
Interpretation: The structures of all the constitutionally isomeric ethers of molecular formula
Concept introduction:
The compounds that have the identical molecular formula but have different connectivity of atoms are termed as constitutional isomers. The constitutional isomers are also known as structural isomers.
The names of the different compounds of ether can be determined by using the IUPAC convention for naming compounds.
Answer to Problem 21P
The structures of all the constitutionally isomeric ethers of molecular formula
Explanation of Solution
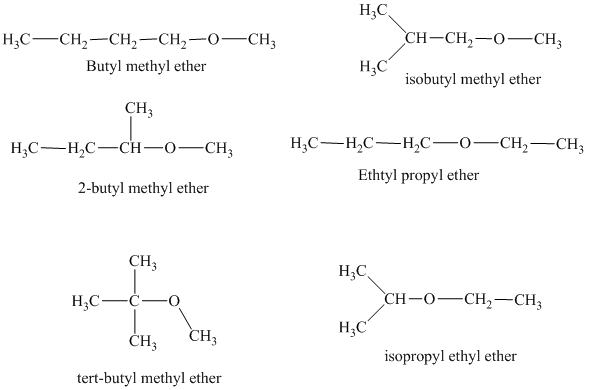
The different constitutionally isomeric ethers that can be written for the given molecular formula, that is,
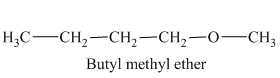
In the above isomeric form, a butyl and a methyl group are attached with the oxygen atom. Therefore, the acceptable name for this isomer is Butyl methyl ether.
Another constitutional isomer for ether of molecular formula
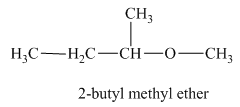
The oxygen atom is attached to the second carbon of the butyl group and a methyl group. Therefore, the acceptable name for this isomer is 2-butyl methyl ether.
Another constitutional isomer for ether of molecular formula
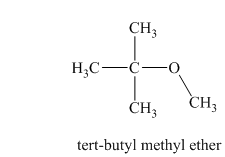
In this isomeric ether, the oxygen atom is attached to a methyl group and a tertiary butyl group. Therefore, the acceptable name for this isomer is tert-butyl methyl ether.
Another constitutional isomer for ether of molecular formula
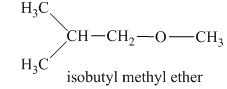
In this isomeric form of ether, the oxygen atom is attached to a methyl group and an isobutyl group. Therefore, the acceptable name for this isomer is isobutyl methyl ether.
Another constitutional isomer for ether of the given molecular formula is shown below.
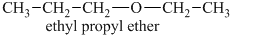
The oxygen atom in the above isomeric form of ether is attached to an ethyl and a propyl group. Therefore, the acceptable name for this isomer is ethyl propyl ether.
Another constitutional isomer for ether of molecular formula
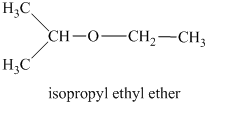
In this isomeric ether, the oxygen atom is attached to an isopropyl and an ethyl group. Therefore, the acceptable name for this isomer is isopropyl ethyl ether.
There are total six constitutional isomers for the ether of molecular formula
Want to see more full solutions like this?
Chapter 17 Solutions
ORGANIC CHEMISTRY (LOOSELEAF)-PACKAGE
- Write structural formulas for all ketones with the molecular formula C6H12O and give each its IUPAC name. Which of these ketones are chiral?arrow_forward(A) Which of the following ethers is the least soluble in water? (B) Which produces ethanol and methyl iodide after treatment with dilute HI?arrow_forwardThree constitutional isomers of molecular formula C 5H 8O can be converted to 1-pentanol (CH 3CH 2CH 2CH 2CH 2OH) on treatment with two equivalents of H 2 in the presence of a Pd catalyst. Draw the structures of the three possible compounds, all of which contain a carbonyl grouparrow_forward
- Predict which member of each group is most soluble in water, and explain the reasons for your predictions.(a) butan-1-ol, pentan-1-ol, or propan-2-ol(b) chlorocyclohexane, cyclohexanol, or cyclohexane-1,2-diol(c) phenol, cyclohexanol, or 4-methylcyclohexanolarrow_forwardDescribe each highlighted bond in terms of the overlap of atomic orbitals. and Draw the structures of ALL of the aldehydes with the molecular formula C5H10O that contain a 5-carbon chain.arrow_forwardAssume that you have samples of the following two compounds, both with formula C7H8O. Both compounds dissolve in ether, but only one of the two dissolves in aqueous NaOH. How could you use this information to distinguish between them?arrow_forward
- Draw and name all constitutionally isomeric alcohols with the molecular formula C4H10Oarrow_forward3. From what Grignard reagent and carbonyl compound are the following alcohols are prepared? (a) (b) CH₂OH H* CH₂O -CH3 OH -CH3arrow_forwardCompound X (molecular formula C10H12O) was treated with NH2NH2,−OH to yield compound Y (molecular formula C10H14). Based on the 1H NMR spectra of X and Y given below, what are the structures of X and Y?arrow_forward
- Dimethyl disulfide, CH,S–SCH3, found in the vaginal secretions of female hamsters, acts as a sexual attractant for the male hamster. Write an equation for its synthesis from methanethiol.arrow_forwardCyclopropenones are described as having aromatic character. How would you account for this, given that the ring contains three π-electrons?arrow_forwardCompound A exhibits a peak in its 1H NMR spectrum at 7.6 ppm, indicating that it is aromatic. (a) How are the carbon atoms of the triple bonds hybridized? (b) In what type of orbitals are the π electrons of the triple bonds contained? (c) How many π electrons are delocalized around the ring in A?arrow_forward
