
(a)
Interpretation: The name of
Concept introduction:
Esters encompass a large family of organic compound with broad application in various fields of science and technology.
In Fischer esterification, a carboxylic acid is reacted with an alcohol in the presence of an acid catalyst, such as concentrated sulfuroc acid.
Esters can be simply represented as,
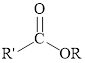
Fischer esterification can be represented as follows,
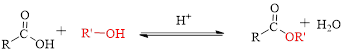
The splitting of an ester into the component acid and alcohol is known as ester hydrolysis.
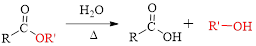
(b)
Interpretation: The name of carboxylic acid and alcohol has to be determined, from which the given eseter is derived.
Concept introduction:
Esters encompass a large family of organic compound with broad application in various fields of science and technology.
In Fischer esterification, a carboxylic acid is reacted with an alcohol in the presence of an acid catalyst, such as concentrated sulfuroc acid.
Esters can be simply represented as,

Fischer esterification can be represented as follows,
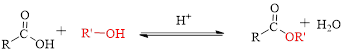
The splitting of an ester into the component acid and alcohol is known as ester hydrolysis.
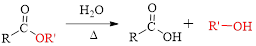
(c)
Interpretation: The name of carboxylic acid and alcohol has to be determined, from which the given eseter is derived.
Concept introduction:
Esters encompass a large family of organic compound with broad application in various fields of science and technology.
In Fischer esterification, a carboxylic acid is reacted with an alcohol in the presence of an acid catalyst, such as concentrated sulfuroc acid.
Esters can be simply represented as,

Fischer esterification can be represented as follows,
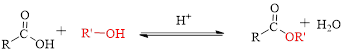
The splitting of an ester into the component acid and alcohol is known as ester hydrolysis.
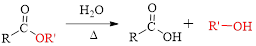
(d)
Interpretation: The name of carboxylic acid and alcohol has to be determined, from which the given eseter is derived.
Concept introduction:
Esters encompass a large family of organic compound with broad application in various fields of science and technology.
In Fischer esterification, a carboxylic acid is reacted with an alcohol in the presence of an acid catalyst, such as concentrated sulfuroc acid.
Esters can be simply represented as,

Fischer esterification can be represented as follows,
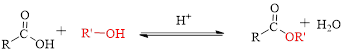
The splitting of an ester into the component acid and alcohol is known as ester hydrolysis.
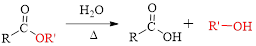
Trending nowThis is a popular solution!

Chapter 17 Solutions
ORGANIC CHEMISTRY-OWL V2 ACCESS
- One of the compounds that gives orange oil its unique odor is an ester formed when acetic acid reacts with octan-1-ol. Draw the structure of this ester and name it.arrow_forwardDescribe the Properties of Carboxylic Acids and Its Derivatives?arrow_forwardExplain the characteristic reaction of aldehydes and ketones ?arrow_forward

 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





