
Concept explainers
Synthesize each compound from toluene
a.
b.
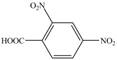 f.
f.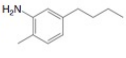
c.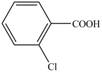 e.
e. 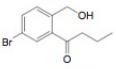 g.
g.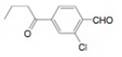
(a)
Interpretation: The synthesis of given compound from toluene and any other organic or inorganic reagent is to be stated.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.66P
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown below:
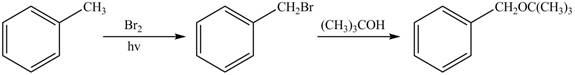
Explanation of Solution
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown below.
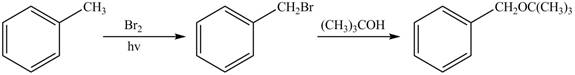
Figure 1
The first step of the synthesis is monobromination of methyl group of toluene in presence of light. The product obtained is treated with tertiary butanol to form desired ether.
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown in Figure 1.
(b)
Interpretation: The synthesis of given compound from toluene and any other organic or inorganic reagent is to be stated.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.66P
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown below:
Explanation of Solution
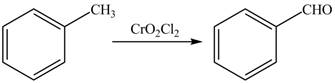
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown in Figure 1.
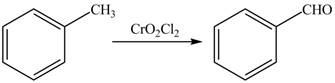
Figure 2
Toluene undergoes Etard’s reaction using inorganic reagent
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown in Figure 2.
(c)
Interpretation: The synthesis of given compound from toluene and any other organic or inorganic reagent is to be stated.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.66P
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown below:
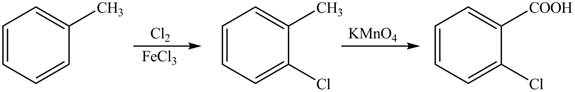
Explanation of Solution
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown below.
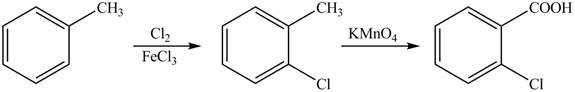
Figure 3
Toluene undergoes chlorination on ortho and para position, but for the synthesis ortho product is required. This ortho product is oxidized using potassium permanganate to form the desired product.
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown in Figure 3.
(d)
Interpretation: The synthesis of given compound from toluene and any other organic or inorganic reagent is to be stated.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.66P
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown below:
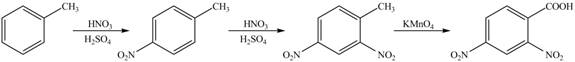
Explanation of Solution
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown below.
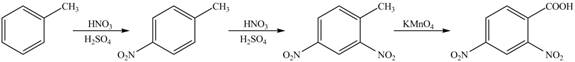
Figure 4
Toluene undergoes nitration on ortho and para position, but for the synthesis para product is required. This product again undergoes nitration on the meta position to nitro product. This product is oxidized using potassium permanganate to form the desired product.
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown in Figure 4.
(e)
Interpretation: The synthesis of given compound from toluene and any other organic or inorganic reagent is to be stated.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.66P
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown below:
Explanation of Solution
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown below.
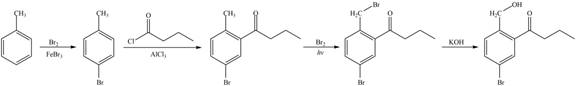
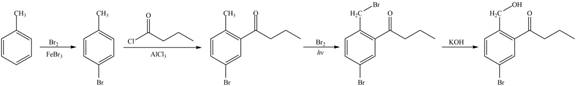
Figure 5
The first step of the synthesis is bromination of toluene which is followed by Friedel-Craft acylation. The product obtained by the Friedel-Craft acylation undergoes bromination at allylic carbon atom and on further reaction with strong base bromine group is replaced by hydroxyl group. This leads to the formation of desired product.
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown in Figure 5.
(f)
Interpretation: The synthesis of given compound from benzene and any other organic or inorganic reagent is to be stated.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.66P
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown below:
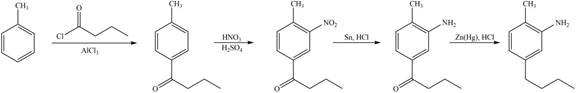
Explanation of Solution
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown below.
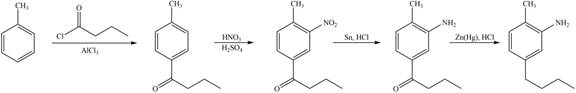
Figure 6
The first step of the synthesis is Friedel-Craft acylation of toluene which is followed by nitration reaction. The product obtained by nitration undergoes reduction in presence of
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown in Figure 6.
(g)
Interpretation: The synthesis of given compound from benzene and any other organic or inorganic reagent is to be stated.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.66P
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown below:
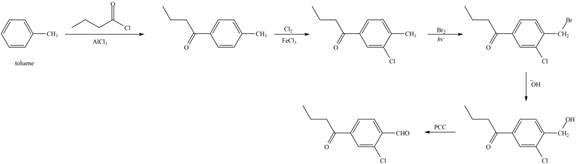
Explanation of Solution
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown below.
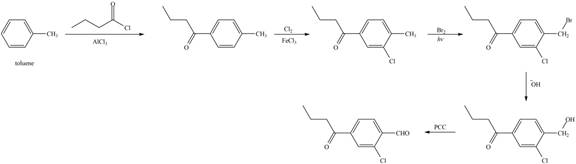
Figure 7
The first step of the synthesis is Friedel-Craft acylation of toluene which is followed by chlorination. The product obtained by chlorination undergoes bromination at allylic carbon atom and on further reaction with strong base bromine group is replaced by hydroxyl group. The
The synthesis of given compound from toluene by the use of organic and inorganic reagent is shown in Figure 7.
Want to see more full solutions like this?
Chapter 18 Solutions
Organic Chemistry-Package(Custom)
- 4b. monosubstituted benzene NO2arrow_forward18. In the IUPAC name for the following compound, the -Br group is located on carbon number CH;CHCH=CH, Br a. 1 b. 2 С. 3 d. 4arrow_forwardComplete and balance each hydrocarbon combustion reaction. a. CH;CH;CH,CH3 + O2 b. CH2=CHCH, + 02 c. CH=CCH,CH3 + O2arrow_forward
- 1. Which functional group is found in aldehydes? a. CHO b. -CH;OH c. -COOH d. RCOR' 2. Which functional group is found in ketones? a. -CHO CH:OH c. -COOH RCOR' 3. Which structural feature is common to aldehydes and ketones? a, an oxygen atom bonded to both a carbon atom and a hydrogen atom b. an oxygen atom bonded to two carbon atoms c. an oxygen atom double bonded to a carbon atom d. two oxygen atoms bonded to the same carbon atom 4. What is the IUPAC name for the following compound? a. I-pentaldehyde b. l-pentanal c. pentanal d. pentanealdehyde Cetoa of Fiarrow_forwardSynthesize each compound from benzene. a. SO₂H 19일 g b. 00 이 Harrow_forward6. What is the product? CH3 осно A. C. A and B A. A B. B C. C CH3 B. DBN CH₂arrow_forward
- Addition reactions of alkenes are characterized by: A. formation of a π bond B. addition of two groups across a double bond C. breaking of a π bond D. A and B E. B and Carrow_forward3. Ethanoic acid (vinegar) when diluted to low concentrations by water can be prepared from ethene by a) reduction with H2, followed by reaction with a strong oxidizer b) addition of HCL followed by reaction with H20 c) addition of H20 followed by reaction with a strong oxidizer d) addition of Br2 e) followed by reduction with H2arrow_forwardtaken in order to gor the product Please explain the mechanisms/stepsarrow_forward
- Show how to convert cyclohexanol to these compounds. a. Cyclohexene b. Cyclohexane c. Cyclohexanone d. Bromocyclohexanearrow_forwardwhat should be the preferred product in the following reaction? CH₂ Som OH CH3 CH3 § § a) b) a H₂SO A C CH₂ c) ? CH3 d)arrow_forward• Whał arc the IUPAC namar of the ff. Carboxylic acias? a. COOH COOH CH3 b. I f. .COOH CHJCH2 ÇHCHCOOH CH3 NO2 tON C. COOH ноос d. COOHarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





