
Concept explainers
Draw the products of each acid-base reaction, and using the
a. 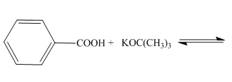 d.
d. 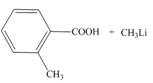
b. 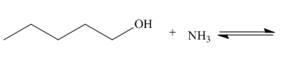 e.
e.
c. 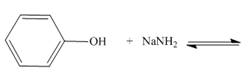 f..
f.. 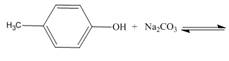
(a)
Interpretation: The products of the given acid-base reaction are to be drawn. Whether the equilibrium favors the reactants or the product is to be determined.
Concept introduction: An acidic substance in a reaction donates a proton, whereas base in a reaction accepts a proton. The
Answer to Problem 19.36P
The products of the given acid-base reaction are,
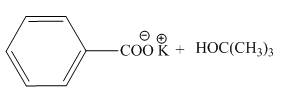
The
Explanation of Solution
The products of the given acid-base reaction are shown below.
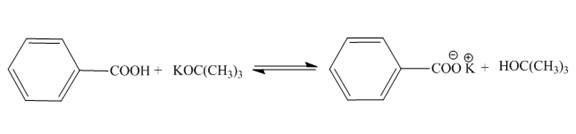
Figure 1
The
The products of the given acid-base reaction are shown in Figure 1. The
(b)
Interpretation: The products of the given acid-base reaction are to be drawn. The equilibrium favors the reactants or a product is to be determined.
Concept introduction: An acidic substance in a reaction donates a proton, whereas base in a reaction accepts a proton. The
Answer to Problem 19.36P
The products of the given acid-base reaction are,
The
Explanation of Solution
The products of the given acid-base reaction are shown below.

Figure 2
The
The products of the given acid-base reaction are shown in Figure 2. The
(c)
Interpretation: The products of the given acid-base reaction are to be drawn. Whether the equilibrium favors the reactants or the product is to be determined.
Concept introduction: An acidic substance in a reaction donates a proton, whereas base in a reaction accepts a proton. The
Answer to Problem 19.36P
The products of the given acid-base reaction are,
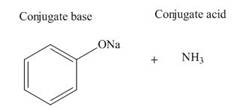
The
Explanation of Solution
The products of the given acid-base reaction are shown below.
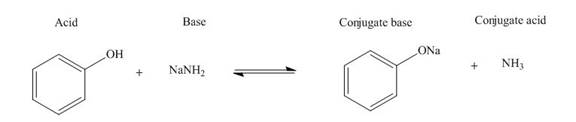
Figure 3
The
The products of the given acid-base reaction are shown in Figure 3. The
(d)
Interpretation: The products of the given acid-base reaction are to be drawn. Whether the equilibrium favors the reactants or the product is to be determined.
Concept introduction: An acidic substance in a reaction donates a proton, whereas base in a reaction accepts a proton. The
Answer to Problem 19.36P
The products of the given acid-base reaction are,
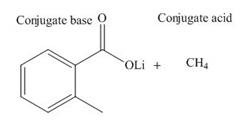
The
Explanation of Solution
The products of the given acid-base reaction are shown below.
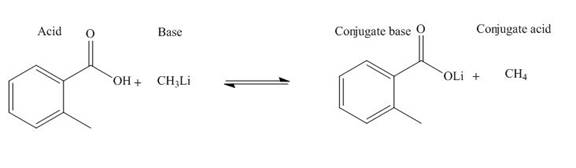
Figure 3
The
The products of the given acid-base reaction are shown in Figure 3. The
(e)
Interpretation: The products of the given acid-base reaction are to be drawn. Whether the equilibrium favors the reactants or the product is to be determined.
Concept introduction: An acidic substance in a reaction donates a proton, whereas base in a reaction accepts a proton. The
Answer to Problem 19.36P
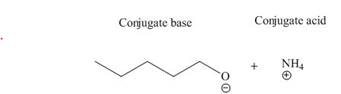
The products of the given acid-base reaction are
The
Explanation of Solution
The products of the given acid-base reaction are,
The
The products of the given acid-base reaction are
(f)
Interpretation: The products of the given acid-base reaction are to be drawn. Whether the equilibrium favors the reactants or the product is to be determined.
Concept introduction: An acidic substance in a reaction donates a proton, whereas base in a reaction accepts a proton. The
Answer to Problem 19.36P
The products of the given acid-base reaction are,
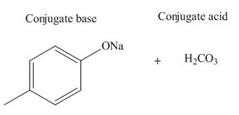
The
Explanation of Solution
The products of the given acid-base reaction are shown below.
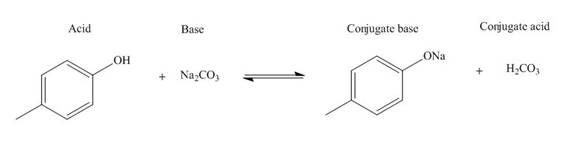
Figure 4
The
The products of the given acid-base reaction are shown in Figure 4. The
Want to see more full solutions like this?
Chapter 19 Solutions
Organic Chemistry-Package(Custom)
- 8. How many curved arrows are needed for the following reaction? H₂C NH₂ H₂C NH₂ ܒܘ H a. 4 b. 5 c. 6 d. 7arrow_forwardA. F- behaves as a Bronsted Lowry base F- + H2O -> ? + ? B. H2CO3 behaves as a Bronsted Lowry acid H2CO3 + H2O -> ? + ?arrow_forwardWhat is the conjugate acid of C6H₂O¯ ? A. B. C6H402- C₂H4O C. CH₂OH ODCH₂OH O E. C6H5OH3+arrow_forward
- Which acid is the strongest? A. H2SO3 B. H3AsO3 C. H2SeO3arrow_forwarddentify the conjugate base in the reaction of propanoic acid CH3CH3COOH reacting with methyl amine, CH3NH2. a. CH3NH2 b. CH3CH3COOH c. CH3NH3+ d. OH-1 e. CH3CH3COO-1arrow_forwardDraw the products of the following acid base reactions and indicate whether products or reactants are favored. CH;CH,00 a. CF3CH2OH + b. H2S Но C. CH3-C=C: H-C d. N-H N-H е. CH3MGB + CH3 1arrow_forward
- Which is a stronger base?a. HS− or HO− b. CH3O− or CH3N-H c. CH3OH or CH3O− d. Cl− or Br−e. CH3COO− or CF3COO− f. CH3CHClCOO− or CH3CHBrCOO−arrow_forwardWhich compound is considered as the conjugate base? * D. OH- B. C2H5NH+3 C. H2O A. C2H5NH2arrow_forwardWhich is the stronger base? a. Br- or I- b. CH3O- or CH3S-c. CH3CH2O− or CH3COO−d. H2C =CH e. FCH2CH2COO− or BrCH2CH2COO− f. ClCH2CH2O− or Cl2CHCH2O−arrow_forward
- Arrange the following carboxylic acids in order of decreasing acid strength ( strongest to weakest). A. CICH2COOH B. BRCH2COOH C. FCH2COOH D. ICH2COOH OC>A> D> B O C> B> A> D OD>A> B >C O C>A> B> Darrow_forwardConsider the acid-base reaction below: Ca(HCO3)2 + Ca(OH)2 ----> Compound A + H2O (OR Ca(HCO3)2 + Ca left parenthesis OH right parenthesis 2 ----> Compound A + H2O) One of the single ions that is used to form compound A is ____.arrow_forwarda. b. C. d. MeO لله OEt OCH3 CI 1 eq Me₂NH Pyridine 1. PhCO₂Me NaOEt EtOH 2. aq H* 1. PrMgBr 2. aq H+ 1. LAH 2. H₂O H₂SO4 EtOH ÷[ aq H+ Aarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





