
Concept explainers
Show by a series of equations how could synthesize each of the following compounds from the indicated starting material and any necessary organic or inorganic reagents:
(a)
(b)
(c)
(d)
(e)
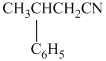
(f) 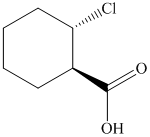 from
from 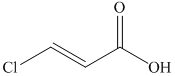
(g)
(h)
(i)
Interpretation:
The way in which each of the given compounds can be synthesized from the indicated starting material and any necessary organic or inorganic reagents is to be shown by using a series of equations.
Concept Introduction:
Hydroboration reaction is a two-step reaction that involves conversion of an alkene into alcohol. This type of reaction follows anti-Markovnikov's rule.In acid-catalyzed dehydration, a saturated compound is converted to an unsaturated compound with the removal of a water molecule in the presence of an acid catalyst.
Grignard reagent is prepared by the reaction of alkyl or aryl bromide with magnesium metal in the presence of ether.
Thionyl chloride
Lithium aluminium hydride is a strong reducing agent. The reduction of carboxylic acid by
Oxidation reaction involves increase in the
Answer to Problem 18P
Solution:
a)The reaction that shows the preparation of
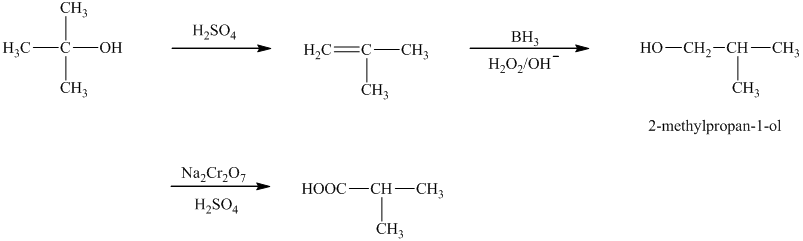
b)The reaction that shows the preparation of
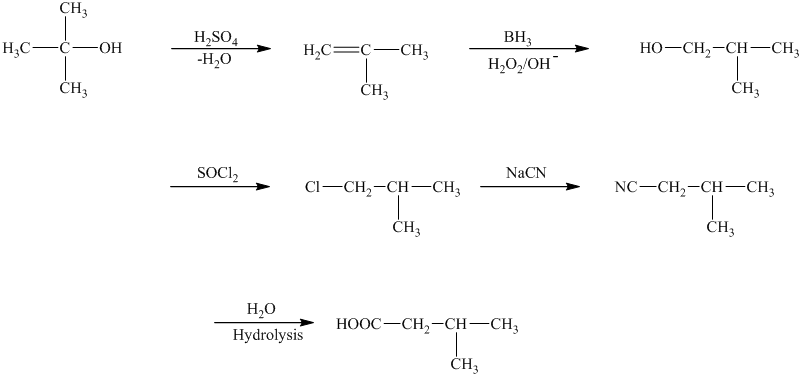
c)The reaction that shows the preparation of
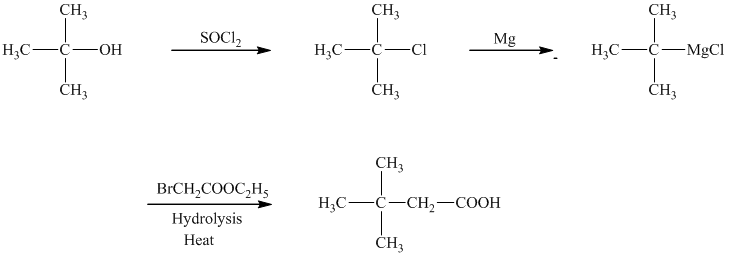
d)The reaction that shows the preparation of
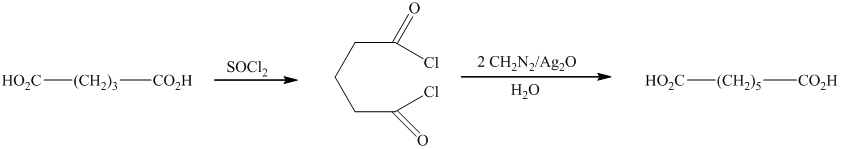
e) The reaction that shows the preparation of
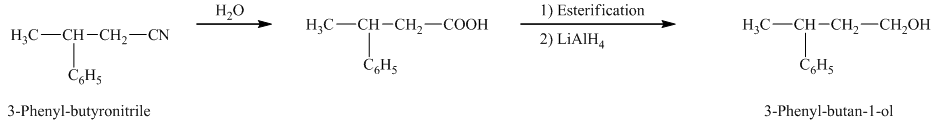
f) The reaction that shows the preparation of
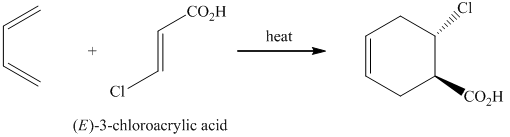
g) The reaction that shows the preparation of
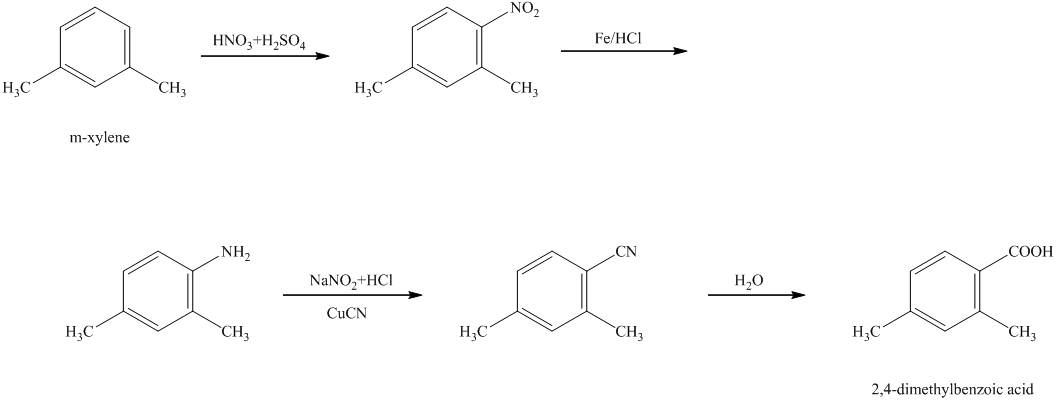
h) The reaction that shows the preparation of
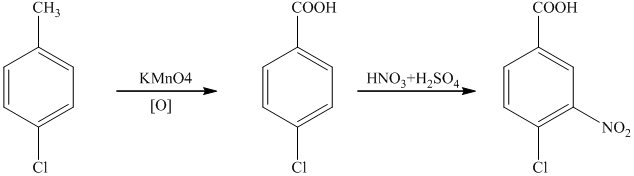
i) The reaction that shows the preparation of
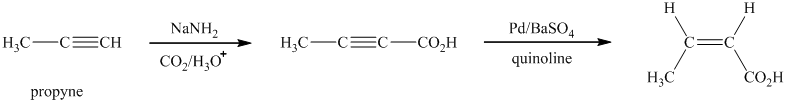
Explanation of Solution
a)
In the synthesis of
Thus, the reaction that shows the preparation of

b)
The reaction that shows the preparation of
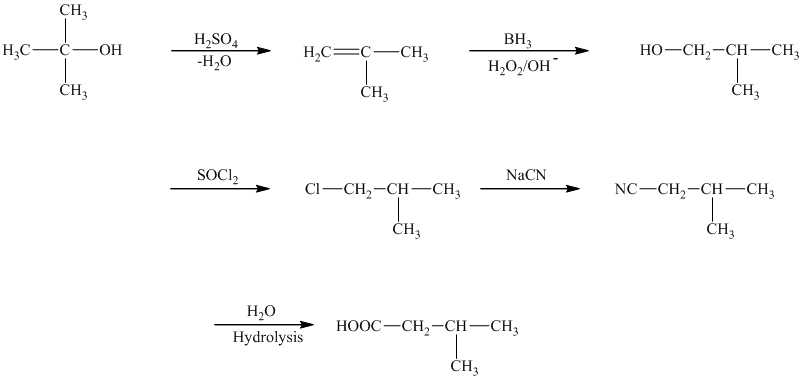
The first step of the required synthesis is the reaction of acid with the given alcohol. This step is the dehydration of alcohol. In the next step, hydroboration of alkene is done, which is then followed by hydrolysis. This results in the formation of
Thus, the required acid is synthesized.
c)
In the required synthesis, the first step is the reaction of the given alcohol with
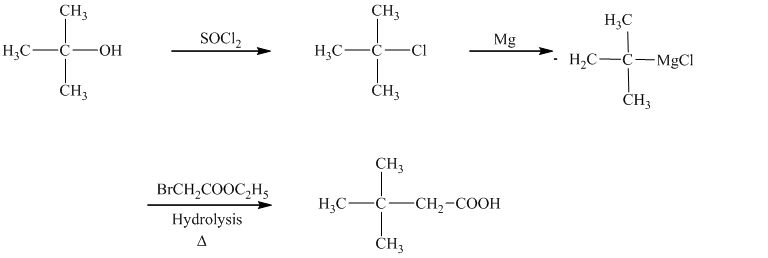
d)
In this synthesis, the first step is the reaction of the given carboxylic acid with thionyl chloride to form acyl chloride. In the next step, acyl chloride reacts with diazomethane to form diazoketones. In the last step of the synthesis reaction, the diazoketones give the final product in the presence of metal catalyst and water. The given reaction is an example of Arndt-Eistert synthesis.
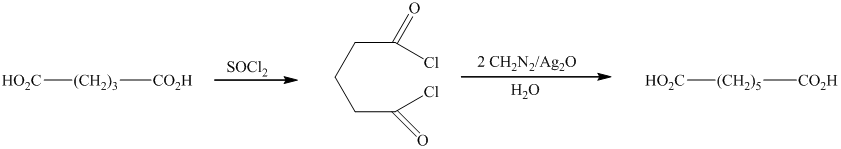
Therefore, the required product is synthesized.
e) 
In the required synthesis, the first step is the hydrolysis of
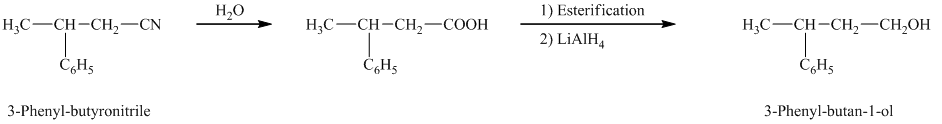
Therefore, the required product is synthesized.
f) 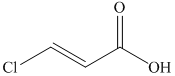
The reaction of
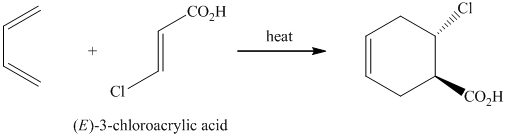
Therefore, the required product is synthesized.
g)
In the required synthesis, the first step is the nitration of
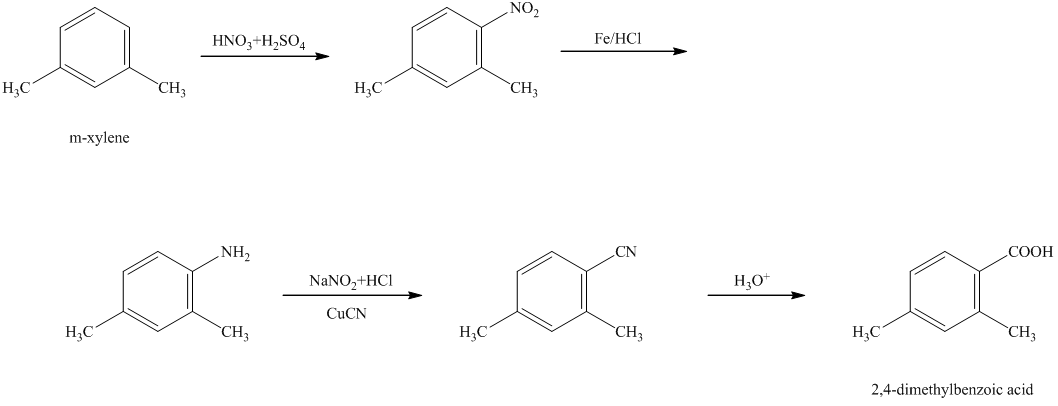
Thus, the required product was synthesized.
h)
In the synthesis of
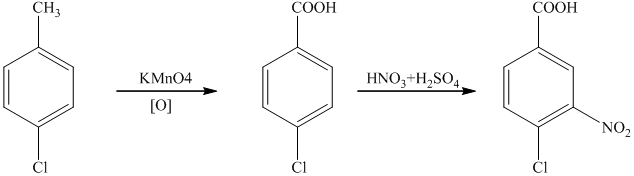
Therefore, the given compound was synthesized.
i)
Sodium amide is a strong base and acts as s strong nucleophile.The reaction that shows the preparation of
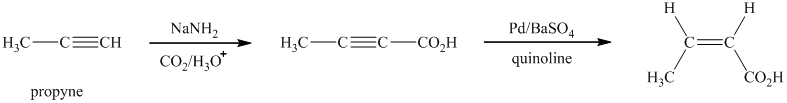
In the above synthesis, the first step is the reaction of
Want to see more full solutions like this?
Chapter 19 Solutions
ORGANIC CHEMISTRY (LL)-W/SOLN.>CUSTOM<
- Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.(a) PhMgBr, then H3O+ (b) Tollens reagent (c) semicarbazide and weak acid(d) excess ethanol and acid (e) propane-1,3-diol, H+ (f) zinc amalgam and dilute hydrochloric acidarrow_forwardNonconjugated , -unsaturated ketones, such as 3-cyclohexenone, are in an acid-catalyzed equilibrium with their conjugated , -unsaturated isomers. Propose a mechanism for this isomerization.arrow_forwardOne frequently used method for preparing methyl esters is by reaction of carboxylic acids with diazomethane, CH2N2. The reaction occurs in two steps: (l) protonation of diazomethane by the carboxylic acid to yield methyldiazonium ion, CH3N2+, plus a carboxylate ion; and (2) reaction of the carboxylate ion with CH3N2+. (a) Draw two resonance structures of diazomethane, and account for step 1. (b) What kind of reaction occurs in step 2?arrow_forward
- Following is the structural formula of the tranquilizer meparfynol (Oblivon). Propose a synthesis for this compound starting with acetylene and a ketone. (Notice the -yn- and -ol in the chemical name of this compound, indicating that it contains alkyne and hydroxyl functional groups.)arrow_forwardClaisen condensation between diethyl phthalate and ethyl acetate followed by saponification, acidification, and decarboxylation forms a diketone, C9H6O2. Propose structural formulas for compounds A and B and the diketone.arrow_forwardCyclopentanecarboxylic acid and 4-hydroxycyclohexanone have the same formula (C6H10O2), and both contain an —OH and a C=O group. How could you distinguish between them using IR spectroscopy?arrow_forward
- The following questions concern ethyl (2-oxocyclohexane)carboxylate.(a) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by a Dieckmann cyclization.(b) Write a chemical equation showing how you could prepare ethyl (2-oxocyclohexane)-carboxylate by acylation of a ketone.(c) Write structural formulas for the two most stable enol forms of ethyl (2-oxocyclohexane)carboxylate.(d) Write the three most stable resonance contributors to the most stable enolate derived from ethyl (2-oxocyclohexane)carboxylate.(e) Show how you could use ethyl (2-oxocyclohexane)carboxylate to prepare 2-methylcyclohexanone.(f) Give the structure of the product formed on treatment of ethyl (2-oxocyclohexane)-carboxylate with acrolein (H2C=CHCH=O) in ethanol in the presence of sodium ethoxidearrow_forwardHow could you convert butanoic acid into the following compounds? Write each step showing the reagents needed. (a) 1-Butanol (b) 1-Bromobutane (c) Pentanoic acid (d) 1-Butene (e) Octanearrow_forwardWhich of the following reactions will synthesize phenol from benzene? 1) HNO3 + H2SO4; 2) Fe, HCl; 3) NaNO2, HCl, 0-10 oC; 4) warm H2SO4 and H2O 1) HNO3 + H2SO4; 2) Fe, HCl; 3) NaNO2, HCl, 0-10 oC; 4) CuCN; 5) dilute acid and heat 1) Acetyl chloride & AlCl3; 2) bleach 1) Ph-N2+ + KI; 2) BrMgCH=CH2 in ether, followed by H3O+; 3) warm, conc'd KMnO4 1) Cl-CH(CH3)-CH2CH2CH3 + FeBr3; 2) hot, conc'd KMnO4arrow_forward
- Use a Grignard reaction to prepare the following alcohols: (a) 2-Methyl-2-propanol (b) 1-Methylcyclohexanol (c) 3-Methyl-3-pentanol (d) 2-Phenyl-2-butanol (e) Benzyl alcohol (f) 4-Methyl-1-pentanolarrow_forward1. How would you prepare the following alkyl halides from the appropriate alcohols? (a) ÇI (b) Br CH3 CH3CH3 CH;CHCH,CHCH3 ČH3 2. Predict the products of the following reaction: (a) он CH3 (b) H2C CH;CH,CHCH,CHCH, Per, socl2. PBr3 ? H3Carrow_forwardA postgraduate student wanted to synthesized two carbonyl compounds known as compounds M and N using oxidation of alcohol. However, these carbonyl compounds should have more than 6 carbon atoms Draw two (2) possible structural formulae for compounds M and N. How would you distinguish between M and N?arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

