
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18, Problem 25P
The African dwarf crocodile secretes a volatile substance believed to be a sex
pheromone. It is a mixture of two stereoisomers, one of which is shown:
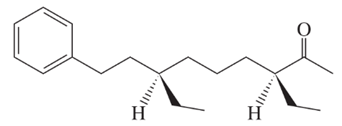
(a) Give the IUPAC name for this compound, including
(b) One component of the scent substance has the
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Coibacin B (shown below) is a natural product that exhibits potent anti-inflammatory activity and potential activity in the treatment of leishmaniasis, a disease caused by certain
parasites
(Org. Lett. 2012, 14, 3878-3881):
(a) Assign the configuration (R or S) of each chirality center (labeled A to C) in coibacin B.
(b) Identify the number of possible stereoisomers for this compound, assuming that the geometry of the alkenes are fixed. Choices are given below and write the CAPITAL
LETTER of your choice.
A. 2
В. 4
С. 8
D.
16
ANSWERS:
(a) A.
В.
C.
(b)
5-Hydroxyhexanal forms a six-membered cyclic hemiacetal, which predominates at equilibrium in aqueous solution.
(a) Draw a structural formula for this cyclic hemiacetal.
(b) How many stereoisomers are possible for 5-hydroxyhexanal?
(c) How many stereoisomers are possible for this cyclic hemiacetal?
(d) Draw alternative chair conformations for each stereoisomer and label groups axial or Also predict which of the alternative chair conformations for each stereoisomer is more stable.
Diltiazem is prescribed to treat hypertension, and simvastatin is a cholesterol-lowering drug. Locate the chirality centers in each.
Chapter 18 Solutions
Organic Chemistry - Standalone book
Ch. 18.1 - Prob. 1PCh. 18.1 - Prob. 2PCh. 18.3 - Prob. 3PCh. 18.4 - Prob. 4PCh. 18.4 - Prob. 5PCh. 18.6 - Prob. 6PCh. 18.7 - Prob. 7PCh. 18.7 - Prob. 8PCh. 18.7 - Prob. 9PCh. 18.8 - Prob. 10P
Ch. 18.8 - Prob. 11PCh. 18.8 - Prob. 12PCh. 18.9 - Prob. 13PCh. 18.10 - Prob. 14PCh. 18.10 - Prob. 15PCh. 18.11 - Problem 18.16 The product of the following...Ch. 18.11 - Prob. 17PCh. 18.12 - Problem 18.18 What other combination of ylide and...Ch. 18.12 - Prob. 19PCh. 18.12 - Prob. 20PCh. 18.12 - Prob. 21PCh. 18.13 - Prob. 22PCh. 18 - (a) Write structural formulas and provide IUPAC...Ch. 18 - Each of the following aldehydes and ketones is...Ch. 18 - The African dwarf crocodile secretes a volatile...Ch. 18 - Prob. 26PCh. 18 - Prob. 27PCh. 18 - Prob. 28PCh. 18 - Prob. 29PCh. 18 - Prob. 30PCh. 18 - Prob. 31PCh. 18 - Each of the following reaction has been reported...Ch. 18 - Prob. 33PCh. 18 - On standing in 17O-labeled water, both...Ch. 18 - Prob. 35PCh. 18 - Prob. 36PCh. 18 - The OH groups at C-4 and C-6 of methyl ...Ch. 18 - Prob. 38PCh. 18 - Prob. 39PCh. 18 - The sex attractant of the female winter moth has...Ch. 18 - Prob. 41PCh. 18 - Prob. 42PCh. 18 - Prob. 43PCh. 18 - Suggest a reasonable mechanism for each of the...Ch. 18 - Prob. 45PCh. 18 - Prob. 46PCh. 18 - Prob. 47PCh. 18 - Prob. 48PCh. 18 - Prob. 49PCh. 18 - Prob. 50PCh. 18 - Prob. 51PCh. 18 - Prob. 52DSPCh. 18 - Prob. 53DSPCh. 18 - Prob. 54DSPCh. 18 - Prob. 55DSPCh. 18 - Prob. 56DSPCh. 18 - Prob. 57DSPCh. 18 - Prob. 58DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 3) How many stereogenic centers are in the following compound:arrow_forwardFollowing are stereorepresentations for the four stereoisomers of 3-chloro-2-butanol. (a) Assign an R or S configuration to each chiral center. (b) Which compounds are enantiomers? (c) Which compound are diastereomers?arrow_forward(a) assign R or S configuration to each chiral center, (b) Which compound are enantiomers? (c) Which compounds are diastereomers?arrow_forward
- (i) What is meant by chirality of a compound? Give an example.(ii) Which one of the following compounds is more easily hydrolyzed by KOH and why?CH3CHClCH2CH3 or CH3CH2CH2Clarrow_forwardClopidogrel is the generic name for Plavix, a drug used to prevent the formation of blood clots in patients that have a history of heart attacks or strokes. A single enantiomer of clopidogrel can be prepared in three steps from the chiral α-hydroxy acid A. Identify B and C in the following reaction sequence, and designate the configuration of the enantiomer formed by this route as R or S.arrow_forwardOne commercial synthesis of flurbiprofen (the active ingredient in Ansaid and a score of other over-the-counter and prescription nonsteroidal anti-inflammatory drug preparations) gives the enantiomer shown in 94% enantiomeric excess. H CH3 он (a) Assign an R or S configuration to this enantiomer of flurbiprofen. OR What are the percentages of R and S enantiomers in the mixture? (Enter unrounded values.) (b) R: % S: 96arrow_forward
- (A) How many chirality centers does the following molecule contain? (B) How many stereoisomers are possible for this molecule? (C) Assign R,S designation to each chiral carbonarrow_forwardIdentify each stereocenter in these molecules (note that not all contain stereocenters): (a) (c) (a) OH gi (b) (c) (d) CHS 2 3 CHS (b) 1 OH 2 the he 2 (d) 3 0 NHarrow_forwardKumepaloxane is a signaling agent in snails. Using the Cahn Ingold Prelog system, determine the configuration of the chirality centers indicated in Kumepaloxane below. 4 2 Br 3 Kumepaloxanearrow_forward
- Salinosporamide A, isolated from a microbe called Salinispora tropica has shown promise as an inhibitor of cancer cell growth. Select the chiral centers in the following structure; the selected centers will appear highlighted green.arrow_forwardThe anti-inflammatory agent, (S)-naproxen sodium salt has a specific rotation of +66°. The commercial preparation of the latter agent results in a mixture that has a 97% enantiomeric excess (ee). coj H3CO Na (S)-naproxen sodium salt (a) Draw the R enantiomer of naproxen sodium salt. (b) What effect does the R enantiomer have on plane-polarized light? (c) What is the observed specific rotation of anti-inflammatory agent mixture? (d) What percentage of each enantiomer is obtained from the above commercial mixture? (e) Provide a reaction scheme for the formation of the (S)-naproxen sodium salt from "acidic" (S)-naproxen.arrow_forward1) The following compound, whose central ring is referred to as a 1,2,4-trioxane, is an anticancer agent that demonstrates activity against canine osteosarcoma. Assign the configuration of each chiral center and then draw all possible stereoisomers of this compound, showing the specific stereochemical relationship between each of your drawings. H -OH O-N O-Oarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole
Enzymes - Effect of cofactors on enzyme; Author: Tutorials Point (India) Ltd;https://www.youtube.com/watch?v=AkAbIwxyUs4;License: Standard YouTube License, CC-BY
Enzyme Catalysis Part-I; Author: NPTEL-NOC IITM;https://www.youtube.com/watch?v=aZE740JWZuQ;License: Standard Youtube License