
Concept explainers
(a)
Interpretation:
The stronger acid has to be determined.
Concept Introduction:
Effect of hybridization on Acidity: The electronegativity of an atom depends on its hybridization.
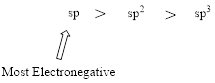
The stronger acid will have its proton attached to the more electronegative atom.
(b)
Interpretation:
The stronger acid has to be determined.
Concept Introduction:
Effect of hybridization on Acidity: The electronegativity of an atom depends on its hybridization.
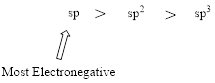
The stronger acid will have its proton attached to the more electronegative atom.
(c)
Interpretation:
The stronger acid has to be determined.
Concept Introduction:
Effect of hybridization on Acidity: The electronegativity of an atom depends on its hybridization.
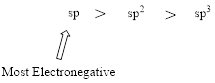
The stronger acid will have its proton attached to the more electronegative atom.
Want to see the full answer?
Check out a sample textbook solution
Chapter 2 Solutions
EBK ESSENTIAL ORGANIC CHEMISTRY
- Which is a stronger base?a. HS− or HO− b. CH3O− or CH3N-H c. CH3OH or CH3O− d. Cl− or Br−e. CH3COO− or CF3COO− f. CH3CHClCOO− or CH3CHBrCOO−arrow_forwardWhich is a stronger acid?arrow_forwardWhich is a stronger acid? a. CH2-CHCOOH or CH3CH2COOH b. H-N-H or H-NO-H c. CH2-CHCOOH or HC CCOOH d. H-N-H or N-Harrow_forward
- Which acid is the strongest? A. H2SO3 B. H3AsO3 C. H2SeO3arrow_forwardRank the compounds in each group in order of increasing acidity. ÇI Br .COOH „COOH COOH а. COOH COOH COOH CH CF3 LOH HO HO HO HO HO b. d. CH O,N Br O,N O,N `NO2 Rank the compounds in each group in order of increasing basicity. NH2 NH2 NH2 a. NH3 -NH2 -NH2 c. O,N CH3 b. d. CgHgNH2 (C,H5)2NH -NH2 N. H H C.arrow_forwardWhich is a stronger base?arrow_forward
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
