to prepare the given alkene are to be predicted, and the preferred route is to be identified, if any.
Concept introduction: One should follow the two steps retrosynthesis to find out the starting materials that are needed to prepare the given alkene. The first step is cleaving of
Answer to Problem 21.57P
The Wittig reagent and carbonyl compound that are needed to prepare the given alkene are shown in Figure 7. The preferred pathway is Route 1.
Explanation of Solution
One should follow the two steps retrosynthesis to find out the Wittig reagent and carbonyl compound that are needed to prepare the given alkene. The first step is cleaving of
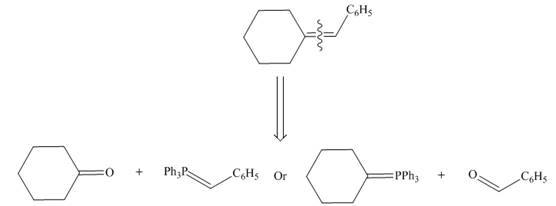 Figure 8
Figure 8
The second step is selection of preferred pathway. The Wittig reaction involves two pathways to synthesize unsymmetrical alkene and only one pathway to synthesize symmetrical alkene.
While selection one should consider that pathway in which Wittig reagent is derived from an unhindered alkyl halide, like
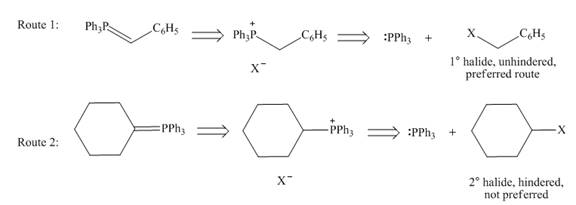
Figure 9
The first step for the preparation of Wittig reagent, formation of phosphonium salts, takes place through the
Thus, the Wittig reagent and carbonyl compound that are needed to prepare the given alkene are,
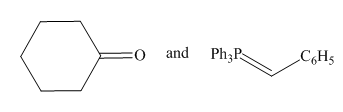
Figure 10
The Wittig reagent and carbonyl compound that are needed to prepare the given alkene are shown in Figure 10. The preferred pathway is Route 1.
Want to see more full solutions like this?
Chapter 21 Solutions
Organic Chemistry-Package(Custom)
- Draw the products formed when phenol (C6H5OH) is treated with each set of reagents. a. [1] HNO3, H2SO4; [2] Sn, HCl b. [1] (CH3CH2)2CHCOCl, AlCl3; [2] Zn(Hg), HCl c. [1] CH3CH2Cl, AlCl3; [2] Br2, hν d. [1] (CH3)2CHCl, AlCl3; [2] KMnO4arrow_forwardGive the IUPAC name for each compound. CH3 CH2CH3 Br a. PHCH(CH3)2 b. С. d.arrow_forwardDraw the organic product(s) formed when CH3CH₂CH₂OH is treated with each reagent. a. H₂SO4 d. HBr g. TsCl, pyridine b. NaH h. [1] NaH; [2] CH₂CH₂Br e. SOCI₂, pyridine f. PBr3 c. HCI + ZnCl₂ Hint: NaH deprotonates the alcohol forming an alkoxidearrow_forward
- Does the equilibrium favor the reactants or products in each substitution reaction? a. CH;CH2-NH2 Br CH;CH2-Br + "NH2 b. "CN CN + I-arrow_forwardClassify each alkyl halide as 1°, 2°, or 3°. CH3 c. CHg-C-CHCH3 ČH3 ČI CH;CH2CH,CH,CH2-Br b. d. a.arrow_forwardWhat is the major organic product obtained from the following reaction? A. CI B. H 1. (CH3CH₂)2CuLi 2. H3O+ C. D. •ay.a OHarrow_forward
- Draw the products of each reaction. CH3 a. CH3-C-CH,CH3 HCI HI OH HBr b. C. OHarrow_forwardcdn.fbsbx.com I. What products are formed when each alcohol is oxidized with K,Cr,0,? a. CH,CH,CH,CH,CH,OH OH OH H3C CH3 b. C. II, Give the structure corresponding to each IUPAC name:arrow_forwardDraw the product formed when each compound is treated with two equivalents of CH;CH,CH,CHgMgBr followed by H,0. OCH3 a. b. C. CH;CH2 OCH2CH3arrow_forward
- I. What products are formed when each alcohol is oxidized with K2Cr207? a. CH;CH,CH2CH;CH2OH ОН ОН H3C CH3 b. C.arrow_forwardRank the compounds in each group in order of increasing water solubility. a. (CH3),CH, CH3OCH2CH3, CH3CH;CH,CH3, CH;CH,CH2OH b. Brarrow_forwardDraw the products of combustion of each alkane. a. CH;CH,CH,CH2CH(CH3)2 b.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





