
Suppose 2.00 mol of a diatomic gas is taken reversibly around the cycle shown in the T-S diagram of Fig. 20-35. where S1 = 6.00 J/K and S2 = 8.00 J/K. The molecules do not rotate or oscillate. What is the energy transferred as heat Q for (a) path 1 → 2, (b) path 2 → 3, and (c) the full cycle? (d) What is the work W for the isothermal process? The volume V1 in state 1 is 0.200 m3. What is the volume in (e) state 2 and (f) state 3?
What is the change ΔEint for (g) path 1 → 2, (h) path 2 → 3, and (i) the full cycle? (Hint: (h) can be done with one or two lines of calculation using Module 19-7 or with a page of calculation using Module 19-9.) (j) What is the work W for the adiabatic process?
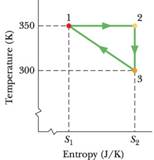
Figure 20-35 Problem 62.
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
WILEY PLUS 1 SEMESTER ACCESS CODE + LOOS
Additional Science Textbook Solutions
Glencoe Physics: Principles and Problems, Student Edition
Physics (5th Edition)
The Cosmic Perspective (8th Edition)
University Physics with Modern Physics (14th Edition)
Matter and Interactions
Glencoe Physical Science 2012 Student Edition (Glencoe Science) (McGraw-Hill Education)
- The insulated cylinder shown below is closed at both ends and contains an insulating piston that is flee to move on frictionless bearings. The piston divides the chamber into two compartments containing gases A and B. Originally, each compartment has a volume of 5.0102 m3 and contains a monatomic ideal gas at a temperature of and a pressure of 1.0 atm. (a) How many moles of gas are in each compartment? (b) Heat Q is slowly added to A so that it expands and B is compressed until the pressure of both gases is 3.0 atm. Use the fact that the compression of B is adiabatic to determine the final volume of both gases. (c) What are their final temperatures? (d) What is the value of Q?arrow_forwardFigure P21.45 shows a cyclic process ABCDA for 1.00 mol of an ideal gas. The gas is initially at Pi = 1.50 105 Pa, Vi = 1.00 103 m3 (point A in Fig. P21.45). a. What is the net work done on the gas during the cycle? b. What is the net amount of energy added by heat to this gas during the cycle? FIGURE P21.45arrow_forwardAn insulated vessel contains 1.5 moles of argon at 2 atm. The gas initially occupies a volume of 5 L. As a result of the adiabatic expansion the pressure of the gas is reduced to 1 atm (a) Find the volume and temperature of the final state. (b) Find the temperature of the gas in the initial state. (c) Find the work done by the gas in the process. (d) Find the change in the internal energy of the gas in the process.arrow_forward
- An ideal gas with specific heat ratio confined to a cylinder is put through a closed cycle. Initially, the gas is at Pi, Vi, and Ti. First, its pressure is tripled under constant volume. It then expands adiabatically to its original pressure and finally is compressed isobarically to its original volume. (a) Draw a PV diagram of this cycle. (b) Determine the volume at the end of the adiabatic expansion. Find (c) the temperature of the gas at the start of the adiabatic expansion and (d) the temperature at the end of the cycle. (e) What was the net work done on the gas for this cycle?arrow_forwardFor a temperature increase of 10 at constant volume, what is the heat absorbed by (a) 3.0 mol of a dilute monatomic gas; (b) 0.50 mol of a dilute diatomic gas; and (c) 15 mol of a dilute polyatomic gas?arrow_forwardA 2.00-mol sample of a diatomic ideal gas expands slowly and adiabatically from a pressure of 5.00 atm and a volume of 12.0 L to a final volume of 30.0 L. (a) What is the final pressure of the gas? (b) What are the initial and final temperatures? Find (c) Q, (d) Eint, and (e) W for the gas during this process.arrow_forward
- Compare the charge in internal energy of an ideal gas for a quasi-static adiabatic expansion with that for a quasi-static isothermal expansion. What happens to the temperature of an ideal gas in an adiabatic expansion?arrow_forwardA sample of a monatomic ideal gas occupies 5.00 L at atmospheric pressure and 300 K (point A in Fig. P21.65). It is warmed at constant volume to 3.00 atm (point B). Then it is allowed to expand isothermally to 1.00 atm (point C) and at last compressed isobarically to its original state, (a) Find the number of moles in the sample. Find (b) the temperature at point B, (c) the temperature at point C, and (d) the volume at point C. (e) Now consider the processes A B, B C, and C A. Describe how to carry out each process experimentally, (f) Find Q, W, and Eint for each of the processes, (g) For the whole cycle A B C A, find Q, W, and Eint.arrow_forwardA multicylinder gasoline engine in an airplane, operating at 2.50 103 rev/min, takes in energy 7.89 103 J and exhausts 4.58 103 J for each revolution of the crankshaft. (a) How many liters of fuel does it consume in 1.00 h of operation if the heat of combustion of the fuel is equal to 4.03 107 J/L? (b) What is the mechanical power output of the engine? Ignore friction and express the answer in horsepower. (c) What is the torque exerted by the crankshaft on the load? (d) What power must the exhaust and cooling system transfer out of the engine?arrow_forward
- During the power stroke in a four-stroke automobile engine, the piston is forced down as the mixture of combustion products and air undergoes an adiabatic expansion. Assume (1) the engine is running at 2 500 cycles/min; (2) the gauge pressure immediately before the expansion is 20.0 atm; (3) the volumes of the mixture immediately before and after the expansion are 50.0 cm3 and 400 cm3, respectively (Fig. P21.31); (4) the time interval for the expansion is one-fourth that of the total cycle; and (5) the mixture behaves like an ideal gas with specific heat ratio 1.40. Find the average power generated during the power stroke.arrow_forwardIf a gas is compressed isothermally, which of the following statements is true? (a) Energy is transferred into the gas by heat. (b) No work is done on the gas. (c) The temperature of the gas increases, (d) The internal energy of the gas remains constant, (e) None of those statements is true.arrow_forwardA sample of a monatomic ideal gas occupies 5.00 L at atmospheric pressure and 300 K (point A in Fig. P17.68). It is warmed at constant volume to 3.00 atm (point B). Then it is allowed to expand isothermally to 1.00 atm (point C) and at last compressed isobarically to its original state. (a) Find the number of moles in the sample. Find (b) the temperature at point B, (c) the temperature at point C, and (d) the volume at point C. (e) Now consider the processes A B, B C, and C A. Describe how to carry out each process experimentally. (f) Find Q, W, and Eint for each of the processes. (g) For the whole cycle A B C A, find Q, W, and Eint. Figure P17.68arrow_forward

 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning




