
Chemistry: Atoms First
3rd Edition
ISBN: 9781259638138
Author: Julia Burdge, Jason Overby Professor
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 20.2, Problem 20.2.3SR
What is the change in mass (in ka) for the following reaction? ΔH° for the reaction is −890.4 kJ/mol.
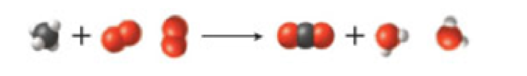
(a) 1.479 × 10−21 kg
(b) 1.479 × 10−24 kg
(c) 1.645 × 10−35kg
(d) 4.932 × 10−33 kg
(e) 9.907 × 10−15 kg
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which of the following reactions is most likely to be exothermic?(a) F2(g) → 2 F(g) (b) 2 Cl(g) → Cl2(g) (c) Cu(s) → Cu(l) (d) CBr4(g) → C(g) + 4 Br(g) (e) CO(g) → C(g) + O(g)
The lead-acid storage battery is the oldest rechargeable battery in existence. It was invented in 1859 by French physician
Gaston Plante and still retains application todaY, more than 150
years later.
There are two reactions that take place during discharge of the lead-acid storage battery. In one step, sulfuric acid
decomposes to form sulfur trioxide and water:
H, SO,() – SO;(9) + H,O()
AH-+113. kJ
In another step, lead, lead(IV) oxide, and sulfur trioxide react to form lead(II) sulfate:
Pb(s) – PbO,(s) - 2 S0, (g) → 2 PbSO,(s)
AH= -775. J
Calculate the net change in enthalpy for the formation of one mole of lead(II) sulfate from lead, lead(IV) oxide, and
sulfuric acid from these reactions.
Round your answer to the nearest kJ.
Consider the combustion of a single molecule of CH4(g),forming H2O(l) as a product. (a) How much energy, in J,is produced during this reaction? (b) A typical X-ray lightsource has an energy of 8 keV (see inside back cover forconversion between eV and J). Is the energy released by thecombustion of a CH4 molecule larger or smaller than the energyof an X-ray from this source?
Chapter 20 Solutions
Chemistry: Atoms First
Ch. 20.1 - Prob. 20.1WECh. 20.1 - Prob. 1PPACh. 20.1 - Prob. 1PPBCh. 20.1 - Prob. 20.1.1SRCh. 20.1 - Prob. 20.1.2SRCh. 20.2 - Prob. 20.2WECh. 20.2 - Prob. 2PPACh. 20.2 - Prob. 2PPBCh. 20.2 - Prob. 2PPCCh. 20.2 - Prob. 20.2.1SR
Ch. 20.2 - Prob. 20.2.2SRCh. 20.2 - What is the change in mass (in ka) for the...Ch. 20.3 - Prob. 20.3WECh. 20.3 - Prob. 3PPACh. 20.3 - Prob. 3PPBCh. 20.3 - Prob. 20.4WECh. 20.3 - Prob. 4PPACh. 20.3 - Prob. 20.3.1SRCh. 20.3 - Prob. 20.3.2SRCh. 20.4 - Prob. 20.5WECh. 20.4 - Prob. 5PPACh. 20.4 - Prob. 5PPBCh. 20.4 - Prob. 5PPCCh. 20.4 - Prob. 20.4.1SRCh. 20.4 - Prob. 20.4.2SRCh. 20 - Prob. 20.1QPCh. 20 - Prob. 20.2QPCh. 20 - Prob. 20.3QPCh. 20 - Prob. 20.4QPCh. 20 - Prob. 20.5QPCh. 20 - Prob. 20.6QPCh. 20 - Prob. 20.7QPCh. 20 - Prob. 20.8QPCh. 20 - Prob. 20.9QPCh. 20 - Prob. 20.10QPCh. 20 - Prob. 20.11QPCh. 20 - Prob. 20.12QPCh. 20 - Prob. 20.13QPCh. 20 - Prob. 20.14QPCh. 20 - Prob. 20.15QPCh. 20 - Prob. 20.16QPCh. 20 - Prob. 20.17QPCh. 20 - Prob. 20.18QPCh. 20 - Prob. 20.19QPCh. 20 - Prob. 20.20QPCh. 20 - Prob. 20.21QPCh. 20 - Prob. 20.22QPCh. 20 - Prob. 20.23QPCh. 20 - Prob. 20.24QPCh. 20 - Prob. 20.25QPCh. 20 - Prob. 20.26QPCh. 20 - Prob. 20.27QPCh. 20 - Prob. 20.28QPCh. 20 - Prob. 20.29QPCh. 20 - Prob. 20.30QPCh. 20 - Prob. 20.31QPCh. 20 - Prob. 20.32QPCh. 20 - Prob. 20.33QPCh. 20 - Prob. 20.34QPCh. 20 - Prob. 20.35QPCh. 20 - Prob. 20.36QPCh. 20 - Prob. 20.37QPCh. 20 - Prob. 20.38QPCh. 20 - Prob. 20.39QPCh. 20 - Prob. 20.1VCCh. 20 - Prob. 20.3VCCh. 20 - Prob. 20.4VCCh. 20 - Prob. 20.40QPCh. 20 - Prob. 20.41QPCh. 20 - Prob. 20.42QPCh. 20 - Prob. 20.43QPCh. 20 - Prob. 20.44QPCh. 20 - Prob. 20.45QPCh. 20 - Prob. 20.46QPCh. 20 - Prob. 20.47QPCh. 20 - Prob. 20.48QPCh. 20 - Prob. 20.49QPCh. 20 - Prob. 20.50QPCh. 20 - Prob. 20.51QPCh. 20 - Prob. 20.52QPCh. 20 - Prob. 20.53QPCh. 20 - Prob. 20.54QPCh. 20 - Prob. 20.55QPCh. 20 - Prob. 20.56QPCh. 20 - Prob. 20.57QPCh. 20 - Prob. 20.58QPCh. 20 - Prob. 20.59QPCh. 20 - Prob. 20.60QPCh. 20 - Prob. 20.61QPCh. 20 - Prob. 20.62QPCh. 20 - Prob. 20.63QPCh. 20 - Prob. 20.64QPCh. 20 - Prob. 20.65QPCh. 20 - Prob. 20.66QPCh. 20 - Prob. 20.67QPCh. 20 - Prob. 20.68QPCh. 20 - Prob. 20.69QPCh. 20 - Prob. 20.70QPCh. 20 - Prob. 20.71QPCh. 20 - Prob. 20.72QPCh. 20 - Prob. 20.73QPCh. 20 - Prob. 20.74QPCh. 20 - Prob. 20.75QPCh. 20 - Prob. 20.76QPCh. 20 - Prob. 20.77QPCh. 20 - Prob. 20.78QPCh. 20 - Prob. 20.79QPCh. 20 - Prob. 20.80QPCh. 20 - Prob. 20.81QPCh. 20 - Prob. 20.82QPCh. 20 - Prob. 20.83QPCh. 20 - Prob. 20.84QPCh. 20 - Prob. 20.85QPCh. 20 - Prob. 20.86QPCh. 20 - Prob. 20.87QPCh. 20 - Prob. 20.88QPCh. 20 - Prob. 20.89QPCh. 20 - Prob. 20.90QPCh. 20 - Prob. 20.91QPCh. 20 - Prob. 20.92QPCh. 20 - Prob. 20.93QPCh. 20 - Prob. 20.94QPCh. 20 - Prob. 20.95QPCh. 20 - Prob. 20.96QPCh. 20 - Prob. 20.97QPCh. 20 - Prob. 20.98QPCh. 20 - Prob. 20.99QPCh. 20 - Prob. 20.100QPCh. 20 - Prob. 20.101QP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Chlorine dioxide, ClO2, is a reddish yellow gas used in bleaching paper pulp. The average speed of a ClO2 molecule at 25C is 306 m/s. What is the kinetic energy (in joules) of a ClO2 molecule moving at this speed?arrow_forwardFormic acid, HCHO2, was first discovered in ants (formica is Latin for ant). In an experiment, 5.48 g of formic add was burned at constant pressure. 2HCHO2(l)+O2(g)2CO2(g)+2H2O(l) If 30.3 kJ of heat evolved, what is H per mole of formic acid?arrow_forwardWhat is the kinetic energy of a 0.146 kilogram baseball moving at a velocity of 44.8 meters per second? (KE=½ mv) Joulesarrow_forward
- 2.84 For each of the following reactions, decide if (i) tendency toward minimum energy favors reactants or products. ie odib (ii) tendency toward maximum randomness favors re- inst zidi an actants or products. (a) 2A12O3 (s) 2 4Al (s) + 302 (g) (b) CaO (s) + H2O (g) 2 Ca(OH)2 (s) (3) doso (c) 2C1 (g) C2 (g) (d) CO(NH2)2 (aq) + H20 (1) CO2 (g) + 2NH3 (g) AH = +28.4 kkal %3D (e) N204 (g) 2 2NO2 (g) AH = +14 kkål (f) 203 (g) 302 (g) AH= -68 kkal (g) 2NH3 (g) N, (g) + 3H2 (g) AH = +22 kkal %3D %3Darrow_forward(a) Dry red beans contain 62% carbohydrate, 22% protein,and 1.5% fat. Estimate the fuel value of these beans.(b) During a very light activity, such as reading or watchingtelevision, the average adult expends about 7 kJ>min. Howmany minutes of such activity can be sustained by the energyprovided by a serving of chicken noodle soup containing13 g protein, 15 g carbohydrate, and 5 g fat?arrow_forwardThe change in enthalpy for the reaction A → P is x kJ/mol. What does the enthalpy change for the reaction P →A ? (A) -x kJ/mol (B) +x kJ/mol (C) 0 kJ/mol (D) -2x kJ/molarrow_forward
- uestion 27 "A new element (Rr) has been synthesized and its properties analyzed. When 0.157g of RrO (s) is added to 125g of water at 22.2° C in a coffee cup calorimeter, the following reaction occurs. RrO(s) + H 20(1) – Rr(OH) 2 (aq) AH = -83.1k/mol Given the molar mass for RrO is 74g/mol calculate the following moles of Rro round answer to 4 decimal places omit trailing zeroes 9 rxn in joules round answer to the nearest whole number 9 water in joules round answer to the nearest whole number Final temperature round answer to 1 decimal place omit trailing zeroes " A Moving to another question will save this response.arrow_forwardThe photodissociation ozone in the stratosphere converts UV light energy into heat. Which of these reactions release heat? (A) O3 + hv --------> O2 + O (B) O + O2 ------> O3* (C) O3* + M -------> O3 + M* (D) O2 + hv -------> 2 O Group of answer choices only B only A A & D both release heat the combination of B & Carrow_forwardTarget Equation: (1) Mg(s) + 1/2 O2 (g) → MgO(s)Reactions: (2) MgO (s) + 2 HCl (aq) → MgCl2 (aq) + H2O (l) ΔH2 = ?(3) Mg (s) + 2 HCl (aq) → MgCl2 (aq) + H2 (g) ΔH3 = ?(4) H2 (g) + 1/2 O2 (g) → H2O (l) ΔH4 = -285.8 kJ Reaction Mass (grams) of MgO(s) Mass (grams) of Mg(s) Volume (mL) of HCl(aq) Concentration of HCl (mol/L) Initial Temp (°C) Final Temp (°C) 2 0.40 n/a 50.0 1.00 21.5 26.5 3 n/a 0.25 50.0 1.00 22.5 34.0 Using the data above, determine the enthalphy change (ΔH) for reactions (2) and (3). List any assumptions you make.Reaction (2): MgO (s) + 2 HCl (aq) → MgCl2 (aq) + H2O (l)Reaction (3): Mg (s) + 2 HCl (aq) → MgCl2 (aq) + H2 (g)arrow_forward
- Arrange these following four molecules in increasing order of their biological energy content (per mole):(a) Sucrose(b) Myristic acid, CH3(CH2)12COOH(c) Glucose(d) Capric acid, CH3(CH2)8COOHarrow_forward(a) What is the theoretical yield of ammonia (in grams) if 16.55 grams of nitrogen gas and 10.15 grams of hydrogen gas are allowed to react? (b)Based on your theoretical yield, what is the percent yield of ammonia if only 8.33 grams of ammonia are produced? (c)How much heat energy (in kJ) will be absorbed or released if 8.33 grams of ammonia are produced? State whether the energy will be absorbed or released.arrow_forwardSulfur → sulfur dioxide → sulfur trioxide → sulfuric acid (i) How is sulfur dioxide made from sulfur? [1] (c) The energy level diagram for the forward reaction in stage 2 is shown. 2SO,(g) + O2(g) energy 2S0,(g) progress of reaction Explain what the diagram shows about the energy changes in the forward reaction.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
Calorimetry Concept, Examples and Thermochemistry | How to Pass Chemistry; Author: Melissa Maribel;https://www.youtube.com/watch?v=nSh29lUGj00;License: Standard YouTube License, CC-BY