
(a)
Interpretation:
The possible open-chain structures for the given molecular formula,
Concept Introduction:
Hydrocarbons:
Hydrocarbons contain only carbon and hydrogen atoms which are connected together by covalent bonds such as
Saturated hydrocarbons:
Alkanes are classified under saturated hydrocarbons whose carbon atoms are connected to each other by the carbon-carbon single bonds. The general molecular formula for linear alkanes is
Unsaturated hydrocarbons:
The unsaturated hydrocarbons can be classified further into alkenes and alkynes.
Alkenes:
Alkenes are unsaturated hydrocarbon that must contain atleast one carbon-carbon double bond in it. The general molecular formula for alkenes is
The general structure of alkene is drawn here:
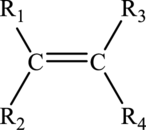
Alkynes:
Alkynes are also unsaturated hydrocarbon that must contain a carbon-carbon triple bond in it. The general molecular formula for linear alkanes is
The general structure of alkyne is shown here:

(b)
Interpretation:
The possible open-chain structures for the given molecular formula,
Concept Introduction:
Refer to part (a).
(c)
Interpretation:
The possible open-chain structures for the given molecular formula,
Concept Introduction:
Refer to part (a).
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
EBK FOUNDATIONS OF COLLEGE CHEMISTRY
- 1: In the sweetening MEROX process for kerosene the mercaptans are converted into (a) Sulpher (b) Disulphide (c) Hydrogen sulphide (d) None of these 2: Which of the following products contain maximum sulphur? (a) Diesel fuel (b) Fuel oil (c) Jet fuel (d) LPG 3: Which of the following petroleum product has a maximum C/H ratio (by weight)? (a) Light diesel oil (b) Fuel oil (c) Naphtha (d) Heating oil 4: Which of the following hydrocarbon are most desirable in gasoline? (a) Paraffins (b) Isoparaffins (c) Naphthenes (d) Aromatic 5: Octane no. of paraffins: (a) Remain constant with change in the number of carbon atoms (b) Increases with increase in the number of carbon atoms (c)Decreases with increase in the number of carbon atoms (d)None of the above 6: Which of the following hydrocarbons has a maximum octane number? (a) Benzene (b) Cyclohexane (c) Hexane (d) Iso-hexane 7: Which of the following petroleum products has a minimum flashpoint? (a)Gasoline (b) Kerosene (c) Fuel oil (d) Heating…arrow_forwardDraw an example of a C6H10 compound with conjugated double bonds.arrow_forwardWrite Lewis structures and describe the molecular geometry at each carbon atom in the following compounds:(a) cis-3-hexene(b) cis-1-chloro-2-bromoethene(c) 2-pentyne(d) trans-6-ethyl-7-methyl-2-octenearrow_forward
- This question is about the chemistry of alkenes, which are unsaturated hydrocarbons. (a) State what is meant by the term unsaturated as applied to a hydrocarbon. (1) (b) An organic compound, X, is an unsaturated hydrocarbon with molecular formula CH₂. (i) Draw the displayed formulae and give the names of two molecules with molecular formula C₂H, which are E/Z isomers. (3) Isomer 1 Isomer 2 Name: Name:arrow_forward3. Draw structures for the following: (a) an acid and an ester having the formula C 2 H 4O 2 (b) 2-alkenes having the formula C 5H 10 (3 isomers) (c) 1° amines having the formula C 4 H 11 N (4 isomers)arrow_forwardDraw the structure of a hydrocarbon with molecular formula C6H10 that also contains: (a) a carbon–carbon triple bond; (b) two carbon–carbon double bonds; (c) one ring and one C=C.arrow_forward
- Draw the bond line structures for C5H12.arrow_forwardCyclopropane (C3H6, a three-membered ring) is more reactive than most other cycloalkanes.(a) Draw a Lewis structure for cyclopropane.(b) Compare the bond angles of the carbon atoms in cyclopropane with those in an acyclic (noncyclic) alkane.(c) Suggest why cyclopropane is so reactive.arrow_forward9) In the boxes provided, draw a structure of C5H8O2 that has no formal charges and that has: a) 2 alcohols and at least 1 ring b) ester and alkene c) alkynearrow_forward
- Draw Lewis structures for the following compounds, andpredict which member of each pair will form hydrogen bonds:(a) NH₃ or AsH₃(b) CH₄ or H₂Oarrow_forwardThe structural formula for the open-chain form of galactose is CH Н—ҫ—ОН Но—С—н НО—С—Н Н—ҫ—ОН CH-ОН (a) Is this molecule a sugar? (b) How many chiral carbons are present in the molecule? (C) Draw the structure of the six-member-ring form of this molecule.arrow_forwardA certain hydrocarbon has a molecular formula of C5H8. Which of the following is not a structural possibility for this hydrocarbon? (a) It is a cycloalkane. (b) It contains one ring and one double bond. (c) It contains two double bonds and no rings. (d) It is an alkyne.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

