
Interpretation:
The set of equations (or a small program) to evaluate the constant-volume heat capacity for a moleculeis to be stated. The graph of the result is to be plotted. The trend for the same is to be stated. The heat capacity versus temperature (say from
Concept introduction:
The heat capacity at constant volume for nonlinear polyatomic molecule is given by the formula,
Where,
•
•
•
•
Answer to Problem 18.59E
The set of equations (or a small program) to evaluate the constant-volume heat capacity for a molecule are,
•
•
•
•
•
•
•
The plot between
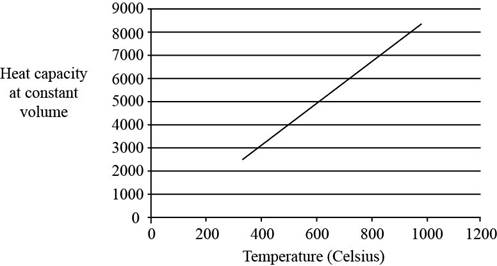
The plot between
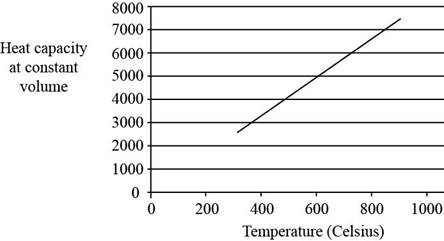
Explanation of Solution
The heat capacity at constant volume for nonlinear polyatomic molecule is given by the formula,
The set of equations(or a small program) to evaluate the constant-volume heat capacity for a molecule are shown below.
•
•
•
•
•
•
•
The vibrational temperatures for
Substitute the value of vibrational temperatures for
The value of
The plot between
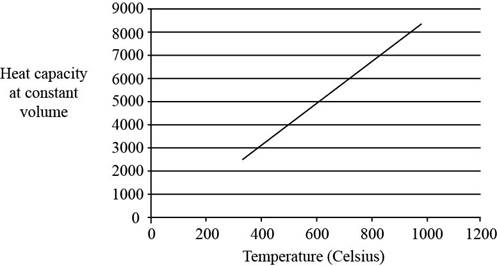
Figure 1
The three vibrational temperatures for
Substitute the value of vibrational temperatures for
The value of
The plot between
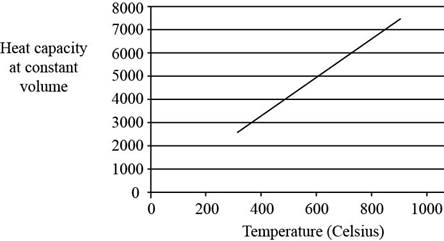
Figure 2
The set of equations (or a small program) to evaluate the constant-volume heat capacity for a molecule are,
•
•
•
•
•
•
•
The plot between
Want to see more full solutions like this?
Chapter 18 Solutions
Student Solutions Manual for Ball's Physical Chemistry, 2nd
- A 1.00 mol sample of H2 is carefully warmed from 22 K to 40 K at constant volume. a What is the expected heat capacity of the hydrogen? b What is q for the process?arrow_forwardDefine the following terms: potential energy, kinetic energy, path-dependent function, state function, system, surroundings.arrow_forwardWhat are the numerical values of the heat capacities c-v and c-p of a monatomic ideal gas,in units of cal/mol.K and L.atm/mol.K?arrow_forward
- Assume that 1.20 g of benzoicacid, C6H5COOH, is burned in a porcelain dish exposed to the air.If 31, 723 J of energy is givenoff andthe surrounding temperature is 24.6C, calculate q, w, H,and U. Compare your answers to those from the previousproblem.arrow_forward• define state functions and explain their importance.arrow_forward
 Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning, Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





