
Concept explainers
Propose a structure consistent with each set of data.
a. Compound A:
b. Compound B:
(a)
Interpretation: A structure consistent with the given set of data is to be predicted.
Concept introduction: Spectroscopy method is used to identify the structure of the molecule. It is based on the interactions between matter and electromagnetic radiations. Proton NMR spectroscopy identifies the number of hydrogen atoms present in a molecule and the nature of the functional group. The value of chemical peaks depends upon the chemical environment around the hydrogen atom.
Answer to Problem 17.56P
A structure consistent with the given set of data is shown below.
Explanation of Solution
The given sets of
Information from
The
Information from
The observed chemical shift value at
The observed chemical shift value at
The observed chemical shift value at
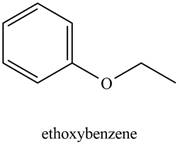
The possible structure of the Compound A, based on the above analysis is,
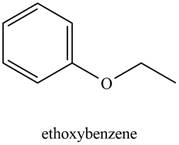
Figure 1
A structure consistent with the given set of data is shown in Figure 1.
(b)
Interpretation: A structure consistent with the given set of data is to be predicted.
Concept introduction: Spectroscopy method is used to identify the structure of the molecule. It is based on the interactions between matter and electromagnetic radiations. Proton NMR spectroscopy identifies the number of hydrogen atoms present in a molecule and the nature of the functional group. The value of chemical peaks depends upon the chemical environment around the hydrogen atom.
Answer to Problem 17.56P
A structure consistent with the given set of data is shown below.
Explanation of Solution
The given sets of
Information from
The
Information from
The observed chemical shift value at
The observed chemical shift value at
The observed chemical shift values at
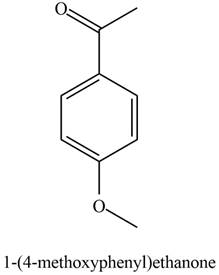
The possible structure of the Compound B, based on the above analysis is,
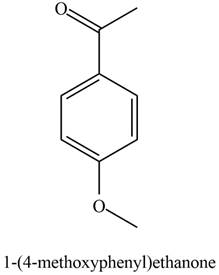
Figure 2
A structure consistent with the given set of data is shown in Figure 2.
Want to see more full solutions like this?
Chapter 17 Solutions
Organic Chemistry
- Compound A, MW 86, shows an IR absorption at 1730 cm-1 and a very simple 1H NMR spectrum with peaks at 9.7 (1 H, singlet) and 1.2 (9 H, singlet). Propose a structure for A.arrow_forwardCompound I (C11H14O2) is insoluble in water, aqueous acid, and aqueous NaHCO3, but dissolves readily in 10% Na2CO3 and 10% NaOH. When these alkaline solutions are acidified with 10% HCl, compound I is recovered unchanged. Given this information and its 1H-NMR spectrum, deduce the structure of compound I.arrow_forwardCompound A, C8H10O, has the IR and 1H NMR spectra shown. Propose a structure consistent with the observed spectra, and label each peak in the NMR spectrum. Note that the absorption at 5.5 î disappears when D2O is added.arrow_forward
- Propose a structure for D (molecular formula C9H9ClO2) consistent with the given spectroscopic data. 13C NMR signals at 30, 36, 128, 130, 133, 139, and 179 ppmarrow_forwardAn unknown compound has a molecular formula of C,H,O. Its IR spectrum shows prominent absorptions at 2980, 2960, and 1718 cm . It exhibits the following signals in its H NMR spectrum (ppm): 1.06 (triplet, 3H), 2.12 (singlet, 3H), 2.45 (quartet, 2H); and the following signals in its ¹3C NMR spectrum ( ppm): 7.6, 29.5, 36.8, 208.8. Draw the structure of the unknown compound. Click and drag to start drawing a structure. 0 X 0:0arrow_forwardPropose a structure for a C,H150,N compound that is unstable in aqueous acid and has the given NMR spectra. 'H NMR: 8 2.30 (6H, s); 8 2.45 (2H, d, J = 6 Hz); 8 3.27 (6H, s); 8 4.50 (1H, t, J = 6 Hz) 13C NMR: 8 46.3, 8 53.2, 8 68.8, 8 102.4 Draw C,H1502N. Select Draw Rings More C N APR étv N Aa MacBook Airarrow_forward
- Compound X (molecular formula C10H12O) was treated with NH2NH2, -OH to yield compound Y (molecular formula C10H14). Based on the 1H NMR spectra of X and Y given below, what are the structures of X and Y?arrow_forwardA compound with molecular formula C5H10Br2 displays the following 13C NMR along with the DEPT-90 and DEPT-135 13CNMR spectra. What is the structure for this compound?arrow_forwardTreatment of compound E (molecular formula C4H8O2) with excess CH3CH2MgBr yields compound F (molecular formula C6H14O) after protonation with H2O. E shows a strong absorption in its IR spectrum at 1743 cm-1. F shows a strong IR absorption at 3600–3200 cm-1. The 1H NMR spectral data of E and F are given. What are the structures of E and F?Compound E signals at 1.2 (triplet, 3 H), 2.0 (singlet, 3 H), and 4.1 (quartet, 2 H) ppmCompound F signals at 0.9 (triplet, 6 H), 1.1 (singlet, 3 H), 1.5 (quartet, 4 H), and 1.55 (singlet, 1 H) ppmarrow_forward
- An unknown compound has a molecular formula of C4H6O2. Its IR spectrum shows absorptions at 3095, 1762, 1254, and 1118 cm -1. It exhibits the following signals in its 1H NMR spectrum (ppm): 2.12 (singlet,3H), 4.55 (doublets of doublets, 1H), 4.85 (doublet of doublets, 1H), 7.25 (doublets of doublets, 1H); and the following signals in its 13C NMR spectrum (ppm): 20.8, 100.4, 141.2, 168.0. Draw the structure of the unknown compoundarrow_forwardA compound of molecular formula C4H8O2 shows no IR peaks at 3600–3200 or 1700 cm-1. It exhibits one singlet in its 1H NMR spectrum at 3.69 ppm, and one line in its 13C NMR spectrum at 67 ppm. What is the structure of this unknown?arrow_forwardPropose a structure for D (molecular formula C9H9ClO2) consistent withthe given spectroscopic data. 13C NMR signals at 30, 36, 128, 130, 133,139, and 179 ppmarrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

