
Give the complete IUPAC name for each of the following compounds:
(a) CH3CH2CBr2CH3
(b) (CH3)3CCl
(c)
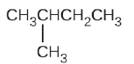
(d)
(e)
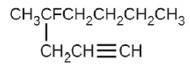
(f)
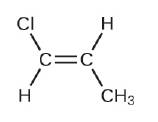
(g)
Trending nowThis is a popular solution!

Chapter 20 Solutions
Chemistry by OpenStax (2015-05-04)
Additional Science Textbook Solutions
College Physics
Chemistry: The Central Science (14th Edition)
General Chemistry: Principles and Modern Applications (11th Edition)
Introductory Chemistry (6th Edition)
Introductory Chemistry (5th Edition) (Standalone Book)
Chemistry: Structure and Properties
- Name or write the condensed structural formula for the fol- lowing compounds: (a) trans-2-pentene (b) 2,5-dimethyl-4-octene (c) CH3 CH;CHCH,CH3 C=C `H. CH;CH2, H СООН H CH3 CH CH3 CH3 `CH2 (d)arrow_forward1. (a) Draw the structures of the eight isomeric pentyl alcohols, C3H11OH. (b) Name each by the IUPAC system and by the carbinol system. (c) Label each as primary, secondary, or tertiary, (d) Which one is isopentyl alcohol? tert-Pentyl alcohol? (e) Give the structure of a primary, a secondary, and a tertiary alcohol of the formula C,H13OH.arrow_forwardPlease name (a) CH3-CH2-CH2-CH-CH3 (b) CH3arrow_forward
- Which of the following alkenes can exist as cis-trans isomers? Write their structures. (a) CH2=CHCH2CH3 (c) CH2=C(CH3)2 (b) CH3CH=CHCH3 (d) CH3CH2CH=CHCIarrow_forward(a) Write the IUPAC names of the following compounds :(i) CH3CO(CH2)4CH3 (ii) Ph — CH = CH — CHO(b) Describe the following conversions in not more than two steps :(i) Ethanol to 3-Hydroxybutanal (ii) Benzoic acid to m-Nitrobenzyl alcohol(iii) Propanone to Propenearrow_forwardWrite the bond line formula of the following compounds: (a) 4-methyl-2-hexene, two geometrical (stereoisomers) isomers (b) 3-fluoro-2-methylheptanol (3-fluoro-2-methylheptan-1-ol) (c) 4-methyl-hex-1-yn-3-olarrow_forward
- Provide IUPAC names for the following compounds. (a) (CH3)2СHCH,СH; (b) CНз — С (CHз)2 — СH3 CH, CH,CH, CH,CH, (c) CH,CH,CH,CH–CH(CH,), (d) CH,—СH—CH,—CH—СH, -arrow_forwardGive the name or condensed structural formula, as appropriate: (a) CH;CHCH3 СHCH-CH-CHCНЗ ČH3 (b) 2,2-dimethylpentane (c) 4-ethyl-1,1-dimethylcyclohexane (d) (CH3),CHCH,CH,C(CH3)3 (e) CH;CH,CH(C,H3)CH,CH,CH,CH3arrow_forwardWrite Lewis structures and describe the molecular geometry at each carbon atom in the following compounds:(a) cis-3-hexene(b) cis-1-chloro-2-bromoethene(c) 2-pentyne(d) trans-6-ethyl-7-methyl-2-octenearrow_forward
- A certain hydrocarbon has a molecular formula of C5H8. Which of the following is not a structural possibility for this hydrocarbon: (d) It contains an alkyne O It contains one ring and one double bond (c) It contains two double bonds and no rings O (b) It contains one ring and no double bondsarrow_forward(ii) Name the following using IUPAC system of nomenclature: (a) CH2 (Cl) CH (NH2) CHO (b) CH3 C (Br) CH2 CH (F) COOH Brarrow_forwardTRUE OR FALSE (a) Both ethylene and acetylene are planar molecules. (b) An alkene in which each carbon of the double bond has two different groups bonded to it will show cis-trans isomerism. (c) Cis-trans isomers have the same molecular formula but a different connectivity of their atoms. (d) Cis-2-butene and trans -2-butene can be interconverted by rotation about the carbon–carbon double bond. (e) Cis-trans isomerism is possible only among appropriately substituted alkenes. (f) Both 2-hexene and 3-hexene can exist as pairs of cis-trans isomers. (g) Cyclohexene can exist as a pair of cis-trans isomers. (h) 1-Chloropropene can exist as a pair of cis-trans isomers.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





