
Interpretation:
The
Concept introduction:
Chromatography is the technique used to identify the components of a mixture. The division of the distance covered by the component of a compound to the distance covered by solvent in the chromatography technique is known as the
Answer to Problem 1ASA
The
Explanation of Solution
The chromatograph for a mixture that is prepared by a student is shown below.

Figure 1
The modified chromatogram with proper divided parts is shown below.
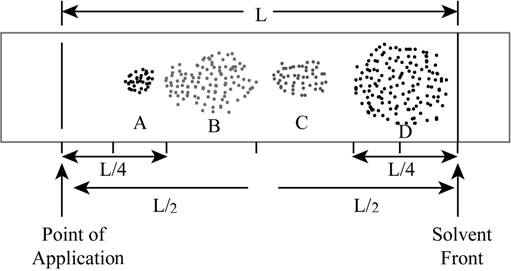
Figure 2
According to the above shown chromatogram, four compounds A, B, C and D are present in the mixture.
The total distance travelled by the solvent is
The total distance travelled by the compound A is
The total distance travelled by the compound B is
The total distance travelled by the compound C is
The total distance travelled by the compound D is
The expression to represent the
To calculate the
To calculate the
To calculate the
To calculate the
Therefore, the
The
Want to see more full solutions like this?
Chapter 2 Solutions
EBK CHEMICAL PRINCIPLES IN THE LABORATO
- A GC-MS was used to analyze an unknown sample. Based off the chromatogram, at least how many different compounds were in this sample? Detector Signal u M Retention Timearrow_forwardGas chromatography cannot analyze mixtures due to the sensitivity of the instrument. True Falsearrow_forwardIn all forms of chromatography, there is a mobile phase and a stationary phase. In thin layer chromatography what is the stationary phase?arrow_forward
- what is chromatography? Explain different types of chromatography and explain how to seperate compounds. indicate stationary phase mobile phase.arrow_forwardQno1: why there is a need of chromatography and chromatography thecniqs. B) when and where we should use normal and reverse phase chromatographyarrow_forwardUse a suitable model to explain how separation and identification of a mixture of organic compounds can be achieved with a thin layer chromatographic (TLC) technique.arrow_forward
- Paper Chromatography and TLC are examples of planar chromatography. True or FalsE?arrow_forwardQUESTION 5 In a chromatographic separation using a polar stationary phase and a mobile phase composed of 60% hexane and 40% ethyl acetate, two compounds, benzene (peak A) and toluene (peak B), are eluted. The width of peak A at its base is 1.2 min, the width of peak B at its base is 1.3 min, and the retention time of peak A is 5.2 min and peak B is 3.5 min. What is the resolution (Rs) between these two peaks and is it acceptable? R = 1.36. It is not an acceptable resolution. R = 0.74. It is an acceptable resolution. R = 1.36. It is an acceptable resolution. R = 0.74. It is not an acceptable resolution.arrow_forwardthin layer chromatography: What would happen to the Rf value of a compound if you increased the polarity of the mobile phase while using a polar stationary phase? please help me this question! Thank you a lotarrow_forward
- What is the Rf value given the following information: Height of Chromatography Paper: 5.0 cm Distance from Bottom of Paper to Origin: 1.5 cm Distance from Origin to Spot: 1.6 cm Distance from Origin to Solvent Front: 2.9 cm Your Answer: Answerarrow_forwardChromatography is a separation technique. What is the physical or chemical basis for separation of compounds during thin layer chromatography?arrow_forwardA new dye (DYE C) was developed and was analyzed against other dyes using paper chromatography. A solution 1:1 ethyl acetate: hexane was used as the eluent. The resulting chromatogram is shown at the photo. Determine the retention factor (Rf) values of each spot on dye C and what are the other dye/s present in dye C.arrow_forward
 Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole
Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT


