
a)
Interpretation:
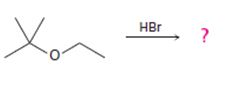
The products formed and the mechanism by which they are formed when tert-butyl ethyl ether is treated with HBr is to be given.
Concept introduction:
Ethers are cleaved by strong acids. The cleavage takes place either by SN1 or SN2 mechanisms, depending upon the structure of the substrate. Ethers with only primary and secondary alkyl groups react by SN2 mechanism. The Br- or I- attacks the protonated ether at the less hindered side to yield a single alcohol and a single
To give:
The products formed and the mechanism by which they are formed when tert-butyl ethyl ether is treated with HBr is to be given.
Answer to Problem 24MP
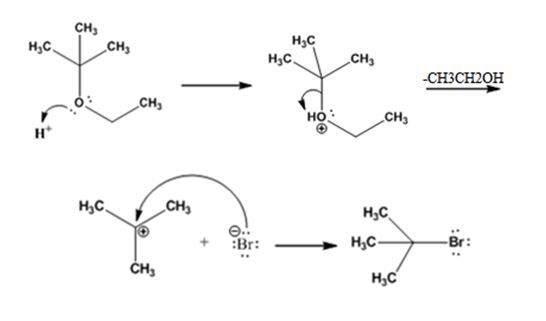
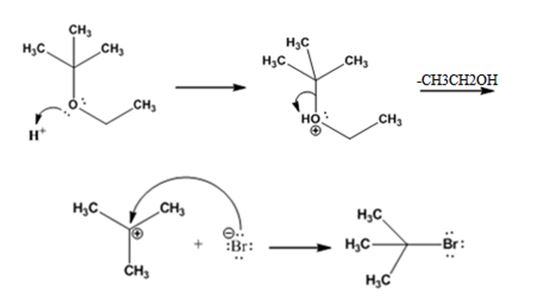
The products formed when tert-butyl ethyl ether is treated with HBr are ethanol and tert-butylbromide.
The mechanism by which they are formed is given below.
Explanation of Solution
The reaction occurs following SN1 mechanism. The acid protonates the ether initially and the protonated ether eliminates ethanol to produce a stable tert-butyl carbocation. In the next step the bromide ion attacks the carbocation to yield tert-butyl bromide as the product.
The products formed when tert-butyl ethyl ether is treated with HBr are ethanol and tert-butylbromide.
The mechanism by which they are formed is given below.
b)
Interpretation:
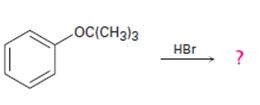
The products formed and the mechanism by which they are formed when tert-butyl phenyl ether is treated with HBr is to be given.
Concept introduction:
Ethers are cleaved by strong acids. The cleavage takes place either by SN1 or SN2 mechanisms, depending upon the structure of the substrate. Ethers with only primary and secondary alkyl groups react by SN2 mechanism. The Br- or I- attacks the protonated ether at the less hindered side to yield a single alcohol and a single alkyl halide. Ethers with a tertiary, benzylic or an allylic group cleave either by SN1 or E1 mechanism because these can produce a stable carbocations yielding alkenes and alcohols.
To give:
The products formed and the mechanism by which they are formed when tert-butyl phenyl ether is treated with HBr.
Answer to Problem 24MP
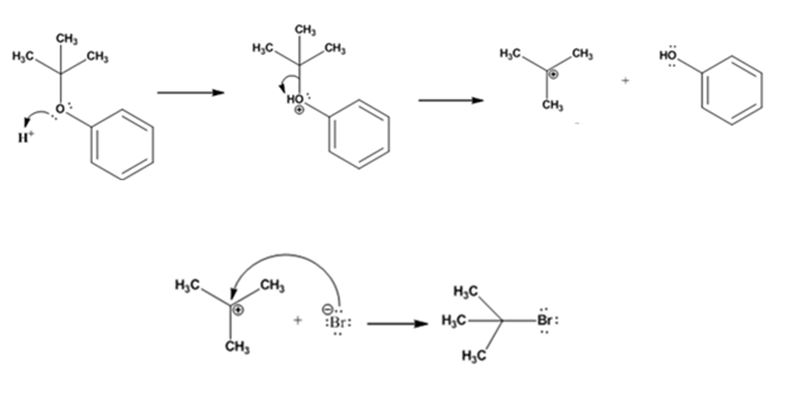
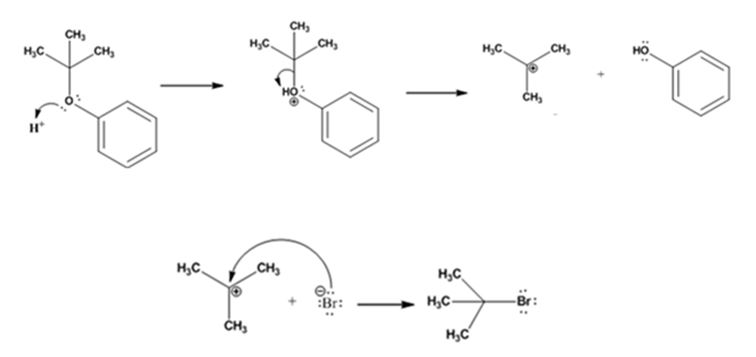
The products formed when tert-butyl phenyl ether is treated with HBr are phenol and tert-butyl bromide.
The mechanism by which they are formed is given below.
Explanation of Solution
The reaction occurs following SN1 mechanism. The acid protonates the ether initially and the protonated ether eliminates phenol to produce a stable tert-butyl carbocation. In the next step the bromide ion attacks the carbocation to yield tert-butyl bromide.
The products formed when tert-butyl phenyl ether is treated with HBr are phenol and tert-butyl bromide.
The mechanism by which they are formed is given below.
c)
Interpretation:
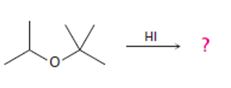
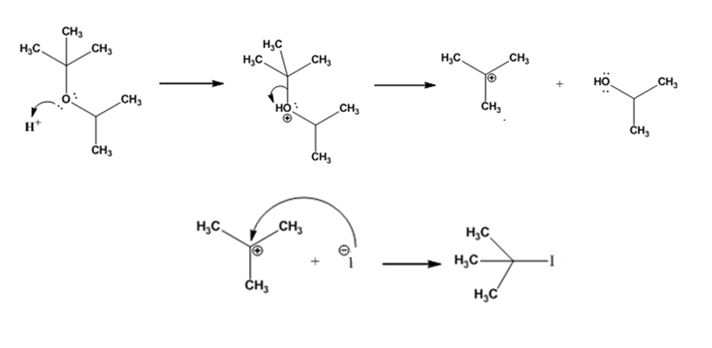
The products formed and the mechanism by which they are formed when tert-butyl isopropyl ether is treated with HI is to be given.
Concept introduction:
Ethers are cleaved by strong acids. The cleavage takes place either by SN1 or SN2 mechanisms, depending upon the structure of the substrate. Ethers with only primary and secondary alkyl groups react by SN2 mechanism. The Br- or I- attacks the protonated ether at the less hindered side to yield a single alcohol and a single alkyl halide. Ethers with a tertiary, benzylic or an allylic group cleave either by SN1 or E1 mechanism because these can produce a stable carbocations yielding alkenes and alcohols.
To give:
The products formed and the mechanism by which they are formed when tert-butyl isopropyl ether is treated with HI.
Answer to Problem 24MP
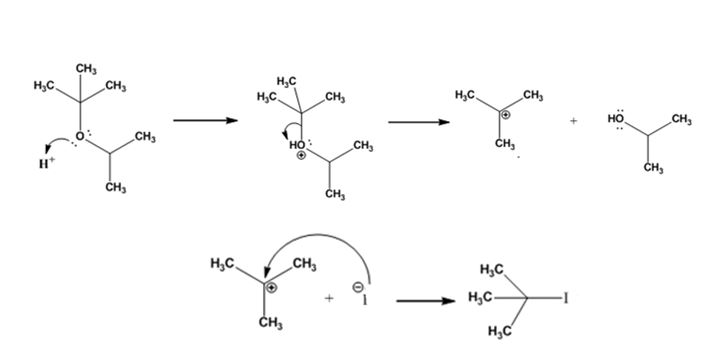
The products formed when tert-butyl isopropyl ether is treated with HI are tert-butyl bromide and 2-propanol.
The mechanism by which they are formed is given below.
Explanation of Solution
The reaction occurs following SN1 mechanism. The acid protonates the ether initially and the protonated ether eliminates 2-propanol to produce a stable tert-butyl carbocation. In the next step the iodide ion attacks the carbocation to yield tert-butyl iodide.
The products formed when tert-butyl isopropyl ether is treated with HI are tert-butyl bromide and 2-propanol.
The mechanism by which they are formed is given below.
d)
Interpretation:
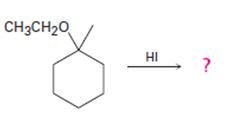
The products formed and the mechanism by which they are formed when ethyl 1-methylcyclohexyl ether is treated with HI is to be given.
Concept introduction:
Ethers are cleaved by strong acids. The cleavage takes place either by SN1 or SN2 mechanisms, depending upon the structure of the substrate. Ethers with only primary and secondary alkyl groups react by SN2 mechanism. The Br- or I- attacks the protonated ether at the less hindered side to yield a single alcohol and a single alkyl halide. Ethers with a tertiary, benzylic or an allylic group cleave either by SN1 or E1 mechanism because these can produce a stable carbocations yielding alkenes and alcohols.
To give:
The products formed and the mechanism by which they are formed when ethyl 1-methylcyclohexyl ether is treated with HI.
Answer to Problem 24MP
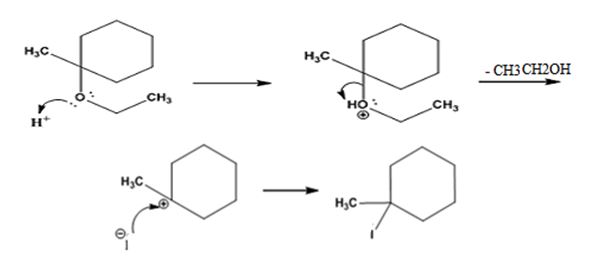
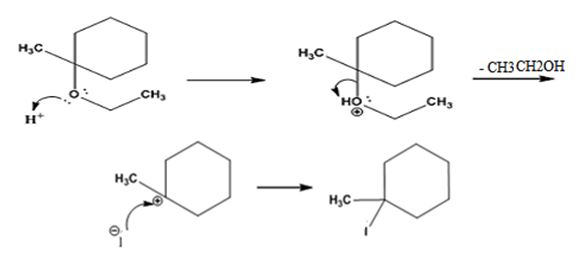
The products formed when ethyl 1-methylcyclohexyl ether is treated with HI are 1-iodo-1-methylcyclohexane and ethanol.
The mechanism by which they are formed is given below.
Explanation of Solution
The reaction occurs following SN1 mechanism. The acid protonates the ether initially and the protonated ether eliminates ethanol to produce a stable tert-butyl carbocation. In the next step the iodide ion attacks the carbocation to yield tert-butyl iodide.
The products formed when ethyl 1-methylcyclohexyl ether is treated with HI are 1-iodo-1-methylcyclohexane and ethanol.
The mechanism by which they are formed is given below.
All the reactions (a), (b), (c) and (d) occur through SN1 mechanism.
All the reactions, (a), (b), (c) and (d) take place following SN1 mechanism. The protonated
Want to see more full solutions like this?
Chapter 18 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Aldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forwardPredict the major product(s) and provide a mechanism for the reaction below. Includestereochemistry.arrow_forwardTreatment of (CH3)2CHCH(OH)CH2CH3 with TsOH affords two products (M and N) with molecular formula C6H12. The 1H NMR spectra of M and N are given below. Propose structures for M and N and draw a mechanism to explain their formation.arrow_forward
- : Treatment of (CHa)CHCH(OH)CH,CH3 with TSOH affords two products (M and N) with molecular formula CgH12. The 'H NMR spectra of M and N are given below. Propose structures for M and N and draw a mechanism to explain their formation. 1H NMR of M 3H 1H NMR of N 3H 3H 3 H 1H 3 H 2 H 2H 2H 8 7 6 4 1 0 9 8. 2 1 ppm ppm 4.arrow_forwardpredict the product for each of the following reactions and propose a mechanism that explains the formation of each product ?arrow_forwardWrite the expected substitution product(s) for each reaction and predict the mechanism by which each product is formed.arrow_forward
