
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19, Problem 68P
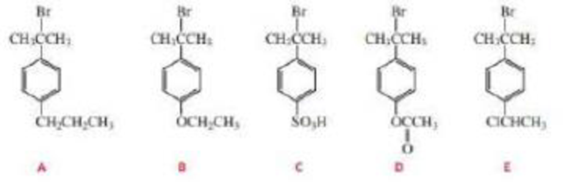
The following tertiary alkyl bromides undergo an SN1 reaction in aqueous acetone to form the corresponding tertiary alcohols. Rank the alkyl bromides from most reactive to least reactive.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw the structures of the initially formed enol tautomers in the reactions of propyne and dicyclohexylethyne with dicyclohexylborane followed by NaOHNaOH and H2O2H2O2
single reaction sequence: a certain ketone undergoes alkylation to give new ketone, when reacted with a base and then an alkylating agent, 1-bromopropane. What is the structure of the final ketone product?
The following tertiary alkyl bromides undergo an SN1 reaction in aqueous acetone to form the corresponding tertiary alcohols. Rank the alkyl bromidesfrom most reactive to least reactive.
Chapter 19 Solutions
Organic Chemistry
Ch. 19.1 - Draw the structure for each of the following: a....Ch. 19.2 - PROBLEM 2
If electrophilic addition to benzene is...Ch. 19.4 - Why does hydration inactivate FeBr3?Ch. 19.6 - Prob. 5PCh. 19.7 - Prob. 6PCh. 19.8 - What is the major product of a Friedel-Crafts...Ch. 19.10 - Describe two ways to prepare each of the following...Ch. 19.12 - Prob. 9PCh. 19.13 - Name the following:Ch. 19.13 - Prob. 12P
Ch. 19.13 - Prob. 13PCh. 19.13 - Prob. 14PCh. 19.14 - Prob. 15PCh. 19.14 - List the compounds in each set from most reactive...Ch. 19.15 - Prob. 18PCh. 19.15 - What product(s) result from nitration of each of...Ch. 19.15 - Prob. 20PCh. 19.16 - Which acid in each of the following pairs is...Ch. 19.16 - Prob. 23PCh. 19.16 - Prob. 24PCh. 19.18 - Show how the following compounds can be...Ch. 19.18 - Give the products, if any, of each of the...Ch. 19.19 - a. Does a coupling reaction have to be used to...Ch. 19.19 - PROBLEM 28
Show how each of the following...Ch. 19.20 - What is the major product(s) of each of the...Ch. 19.20 - Prob. 30PCh. 19.21 - Why isn't FeBr3 used as a catalyst in the first...Ch. 19.21 - Prob. 33PCh. 19.21 - Write the sequence of steps required for the...Ch. 19.21 - Prob. 35PCh. 19.22 - What product is formed from reaction of...Ch. 19.22 - Prob. 37PCh. 19.22 - Draw the structure of the activated ring and the...Ch. 19.23 - Prob. 39PCh. 19.23 - Prob. 40PCh. 19.23 - Diazomethane can be used to convert a carboxylic...Ch. 19.24 - Prob. 42PCh. 19.24 - Prob. 43PCh. 19.24 - Prob. 44PCh. 19.25 - Prob. 45PCh. 19 - Draw the structure for each of the following: a....Ch. 19 - Prob. 47PCh. 19 - Prob. 48PCh. 19 - Prob. 49PCh. 19 - For each of the statements in Column I, choose a...Ch. 19 - What product is obtained from the reaction of...Ch. 19 - Draw the product(s) of each of the following...Ch. 19 - Rank the following substituted anilines from most...Ch. 19 - Prob. 54PCh. 19 - The compound with the 1H NMR spectrum shown below...Ch. 19 - Prob. 56PCh. 19 - Show how the following compounds can be...Ch. 19 - Prob. 58PCh. 19 - Rank each group of compounds from most reactive to...Ch. 19 - Prob. 60PCh. 19 - Describe two ways to prepare anisole from benzene.Ch. 19 - For each of the following components, indicate the...Ch. 19 - Prob. 63PCh. 19 - Prob. 64PCh. 19 - Prob. 65PCh. 19 - Prob. 66PCh. 19 - An aromatic hydrocarbon with a molecular formula...Ch. 19 - The following tertiary alkyl bromides undergo an...Ch. 19 - Show how the following compounds can be...Ch. 19 - Use the four compounds shown below to answer the...Ch. 19 - a. Rank the following esters from most reactive to...Ch. 19 - A mixture of 0.10 mol benzene and 0.10 mol...Ch. 19 - Prob. 73PCh. 19 - Benzene underwent a Friedel-Crafts acylation...Ch. 19 - Prob. 75PCh. 19 - Prob. 76PCh. 19 - Prob. 77PCh. 19 - Friedel-Crafts alkylations can be carried out with...Ch. 19 - Show how the following compounds can be prepared...Ch. 19 - Prob. 80PCh. 19 - Prob. 81PCh. 19 - a. Describe four ways the following reaction can...Ch. 19 - Propose a mechanism for each of the following...Ch. 19 - Prob. 84PCh. 19 - Describe how naphthalene can he prepared from the...Ch. 19 - Prob. 86PCh. 19 - Using resonance contributors for the carbocation...Ch. 19 - Prob. 88PCh. 19 - When heated with chromic acid, compound A forms...Ch. 19 - Prob. 90PCh. 19 - Prob. 91PCh. 19 - What reagents are required to carry out the...Ch. 19 - Show how the following compounds can be prepared...Ch. 19 - An unknown compound reacts with ethyl chloride and...Ch. 19 - How can you distinguish the following compounds...Ch. 19 - P-Fluoronitrobenzene is more reactive toward...Ch. 19 - a. Explain why the following reaction leads to the...Ch. 19 - Describe how mescaline can be synthesized from...Ch. 19 - Propose a mechanism for the following reaction...Ch. 19 - Explain why hydroxide ion catalyzes the reaction...Ch. 19 - Propose a mechanism for each of the following...Ch. 19 - Prob. 102PCh. 19 - Prob. 103PCh. 19 - Describe how 3-methyl-1-phenyl-3-pentanol can he...Ch. 19 - a. How can aspirin be synthesized from benzene? b....Ch. 19 - Prob. 106PCh. 19 - Show how Novocain, a painkiller used frequently by...Ch. 19 - Prob. 108PCh. 19 - Saccharin, an artificial sweetener, is about 300...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- If phenoxide ion is allowed to react with 1-bromopentane, pentyl phenyl ether is obtained. However, if cyclohexane is used as the alkyl halide, the major products are phenol and cyclohexene. Explain how these products were formed.arrow_forwardPlease provide the reagents for the following transformations.arrow_forwardWhen cyclohexene is reacted with hydrobromic acid in acetic acid, the major product is bromocyclohexane. There is a small amount of cyclohexyl acetate formed. What is the mechanism that forms both compounds? What is the purification procedure that isolates both compounds?arrow_forward
- Which of the following undergoes the most rapid reaction with HBr to give the corresponding alkyl bromide? Methanol Ethanol Isopropanol tert - Butyl alcoholarrow_forwardA synthetic organic molecule, G, which contains both aldehyde and ether functional groups, is subjected to a series of reactions in a multi-step synthesis pathway. In the first step, G undergoes a Wittig reaction, leading to the formation of an alkene, H. Subsequently, H is treated with an ozone (O3) reagent followed by a reducing agent in an ozonolysis reaction, resulting in the formation of two different products, I and J. Considering the functional groups present in G and the nature of the reactions involved, what are the most probable structures or functional groups present in products I and J? A. I contains a carboxylic acid group, and J contains an aldehyde group. B. I contains a ketone group, and J contains an alcohol group. C. I and J both contain aldehyde groups. D. I contains an ester group, and J contains a ketone group. Don't use chat gpt.arrow_forwardWhat reagents should be used to carry out the following syntheses?arrow_forward
- Nonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardA problem often encountered in the oxidation of primary alcohols to acids is that esters are sometimes produced as by-products. For example, oxidation of ethanol yields acetic acid and ethyl acetate: Propose a mechanism to account for the formation of ethyl acetate. Take into account the reversible reaction between aldehydes and alcohols:arrow_forwardEthylene oxide is the starting material for the synthesis of 1,4-dioxane. Propose a mechanism for each step in this synthesis.arrow_forward
- Cyclohexene can be converted to 1-cyclopentenecarbaldehyde by the following series of reactions. Propose a structural formula for each intermediate compound.arrow_forwardThe following molecule can undergo keto–enol tautomerization upon addition of hydroxide. Draw the structure of the enol form and the mechanism by which it forms.arrow_forwardTreatment of cis-4-bromocyclohexanol with HO– affords compound A and cyclohex-3-en-1-ol. Treatment of trans-4- bromocyclohexanol under the same conditions forms compound B and cyclohex-3-en-1-ol. A and B contain different functional groups and are not isomers of each other. Propose structures for A and B and offer an explanation for their formation.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY