
Interpretation:
The mechanism for the reaction of tert-butyl alcohol and 1-butanol under the reaction conditions in the experiment needs to be explained.
Concept Introduction:
Generally, alcohols do not react in simple nucleophilic displacement reactions. If alcohols are attracted directly by nucleophile, hydroxide ion which is a strong base can be displaced easily from the alcohol. But the reaction is not energetically favorable and cannot occur to an extent.
To avoid this, the nucleophilic substitution reaction of alcohol takes place in acidic medium. Initially, protonation of alcohol takes place followed by the removal of water molecule. The reaction is energetically stable.
This is represented as follows:
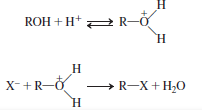
Explanation of Solution
This reaction examines the capability of two halides to replace an alcohol bynucleophilic substitution.
Initially, suppose the reaction of bromide and chloride ions with tert-butyl alcohol. Below possible reaction will take place:
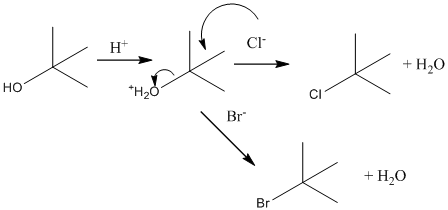
Now suppose, reaction of bromide and chloride ions with 1-butanol. Below possible reaction will take place:
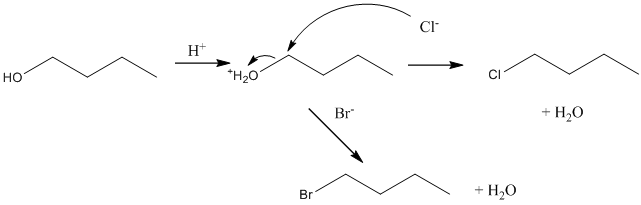
Want to see more full solutions like this?
Chapter 20 Solutions
EBK A SMALL SCALE APPROACH TO ORGANIC L
- Based on the theoretical result in the table, what is the order of reactivity of primary, secondary, and tertiary alkyl halides with sodium iodide in acetone?arrow_forwardcomplete overall reaction when taking KAuCl4 in methanol mixed with 2-phenyl-1,10-phenanthroline in dichloromethanearrow_forwardThis experiment is about synthesis of 1,3-diphenyl-2-propenone.Before the enone formation, how is the product called? Write the mechanism of its base cata-lyzed enal formation.arrow_forward
- Which qualitative analysis test from this module can help distinguish between different kinds of ketones? Can this same test give a positive result for 4-methylacetophenone? Why?arrow_forwardWhy is it difficult to stop the reaction at the mono-brominated stage?arrow_forwardShow the sequence of Heck reactions by which the following conversion takes place. Note from the molecular formula given under each structural formula that this conversion corresponds to a loss of H and I from the starting material.arrow_forward
- Write the complete mechanism (including intermediates) for the reaction of trans-stilbene with bromine in dichloromethane.arrow_forwardWrite the complete mechanism for the hydroxide ion catalyze reaction of cyclohexanone and 1-methylcyclohexanecarbaldehydearrow_forwardExplain in detail the mechanism of how benzoic acid is converted to m-Nitrobenzyl alcohol. Use the picture as a reference.arrow_forward
- What would be the product ratio in the chlorination of propane if all the hydrogens were abstracted at equal rates?arrow_forwardWrite the mechanism for the nitration of nitrobenzene. Use electron arrow pushing to show the formation of the electrophile (NO2+), the intermediate arenium ion and the final product of the rxnarrow_forwardFriedel-Crafts Alkylation, based on substituted benzene reaction, including reactivity based on electron donating and withdrawing group and also discuss the possible positions (Ortho, Meta and Para) for the new group. Explain if there is polysubstitution occurs. Keeping the reaction mixture in the ice bath and adding the reagent slowly by continuously stirring is the key point of success.arrow_forward
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

