
Basic Chemistry
6th Edition
ISBN: 9780134878119
Author: Timberlake, Karen C. , William
Publisher: Pearson,
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 88UTC
The chapter sections to review are shown in parentheses at the end of each problem
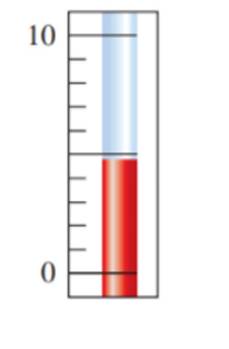
State the temperature on the Celsius thermometer to the correct number ofsignificant figures: (2.3)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Perform the calculation and record the answer with the correct number of significant figures.
(6.5-6.09)
2.15
=
X10
TOOLS
A sample is put into 400-mL of water. It releases 58.829 kJ of heat energy when it is
combusted. What was the temperature change of the water during this combustion?
(1:3)
(1.6) Which of the following is an example of intensive properties?
mass
melting point
O None of these
volume
Chapter 2 Solutions
Basic Chemistry
Ch. 2.1 - Write the abbreviation for each of the following:...Ch. 2.1 - Write the abbreviation for each of the following:...Ch. 2.1 - State the type of measurement in each of the...Ch. 2.1 - State the type of measurement in each of the...Ch. 2.1 - State the name of the unit and the type of...Ch. 2.1 - State the name of the unit and the type of...Ch. 2.1 - On a typical day, medical personnel may encounter...Ch. 2.1 - On a typical day, medical personnel may encounter...Ch. 2.2 - Use the metric ruler to measure the length in each...Ch. 2.2 - Determine the volume, in milliliters, of each...
Ch. 2.2 - How many significant figures are in each of the...Ch. 2.2 - How many significant figures are in each of the...Ch. 2.2 - In which of the following pairs do both numbers...Ch. 2.2 - In which of the following pairs do both numbers...Ch. 2.2 - Prob. 15PPCh. 2.2 - Prob. 16PPCh. 2.2 - Write each of the following in scientific notation...Ch. 2.2 - Write each of the following in scientific notation...Ch. 2.2 - Identify the numbers in each of the following...Ch. 2.2 - Prob. 20PPCh. 2.2 - Prob. 21PPCh. 2.2 - Identify the exact number(s), if any, in each of...Ch. 2.2 - Prob. 23PPCh. 2.2 - Identify each of the following as measured or...Ch. 2.3 - Round off each of the following calculator answers...Ch. 2.3 - Round off each of the calculator answers in...Ch. 2.3 - Round off or add zeros to each of the following to...Ch. 2.3 - Round off or add zeros to each of the following to...Ch. 2.3 - Perform each of the following operations, and give...Ch. 2.3 - Perform each of the following operations, and give...Ch. 2.3 - Prob. 31PPCh. 2.3 - Perform each of the following operations, and give...Ch. 2.4 - Write the abbreviation for each of the following...Ch. 2.4 - Write the abbreviation for each of the following...Ch. 2.4 - Write the complete name for each of the following...Ch. 2.4 - Prob. 36PPCh. 2.4 - Prob. 37PPCh. 2.4 - Prob. 38PPCh. 2.4 - Prob. 39PPCh. 2.4 - Prob. 40PPCh. 2.4 - Complete each of the following metric...Ch. 2.4 - Prob. 42PPCh. 2.4 - For each of the following pairs, which is the...Ch. 2.4 - For each of the following pairs, which is the...Ch. 2.5 - Why can two conversion factors be written for an...Ch. 2.5 - How can you check that you have written the...Ch. 2.5 - Write the equality and two conversion factors for...Ch. 2.5 - Write the equality and two conversion factors for...Ch. 2.5 - Prob. 49PPCh. 2.5 - Write the equality and two conversion factors, and...Ch. 2.5 - Write the equality and two conversion factors, and...Ch. 2.5 - Write the equality and two conversion factors, and...Ch. 2.5 - Write the equality and two conversion factors, and...Ch. 2.5 - Prob. 54PPCh. 2.5 - Prob. 55PPCh. 2.5 - Write an equality and two conversion factors for...Ch. 2.6 - Perform each of the following conversions using...Ch. 2.6 - Perform each of the following conversions using...Ch. 2.6 - Perform each of the following conversions using...Ch. 2.6 - Prob. 60PPCh. 2.6 - Use metric conversion factors to solve each of the...Ch. 2.6 - Use metric conversion factors to solve each of the...Ch. 2.6 - Solve each of the following problems using one or...Ch. 2.6 - Solve each of the following problems using one or...Ch. 2.6 - Using conversion factors, solve each of the...Ch. 2.6 - Using conversion factors, solve each of the...Ch. 2.6 - Using conversion factors, solve each of the...Ch. 2.6 - Using conversion factors, solve each of the...Ch. 2.7 - Determine the density (g/mL) for each of the...Ch. 2.7 - Determine the density (g/mL) for each of the...Ch. 2.7 - Prob. 71PPCh. 2.7 - What is the density (g/mL) of each of the...Ch. 2.7 - Use the density values in Table 2.10 to solve each...Ch. 2.7 - Use the density values in Table 2.10 to solve each...Ch. 2.7 - Use the density values in Table 2.10 to solve each...Ch. 2.7 - Use the density values in Table 2.10 to solve each...Ch. 2.7 - In an old trunk, you find a piece of metal that...Ch. 2.7 - Suppose you have two 100-mL graduated cylinders....Ch. 2.7 - Solve each of the following problems: a. A urine...Ch. 2.7 - Prob. 80PPCh. 2.7 - Prob. 81PPCh. 2.7 - a. Write an equality and two conversion factors...Ch. 2 - The chapter sections to review are shown in...Ch. 2 - The chapter sections to review are shown in...Ch. 2 - The chapter sections to review are shown in...Ch. 2 - Prob. 86UTCCh. 2 - Prob. 87UTCCh. 2 - The chapter sections to review are shown in...Ch. 2 - Prob. 89UTCCh. 2 - Prob. 90UTCCh. 2 - Prob. 91UTCCh. 2 - Prob. 92UTCCh. 2 - Prob. 93UTCCh. 2 - Prob. 94UTCCh. 2 - Prob. 95UTCCh. 2 - Prob. 96UTCCh. 2 - Prob. 97APPCh. 2 - The chapter sections to review are shown in...Ch. 2 - The chapter sections to review are shown in...Ch. 2 - The chapter sections to review are shown in...Ch. 2 - Prob. 101APPCh. 2 - Prob. 102APPCh. 2 - Prob. 103APPCh. 2 - Prob. 104APPCh. 2 - Prob. 105APPCh. 2 - Prob. 106APPCh. 2 - Prob. 107APPCh. 2 - Prob. 108APPCh. 2 - Prob. 109APPCh. 2 - Prob. 110APPCh. 2 - Prob. 111APPCh. 2 - Prob. 112APPCh. 2 - Prob. 113APPCh. 2 - Prob. 114APPCh. 2 - Prob. 115APPCh. 2 - Prob. 116APPCh. 2 - The water level in a graduated cylinder initially...Ch. 2 - Prob. 118APPCh. 2 - Prob. 119APPCh. 2 - Prob. 120APPCh. 2 - Prob. 121APPCh. 2 - Prob. 122APPCh. 2 - Prob. 123APPCh. 2 - Prob. 124APPCh. 2 - Prob. 125APPCh. 2 - Prob. 126APPCh. 2 - The following problems at related io the topics in...Ch. 2 - The following problems at related io the topics in...Ch. 2 - The following problems at related io the topics in...Ch. 2 - The following problems at related io the topics in...Ch. 2 - Prob. 131CPCh. 2 - The following problems at related io the topics in...Ch. 2 - Prob. 133CPCh. 2 - The following problems at related io the topics in...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Perform the calculation and record the answer with the correct number of significant figures. (6.5 – 6.35) 4.32 x10 TOOLSarrow_forward(1.3)Which of the following techniques can separate the homogeneous mixture of two liquids with different boiling points? O All of these O decanting O filtration O distillationarrow_forward1.48 Write each of the following in scientific notation: (1.5) a. 0.0042 b. 310 c. 890 000 000 d. 0.000 000 056 Iarrow_forward
- (1.4)Which of the following is a chemical change? O cutting papers O rusting iron O breaking glasses dissolving sugar in waterarrow_forward(4.3)(0.267) (7.40)(30.0) Above is written as a fraction Express your answer using the correct number of significant figures.arrow_forwardA heated lead ball is added to 158 grams of water in a coffee cup calorimeter. If the ball loses 47.35 °C of heat and the water gains 1.3 °C of heat, what is the mass of the lead ball? (3 sf)arrow_forward
- Complete the following operations and write your answer with the correct number of significant figures. Use scientific notation when appropriate. (40.25)(23.4)(1.0001) =arrow_forward2.92 A graduated cylinder contains three liquids A, B, and C, which have different densities and do not mix: mercury (D = 13.6 g/mL), vegetable oil (D 0.92 g/mL), and water (D == 1.00 g/mL). Identify the liquids A, B, and C in the cylinder. (2.7) 50 40 30 -B 20 10 2.93 The gray cube has a density of 4.5 g/em2. Is the density of the green cube the same, lower than, or higher than that of the grayarrow_forwardlead ball is added to 158 grams of water in a coffee cup calorimeter. If the ball loses 47.35 °C of heat and the water gains 1.3 °C of heat, what would the mass of the lead ball be? (3 sf)arrow_forward
- (1.8)The area of a paper is 758 cm². What is the area of the paper in m²? O 7.58 x 104 m² O 7.58 x 102 m² O 0.0379 m² O 7.58 m²arrow_forward0 The average adult female should consume 2000 Cal per day. If you've consumed 35% of your daily Calories with breakfast, how many Calories did you eat at breakfast? 78°F Rain ? (8) 2 W # 3 E f4 IOI 4 R 15 do % 5 T f6 10 6 17 + & Y 7 f8 8arrow_forwardEvaluate the following and express your answer in standard scientific notation. (3.5-10 2) (7.1210 4) (9.6z10) O 5.1x10-2 O 5.1x10-3 O 4.71x10-4 O 4.7x100arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Types of Matter: Elements, Compounds and Mixtures; Author: Professor Dave Explains;https://www.youtube.com/watch?v=dggHWvFJ8Xs;License: Standard YouTube License, CC-BY