
Concept explainers
(a)
Interpretation: The products formed by the treatment of
Concept introduction: Organometallic reagents like
Answer to Problem 20.36P
The products formed by the treatment of
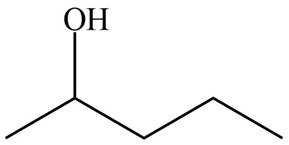
Figure 1
Compound B do not react with
Explanation of Solution
The ball and stick model of compound A and B shows that black balls represent carbon atom, white balls represent hydrogen atom and red balls represent oxygen atom. Therefore, the structure of A and B is,
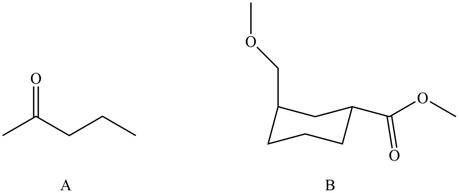
Figure 2
Ketones on reaction with
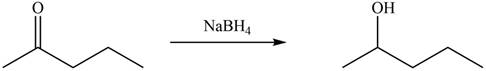
Figure 3
The products formed by the treatment of
(b)
Interpretation: The products formed by the treatment of
Concept introduction: Organometallic reagents like
Answer to Problem 20.36P
The products formed by the treatment of
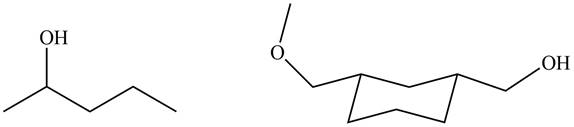
Figure 4
Explanation of Solution
The structure of A and B is shown in Figure 2.
Ketones on reaction with
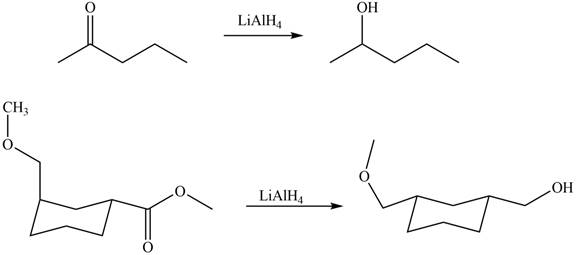
Figure 5
The products formed by the treatment of
(c)
Interpretation: The products formed by the treatment of
Concept introduction: Organometallic reagents like
Answer to Problem 20.36P
The products formed by the treatment of
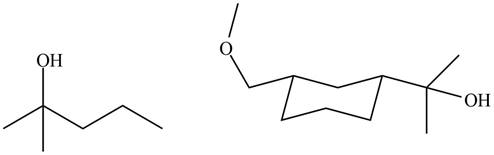
Figure 6
Explanation of Solution
The structure of A and B is shown in Figure 2.
Ketones and esters on reaction with Grignard reagent form tertiary alcohol. Therefore, the products formed by the treatment of
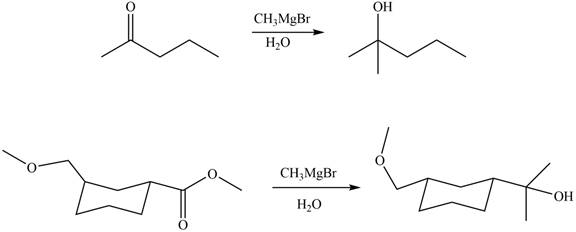
Figure 7
The products formed by the treatment of
(d)
Interpretation: The products formed by the treatment of
Concept introduction: Organometallic reagents like
Answer to Problem 20.36P
The products formed by the treatment of
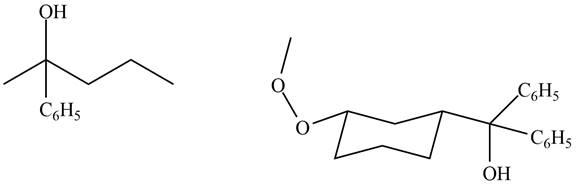
Figure 8
Explanation of Solution
The structure of A and B is shown in Figure 2.
Ketones and esters on reaction with organolithium reagent form tertiary alcohol. Therefore, the products formed by the treatment of
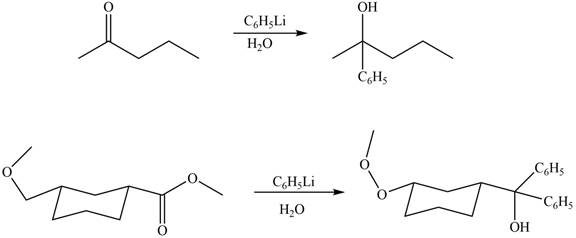
Figure 9
The products formed by the treatment of
(e)
Interpretation: The products formed by the treatment of
Concept introduction: Organometallic reagents like
Answer to Problem 20.36P
Compound A and B do not reaction with
Explanation of Solution
The structure of A and B is shown in Figure 2.
Ketones and esters do not undergo oxidation reaction with
Compound A and B do not reaction with
Want to see more full solutions like this?
Chapter 20 Solutions
ORGANIC CHEMISTRY
- Draw the product formed when (CH3)2CHOH is treated with each reagent. a. SOCl2, pyridine b. TsCl, pyridine c. H2SO4 d. HBr e. PBr3, then NaCN f. POCl3, pyridinearrow_forward3. What product is formed when HO(CH₂) CHO is treated with each reagent? c. Ag₂O, NH OH d. Na₂Cr₂O₂, H₂SO4, H₂O a. NaBH, CH₂OH b. PCC 400arrow_forwardDraw the organic product(s) formed when CH3CH₂CH₂OH is treated with each reagent. a. H₂SO4 d. HBr g. TsCl, pyridine b. NaH h. [1] NaH; [2] CH₂CH₂Br e. SOCI₂, pyridine f. PBr3 c. HCI + ZnCl₂ Hint: NaH deprotonates the alcohol forming an alkoxidearrow_forward
- Draw the product formed when phenylacetic acid (C6H5CH2COOH) is treated with each reagent. With some reagents, no reaction occurs. a. NaHCO3 b. NaOH c. SOCl2 d. NaCl e. NH3(1equiv) f. NH3, ∆ g. CH3OH, H2SO4 h. CH3OH, −OH i. [1] NaOH; [2] CH3COCl j. CH3NH2, DCC k. [1] SOCl2; [2] CH3CH2CH2NH2 (excess) l. [1] SOCl2; [2] (CH3)2; [2] (CHarrow_forward4. Draw the products formed when 3-hexyne is treated with each reagent a. HBr(2 equivalents) b. Bra (2 equivalents) c. H₂ (2 equivalents) d. [1] 2Na [2]NH,(1)arrow_forward1. Draw the products in each reaction. a. H b. CH3-C-CH₂CH3 CI -OH K+ -OC(CH3)3 CH₂CH3 a Xa C. d. CH₂CH₂OH CH3CH₂O™arrow_forward
- Draw the products of each reaction. NANH2 NaOH KNH2 b. CH,0- -CI a. NH3 H20 NH3 Aarrow_forwardDraw the products of each reaction. CH3 a. CH3-C-CH,CH3 HCI HI OH HBr b. C. OHarrow_forwardDraw the products formed when each compound is treated with CH;CH,COCI, AICI3. CH(CH3)2 N(CH)2 Br CH3 CH(CH)2 a. b. C. d. е.arrow_forward
- Does the equilibrium favor the reactants or products in each substitution reaction? a. CH;CH2-NH2 Br CH;CH2-Br + "NH2 b. "CN CN + I-arrow_forwardWhat is the major organic product obtained from the following reaction? A. CI B. H 1. (CH3CH₂)2CuLi 2. H3O+ C. D. •ay.a OHarrow_forwardDraw the product formed when pentanal (CH3 CH₂ CH₂ CH₂ CHO) is treated with each reagent. With some reagents, no reaction occurs. a. NaBH4, CH3OH b. [1] LiAiH4: [2] H₂O c. H₂, Pd-C d. PCC e. Na₂Cr₂O7, H₂SO4, H₂O f. Ag₂O, NH4OH g. [1] CH3 MgBr; [2] H₂O h. [1] C6H5 Li: [2] H₂O i. [1] (CH3)2 CuLi; [2] H₂O J. [1] HC=CNa; [2] H₂O k. [1] CH 3 C=CLI; [2] H₂O 1. The product in (a), then TBDMS-CI, imidazolearrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





