
Concept explainers
Where on the following outline of a periodic table do the indicated elements or groups of elements appear?
(a) Alkali metals
(b) Halogens
(c) Alkaline earth metals
(d)
(e) Hydrogen
(f) Helium
(g) Metalloids
Interpretation:
The given elements or group of elements occurs where in the periodic table has to be given.
Explanation of Solution
The elements and groups are indicated below,
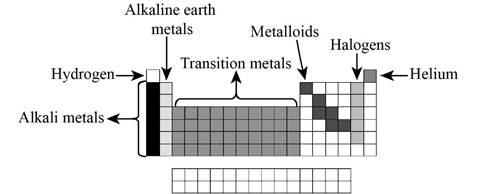
Alkali metals are 1A group elements (other than hydrogen), Halogens are 7A group elements, Alkaline earth metals are 2A group elements, Transition metals are 1B to 8B group elements, Hydrogen is the first element in the periodic table, helium is the second element in the periodic table located in 8A group. Metalloids are main group elements (which is located in 3A to 8A).
Want to see more full solutions like this?
Chapter 2 Solutions
EBK FUNDAMENTALS OF GENERAL, ORGANIC, A
- A principal shell with a value of n=3 would contain s,p,d, and f orbitals true or falsearrow_forward(a) A homogeneous mixture which contains water as a solvent is called (b) Ni(CIO4)2-6H2O is hydrated whereas Ni(CIO.)e is (c) NaCl contains an bond whereas O2(g) contains a bond (d) A homogeneous mixture has a and composition (e) Temperature is an because it does not depend on the amount of substance (f) The maximum number of electrons that an orbital can have is (9) The energy of the lowest level in the H atom is (h) Arrange the following subshells in the H atom in order of increasing energy: 3s 4d 2р 4f 3d 2s 3p () Wavelength and frequency of radiation have an relationshiparrow_forwardWhich of the following elements would you expect to form (i) diatomic molecules, (ii) mainly covalent bonds, (iii) mainly ionic bonds, and (iv) both covalent and ionic bonds? (More than one answer may apply; rememberthat some nonmetals can form ionic bonds with metals.) Explain your answers.arrow_forward
- Look up antimony in the periodic table 1Z = 512. How many covalent bonds would you expect it to form? Based on this information, which of the following antimony compounds is covalent and which is ionic: SbCl3 or SbCl5?arrow_forwardOxygen has 8 protons, 8 neutrons, and 8 electrons. What is its atomic mass?(a) 8 (b) 16 (c) 24 (d) 32.arrow_forwardThe atomic mass number of an atom is determined by the sum of the number of in the atom. (a) protons plus electrons plus neutrons (b) protons plus electrons (c) neutrons plus electrons (d) neutrons plus protonsarrow_forward
- Another major use of bismuth has been as an ingredient in low-melting metal alloys, such as those used in firesprinkler systems and in typesetting. The element itself is a brittle white crystalline solid. How do these characteristicsfit with the fact that bismuth is in the same periodic group with such nonmetallic elements as nitrogenand phosphorus?arrow_forwardArrange the elements Na, Si, and S in the order of(a) decreasing atomic radius.(h)increasing first ionization energy.arrow_forwardUse your copy of the periodic table to answer this question: If you add an electron to fluorine, what will result? A) a negatively charged anion B) a different atomic number C) a different isotope D) a different element E) a positively charged cationarrow_forward
- What two elements besides H and N make up the bulk of living matter?arrow_forward1.1. An atom of element X has an atomic number of 14 and a mass number of 30. Which of the following statements is correct? (A) An atom of sodium has 14 protons, 14 electrons, and 14 neutrons.(B) An atom of sodium has 14 protons, 16 electrons, and 14 neutrons(C) An atom of sodium has 14 protons, 14 electrons, and 16 neutrons.(D) An atom of sodium has 14 protons, 16 electrons, and 16 neutrons.1.2. The ability of carbon to form four covalent bonds with a variety of atoms is(A) Isomerism(B) Tetravalence(C) Hydrolysis(D) Quadravalence1.3. Water resists changing its temperature because of its … (A) High heat of vaporization(B) Low specific heat(C) High specific heat(D) Low heat of vaporization1.4. A solution where [H+] < [OH−] is more:(A) Acidic(B) Basic(C) Neutral(D) None of the above1.5. Malnourishment results when there is(A) A diet that does not provide enough fatty acids(B) A diet with short-term absence of one or more essential nutrients(C) A diet that does not provide enough…arrow_forwardLook up the valence electron configuration, covalent atomic radius, effective nuclear charge, first ionization energy and Pauling electronegativity in Chapter 8 (tables are attached). Examine the above data and answer the following questions. a) Explain why some of the elements like TI and Pb on the lower left of the p block are metallic. b) Explain why some of the elements like C, Si in the center of the p block form covalent bonds. Explain why these bonds formed by the network of these elements (as studied in Chapter 25) tend to be unreactive. c) Explain why the noble Group 8A elements are highly unreactive gases. d) Explain why some elements like F, CI, Br etc, on the upper right of the p block are highly reactive nonmetals.arrow_forward

 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning

