
Concept explainers
(a)
Interpretation:
Organic product that is formed in the given reaction has to be classified as primary amine, secondary amine, tertiary amine or quaternary salt.
Concept Introduction:
Alkylation reaction is a reaction in which the transfer of alkyl group from one molecule to another molecule takes place. While considering
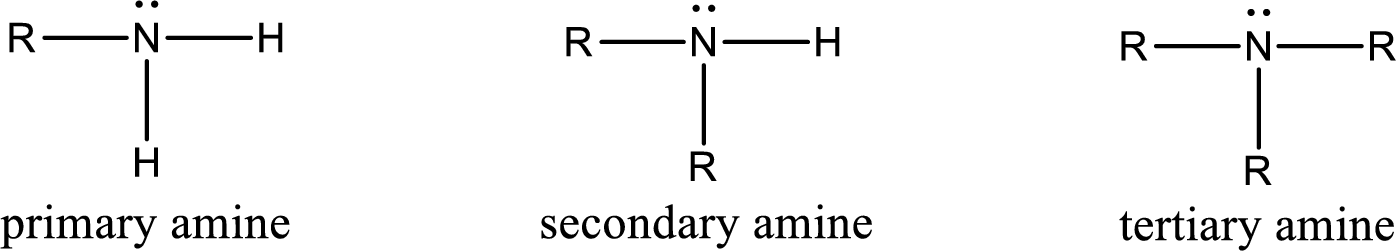
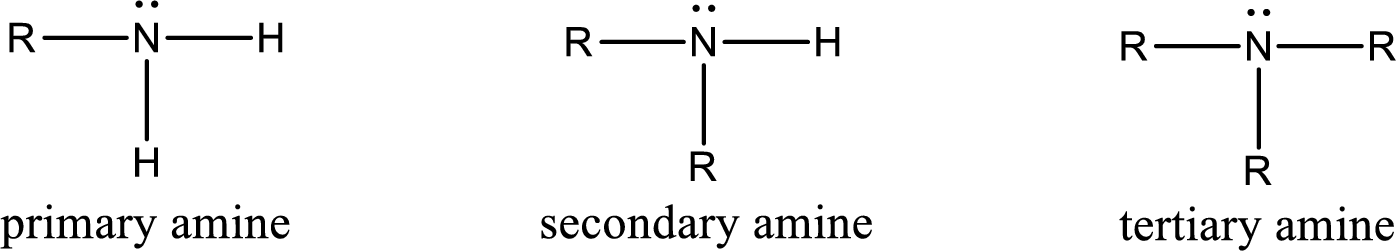
Amine is an organic derivative. If in ammonia one or more alkyl, cycloalkyl, or aryl groups are substituted instead of hydrogen atom then it is known as amine. Depending on the number of substitution the amines are classified as primary, secondary or tertiary amine. Primary amine is the one in which only one hydrogen atom in ammonia is replaced by a hydrocarbon group. Secondary amine is the one in which only two hydrogen atoms in ammonia is replaced by a hydrocarbon group. Tertiary amine is the one in which all three hydrogen atoms in ammonia is replaced by a hydrocarbon group. The generalized structural formula for all the amines is,
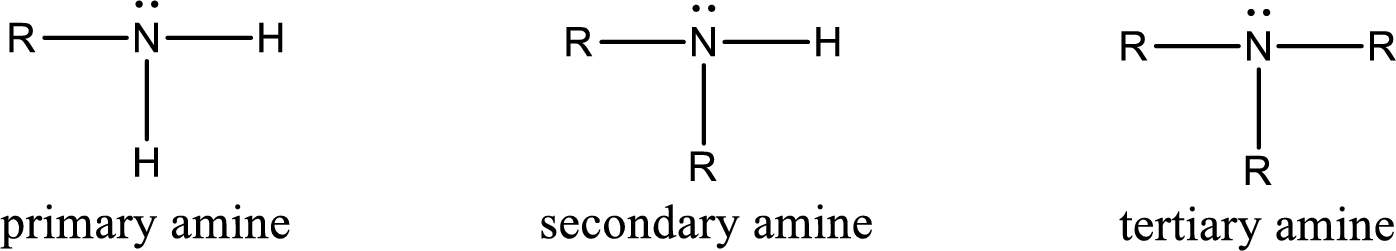
Quaternary ammonium salt is the one that has four carbon atoms attached to the nitrogen atom. This is formed by the reaction of tertiary amine with alkyl halide in presence of a strong base.
(a)
Answer to Problem 17.63EP
Organic product is a primary amine.
Explanation of Solution
Given reaction is,

The reactants given in the above reaction are ammonia, propyl chloride. Sodium hydroxide is a reagent that is used for basic condition in this case. As the reaction between ammonia and propyl chloride gives propylamine as the product, this is an alkylation reaction. The complete reaction can be given as,
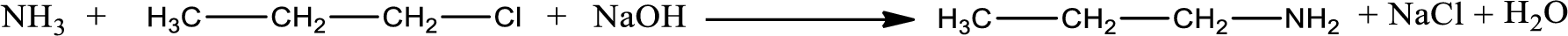
The organic product formed is propylamine. Considering the nitrogen atom in propylamine, it is bonded to one carbon atom and two hydrogen atoms. Hence, it is a primary amine.
The organic product formed in the given reaction is classified.
(b)
Interpretation:
Organic product that is formed in the given reaction has to be classified as primary amine, secondary amine, tertiary amine or quaternary salt.
Concept Introduction:
Alkylation reaction is a reaction in which the transfer of alkyl group from one molecule to another molecule takes place. While considering amines, the alkylating agent that is used is alkyl halides. Alkylation is done under basic conditions. The general equations for amines alkylation process is,
Amine is an organic derivative. If in ammonia one or more alkyl, cycloalkyl, or aryl groups are substituted instead of hydrogen atom then it is known as amine. Depending on the number of substitution the amines are classified as primary, secondary or tertiary amine. Primary amine is the one in which only one hydrogen atom in ammonia is replaced by a hydrocarbon group. Secondary amine is the one in which only two hydrogen atoms in ammonia is replaced by a hydrocarbon group. Tertiary amine is the one in which all three hydrogen atoms in ammonia is replaced by a hydrocarbon group. The generalized structural formula for all the amines is,
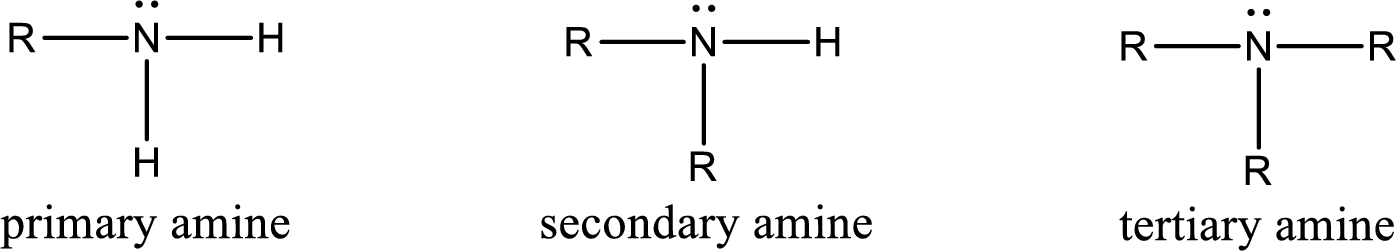
Quaternary ammonium salt is the one that has four carbon atoms attached to the nitrogen atom. This is formed by the reaction of tertiary amine with alkyl halide in presence of a strong base.
(b)
Answer to Problem 17.63EP
Organic product is a tertiary amine.
Explanation of Solution
Given reaction is,
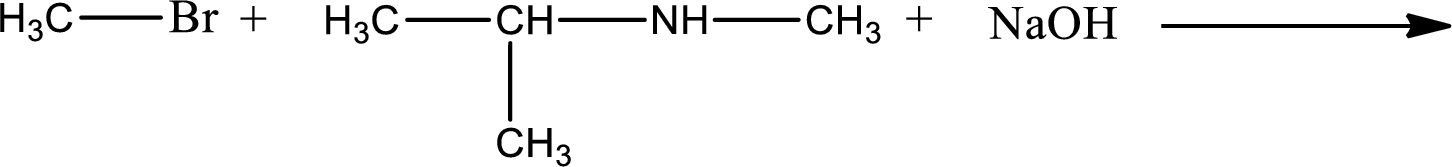
The reactants given in the above reaction are isopropylmethylamine, methyl bromide. Sodium hydroxide is a reagent that is used for basic condition in this case. As the reaction between isopropylmethylamine and methyl bromide gives isopropyldimethylamine as the product, this is an alkylation reaction. The complete reaction can be given as,
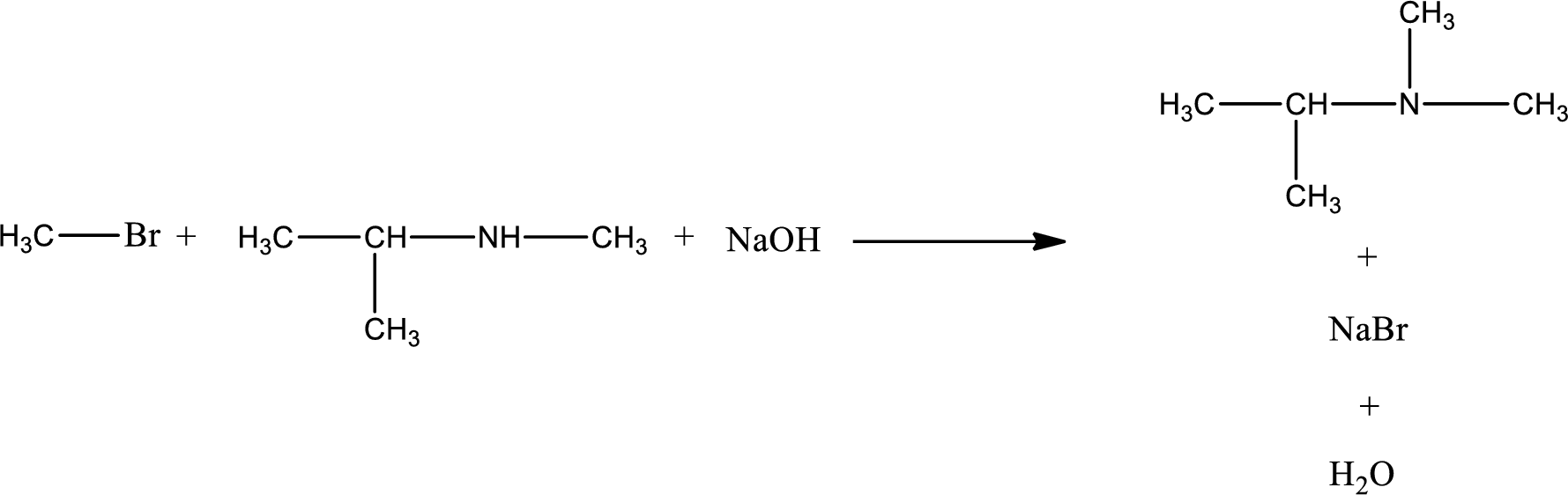
The organic product that is formed has a nitrogen atom that is bonded to three carbon atoms. Hence, it is a tertiary amine.
The organic product formed in the given reaction is classified.
(c)
Interpretation:
Organic product that is formed in the given reaction has to be classified as primary amine, secondary amine, tertiary amine or quaternary salt.
Concept Introduction:
Alkylation reaction is a reaction in which the transfer of alkyl group from one molecule to another molecule takes place. While considering amines, the alkylating agent that is used is alkyl halides. Alkylation is done under basic conditions. The general equations for amines alkylation process is,
Amine is an organic derivative. If in ammonia one or more alkyl, cycloalkyl, or aryl groups are substituted instead of hydrogen atom then it is known as amine. Depending on the number of substitution the amines are classified as primary, secondary or tertiary amine. Primary amine is the one in which only one hydrogen atom in ammonia is replaced by a hydrocarbon group. Secondary amine is the one in which only two hydrogen atoms in ammonia is replaced by a hydrocarbon group. Tertiary amine is the one in which all three hydrogen atoms in ammonia is replaced by a hydrocarbon group. The generalized structural formula for all the amines is,
Quaternary ammonium salt is the one that has four carbon atoms attached to the nitrogen atom. This is formed by the reaction of tertiary amine with alkyl halide in presence of a strong base.
(c)
Answer to Problem 17.63EP
Organic product is a secondary amine.
Explanation of Solution
Given reaction is,
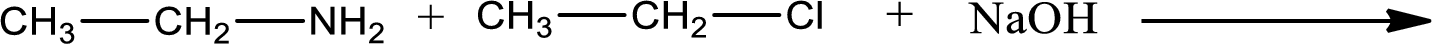
The reactants given in the above reaction are ethylamine, ethyl chloride. Sodium hydroxide is a reagent that is used for basic condition in this case. As the reaction between ethylamine and ethyl chloride gives diethylamine as the product, this is an alkylation reaction. The complete reaction can be given as,
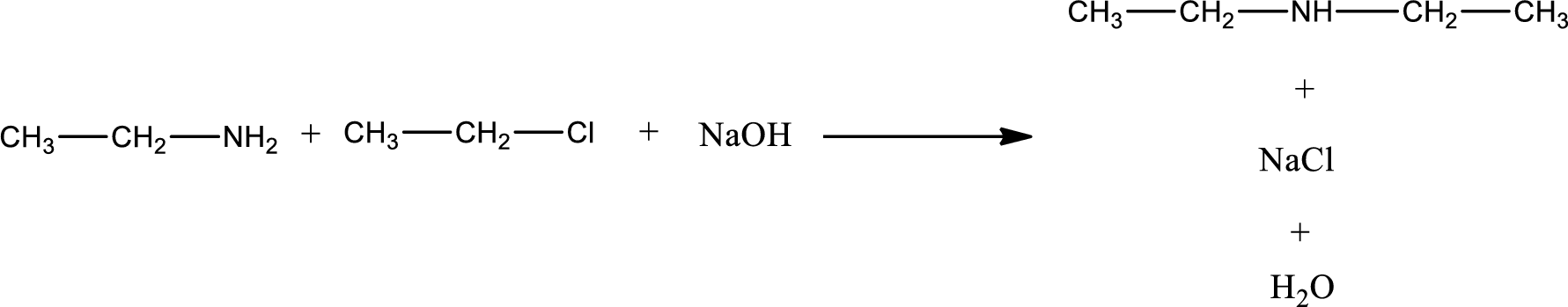
The organic product obtained has a nitrogen atom that is bonded to two carbon atoms and one hydrogen atom. Hence, it is a secondary amine.
The organic product formed in the given reaction is classified.
(d)
Interpretation:
Organic product that is formed in the given reaction has to be classified as primary amine, secondary amine, tertiary amine or quaternary salt.
Concept Introduction:
Alkylation reaction is a reaction in which the transfer of alkyl group from one molecule to another molecule takes place. While considering amines, the alkylating agent that is used is alkyl halides. Alkylation is done under basic conditions. The general equations for amines alkylation process is,
Amine is an organic derivative. If in ammonia one or more alkyl, cycloalkyl, or aryl groups are substituted instead of hydrogen atom then it is known as amine. Depending on the number of substitution the amines are classified as primary, secondary or tertiary amine. Primary amine is the one in which only one hydrogen atom in ammonia is replaced by a hydrocarbon group. Secondary amine is the one in which only two hydrogen atoms in ammonia is replaced by a hydrocarbon group. Tertiary amine is the one in which all three hydrogen atoms in ammonia is replaced by a hydrocarbon group. The generalized structural formula for all the amines is,
Quaternary ammonium salt is the one that has four carbon atoms attached to the nitrogen atom. This is formed by the reaction of tertiary amine with alkyl halide in presence of a strong base.
(d)
Answer to Problem 17.63EP
Organic product is a primary amine.
Explanation of Solution
Given reaction is,
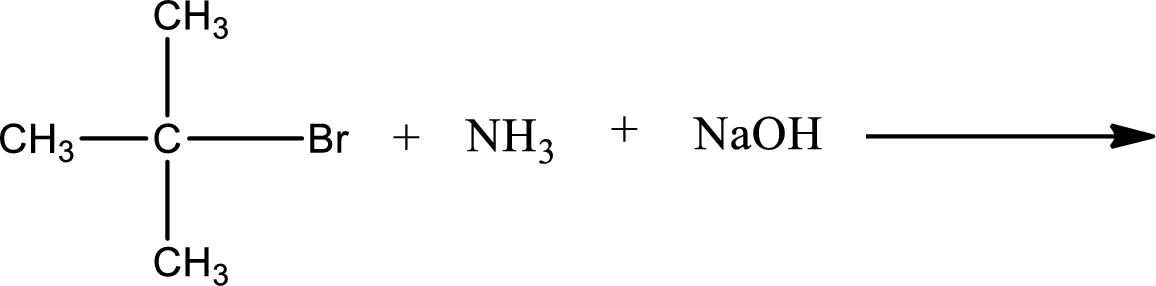
The reactants given in the above reaction are ammonia, tert-butyl bromide. Sodium hydroxide is a reagent that is used for basic condition in this case. As the reaction between ammonia and tert-butyl bromide gives tert-butylamine as the product, this is an alkylation reaction. The complete reaction can be shown as,
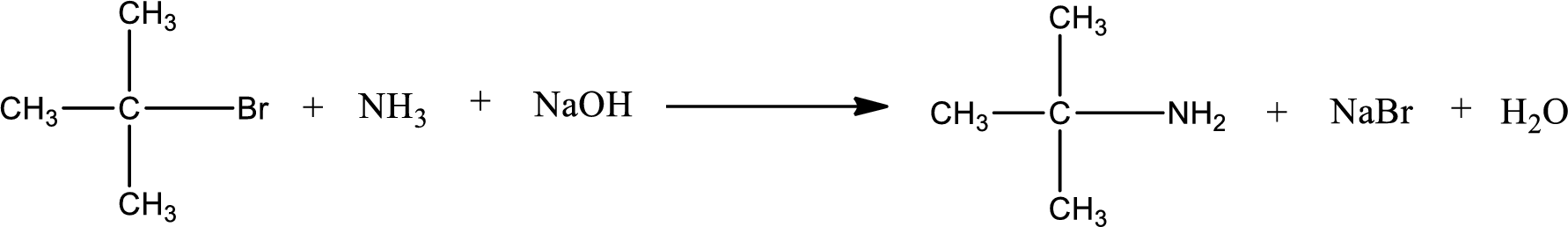
The organic product formed has a nitrogen atom that is bonded to one carbon atom and two hydrogen atoms. Hence, it is a primary amine.
The organic product formed in the given reaction is classified.
Want to see more full solutions like this?
Chapter 17 Solutions
EBK GENERAL, ORGANIC, AND BIOLOGICAL CH
- Consider the intermolecular forces present in a pure sample of each of the following compounds: CH₃CH₂OH and CH₃COCH₃. Identify the intermolecular forces that these compounds have in common.arrow_forwardFumaric acid is a metabolic intermediate that has the systematic name trans-2-butenedioic acid. Draw its structure.arrow_forwardIn the preparation of aspirin, You can do the functional group test to ensure the completion of the reaction. (a) What is the name of reagent used? (b) What is your observation if any unreacted starting material is present? (c) What is the name of the functional group responsible for this reaction?arrow_forward
- Give 5 examples of molybdenum complexes. State their physical and chemical properties and their importance or applications. Additional question, what are the oxidation numbers of molybdenumarrow_forwardPropose a scheme to separate and isolate a mixture containing 4-aminobenzoate, 1,2,4,5-tetrachlorobenzene, and napthalene. Isolate two of the three solids by extraction. (please explain what solvents to use and the process step by step)arrow_forward(a) Label all the O atoms that are part of a glycoside in rebaudioside A. Rebaudioside A, marketed under the trade name Truvia, is a sweet glycoside obtained from the stevia plant, which has been used for centuries in Paraguay to sweeten foods. (b) The alcohol or phenol formed from the hydrolysis of a glycoside is called an aglycon. What aglycon and monosaccharides are formed by the hydrolysis of rebaudioside A?arrow_forward
- Potentiometric titration curve is given below, which is obtained during the potentiometric titration between strong base KOH (0.2 M) with strong acid HI, label the point in the curve from the following options. If more than one points are present than write as x, y(means separate by using comma) a)The point where pH is because of excess OH - ions. b) The point where pH is only because of HI in water. c)The point where [HI]= [I] in water. d)The point where pH=pka e) The point where all HI is neutralized. f) The point where pH corresponds to solution of [I- ] in water. 14 13 12 11 10 9 pH 6. TITIT TITarrow_forwardWhich of the following is the cyclic form of an L saccharide? 1) II) CH2OH OH H CH2OH H H H ОН OH H ОН ОН OH H ОН II) IV) H OH ОН CH2OH CH2OH CH2OH H H. H ОН H H H OH ОН ОН OHarrow_forwardName the following compound and indicate whether or not is a reducing sugar:arrow_forward
- Draw the structures of the following acids:(a) 2-Ethyl-3-hydroxyhexanoic acid (b) m-Nitrobenzoic acidarrow_forwardThe linear tripeptides are formed from the following three amino acids as the starting materials in the condensation reaction. Select the correct statement: (a) There are 27 possible tripeptides and among them, 13 tripeptides are optically active. (b) There are 26 possible tripeptides and all are optically active. (c) There are 27 possible tripeptides and among them, 26 tripeptides are optically active. (d) There are 26 possible tripeptides and among them, 15 tripeptides are optically active.arrow_forwardDrawn are four isomeric dimethylcyclopropanes. How are the compounds in each pair related (enantiomers, diastereomers, constitutional isomers): A and B; A and C; B and C; C and D?arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





