
Organic Chemistry
4th Edition
ISBN: 9780073402772
Author: Janice G. Smith
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 19, Problem 19.47P
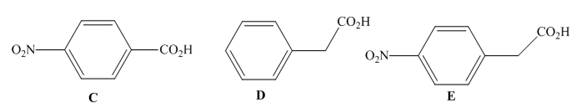
Rank the following compounds in order of increasing acidity and explain in detail your choice of order.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Don't used Ai solution
Don't used Ai solution
Please correct answer and don't used hand raiting
Chapter 19 Solutions
Organic Chemistry
Ch. 19 - Give the IUPAC name for each compound.Ch. 19 - Problem 19.2 Give the structure corresponding to...Ch. 19 - Problem 19.3 Draw the structure corresponding to...Ch. 19 - Prob. 19.4PCh. 19 - Prob. 19.5PCh. 19 - Rank the following compounds in order of...Ch. 19 - Explain how you could use IR spectroscopy to...Ch. 19 - Identify the structure of a compound of molecular...Ch. 19 - Problem 19.9 How many tetrahedral stereogenic...Ch. 19 - What alcohol can be oxidized to each carboxylic...
Ch. 19 - Prob. 19.11PCh. 19 - Draw the products of each acid-base reaction.Ch. 19 - Problem 19.14 Given the values in Appendix A,...Ch. 19 - Problem 19.15 Rank the labeled protons in...Ch. 19 - Problem 19.16 Match each of the following values ...Ch. 19 - Rank the compounds in each group in order of...Ch. 19 - Rank the compounds in each group in order of...Ch. 19 - Prob. 19.18PCh. 19 - Which of the following pairs of compounds can be...Ch. 19 - Problem 19.21 Two other commonly used sulfonic...Ch. 19 - Problem 19.22 Draw both enantiomers of each amino...Ch. 19 - Problem 19.23 Explain why amino acids, unlike most...Ch. 19 - Problem 19.24 Draw the positively charged,...Ch. 19 - Prob. 19.24PCh. 19 - Problem 19.26 Explain why the of the group of...Ch. 19 - Answer each question for A and B depicted in the...Ch. 19 - Prob. 19.27PCh. 19 - Give the IUPAC name for each compound. a....Ch. 19 - Prob. 19.29PCh. 19 - Draw the structures and give the IUPAC names for...Ch. 19 - Prob. 19.31PCh. 19 - Rank the compounds in each group in order of...Ch. 19 - 19.33 Draw the organic products formed in each...Ch. 19 - 19.34 Identify the lettered compounds in each...Ch. 19 - 19.35 Using the table in Appendix A, determine...Ch. 19 - Draw the products of each acid-base reaction, and...Ch. 19 - Which compound in each pair has the lower pKa?...Ch. 19 - 19.38 Rank the compounds in each group in order of...Ch. 19 - Rank the compounds in each group in order of...Ch. 19 - 19.40 Match the values to the appropriate...Ch. 19 - Prob. 19.41PCh. 19 - 19.42 Which carboxylic acid has the lower ,...Ch. 19 - Prob. 19.43PCh. 19 - 19.44 Explain the following statement. Although...Ch. 19 - Prob. 19.45PCh. 19 - 19.46 Explain why the of compound A is lower than...Ch. 19 - 19.47 Rank the following compounds in order of...Ch. 19 - Explain the following result. Acetic acid...Ch. 19 - 19.50 Draw all resonance structures of the...Ch. 19 - As we will see in Chapter 23, CH bonds are...Ch. 19 - Prob. 19.51PCh. 19 - The pKa of acetamide (CH3CONH2) is 16. Draw the...Ch. 19 - 19.54 Write out the steps needed to separate...Ch. 19 - Prob. 19.54PCh. 19 - Can octane and octan -1- ol be separated using an...Ch. 19 - 19.57 Identify each compound from its spectral...Ch. 19 - 19.58 Use the NMR and IR spectra given below to...Ch. 19 - 19.59 An unknown compound (molecular formula )...Ch. 19 - 19.60 Propose a structure for (molecular formula...Ch. 19 - Prob. 19.60PCh. 19 - 19.61 Match the data to the appropriate...Ch. 19 - Prob. 19.62PCh. 19 - Prob. 19.63PCh. 19 - Prob. 19.64PCh. 19 - 19.65 For each amino acid ,draw its neutral,...Ch. 19 - Calculate the isoelectric point for each amino...Ch. 19 - 19.67 Lysine and tryptophan are two amino acids...Ch. 19 - Prob. 19.68PCh. 19 - Prob. 19.69PCh. 19 - Prob. 19.70PCh. 19 - Prob. 19.71PCh. 19 - 19.71 Hydroxy butanedioic acid occurs naturally in...Ch. 19 - 19.72 Although it was initially sold as a rat...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- (12) Which one of the following statements about fluo- rometry is FALSE? a) Fluorescence is better detected at 90 from the exci- tation direction. b) Fluorescence is typically shifted to longer wave- length from the excitation wavelength. c) For most fluorescent compounds, radiation is pro- duced by a transitionarrow_forwardDon't used Ai solutionarrow_forwardDon't used Ai solutionarrow_forward
- Don't used Ai solutionarrow_forwardIndicate the correct option.a) Graphite conducts electricity, being an isotropic materialb) Graphite is not a conductor of electricityc) Both are falsearrow_forward(f) SO: Best Lewis Structure 3 e group geometry:_ shape/molecular geometry:, (g) CF2CF2 Best Lewis Structure polarity: e group arrangement:_ shape/molecular geometry: (h) (NH4)2SO4 Best Lewis Structure polarity: e group arrangement: shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles): Sketch (with angles):arrow_forward
- 1. Problem Set 3b Chem 141 For each of the following compounds draw the BEST Lewis Structure then sketch the molecule (showing bond angles). Identify (i) electron group geometry (ii) shape around EACH central atom (iii) whether the molecule is polar or non-polar (iv) (a) SeF4 Best Lewis Structure e group arrangement:_ shape/molecular geometry: polarity: (b) AsOBr3 Best Lewis Structure e group arrangement:_ shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles):arrow_forward(c) SOCI Best Lewis Structure 2 e group arrangement: shape/molecular geometry:_ (d) PCls Best Lewis Structure polarity: e group geometry:_ shape/molecular geometry:_ (e) Ba(BrO2): Best Lewis Structure polarity: e group arrangement: shape/molecular geometry: polarity: Sketch (with angles): Sketch (with angles): Sketch (with angles):arrow_forwardDon't used Ai solutionarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY