
Concept explainers
(a)
Interpretation:
The organic product formed in the given reaction has to be indicated as primary amide, secondary amide, tertiary amide, or not an amide.
Concept Introduction:
Amides are synthesized using amidification reaction. This involves a reaction between
(a)
Answer to Problem 17.131EP
Organic product obtained is a primary amide.
Explanation of Solution
Given reaction is,
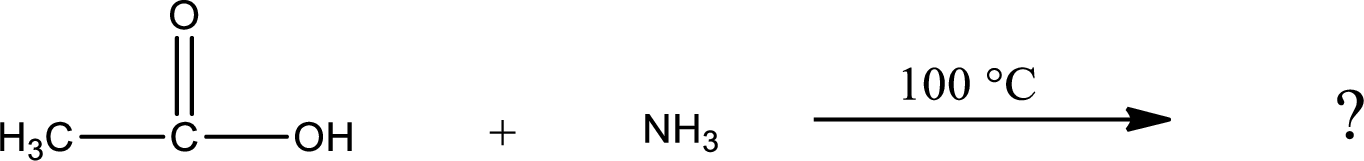
In amidification process,
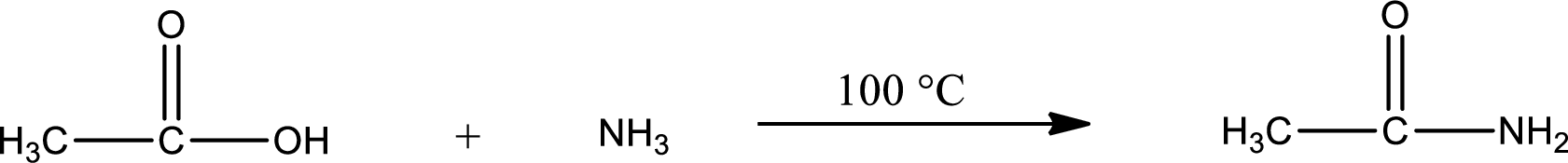
The product obtained is indicated.
(b)
Interpretation:
The organic product formed in the given reaction has to be indicated as primary amide, secondary amide, tertiary amide, or not an amide.
Concept Introduction:
Amides are synthesized using amidification reaction. This involves a reaction between amine and carboxylic acid. In this reaction, the
(b)
Answer to Problem 17.131EP
Organic product obtained is a secondary amide.
Explanation of Solution
Given reaction is,
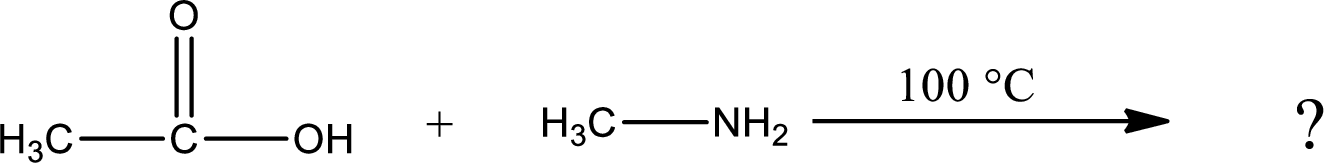
In amidification process,
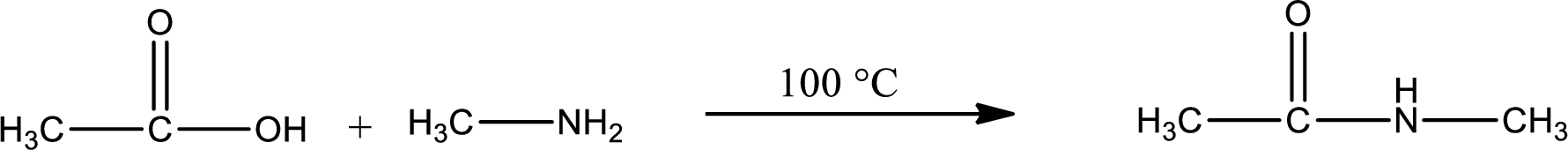
The product obtained is indicated.
(c)
Interpretation:
The organic product formed in the given reaction has to be indicated as primary amide, secondary amide, tertiary amide, or not an amide.
Concept Introduction:
Amides are synthesized using amidification reaction. This involves a reaction between amine and carboxylic acid. In this reaction, the
(c)
Answer to Problem 17.131EP
Organic product obtained is a tertiary amide.
Explanation of Solution
Given reaction is,
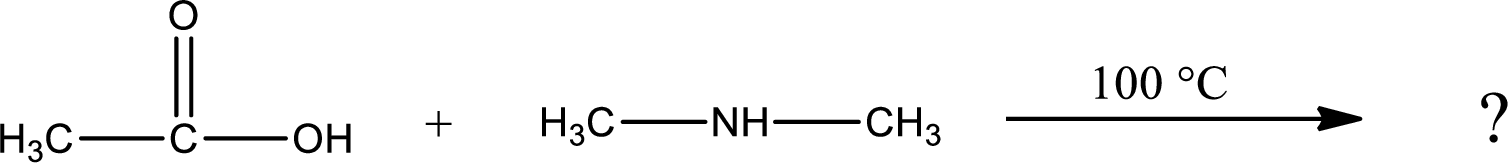
In amidification process,
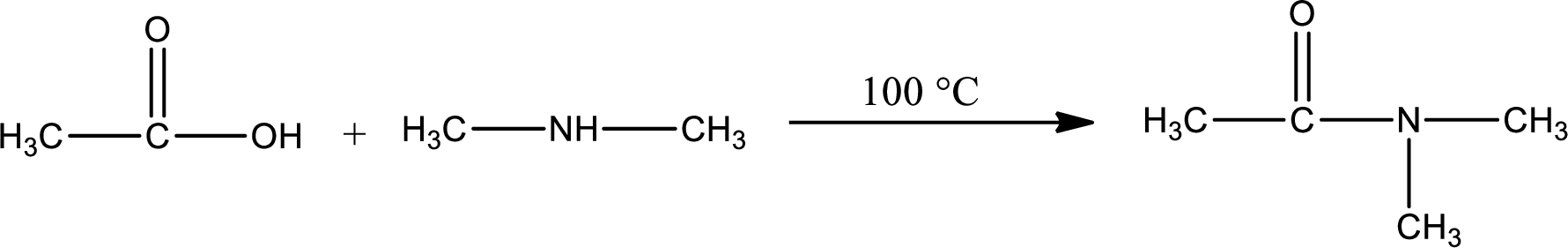
The product obtained is indicated.
(d)
Interpretation:
The organic product formed in the given reaction has to be indicated as primary amide, secondary amide, tertiary amide, or not an amide.
Concept Introduction:
Amides are synthesized using amidification reaction. This involves a reaction between amine and carboxylic acid. In this reaction, the
(d)
Answer to Problem 17.131EP
Organic product obtained is a tertiary amide.
Explanation of Solution
Given reaction is,

In amidification process,
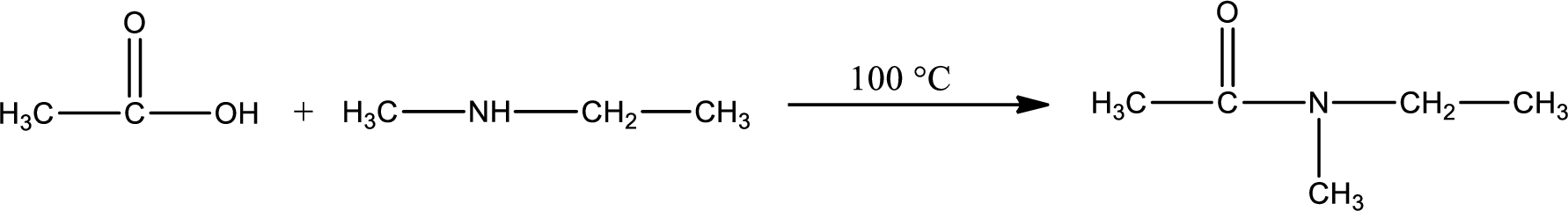
The product obtained is indicated.
Want to see more full solutions like this?
Chapter 17 Solutions
Bundle: General, Organic, and Biological Chemistry, 7th + OWLv2 Quick Prep for General Chemistry, 4 terms (24 months) Printed Access Card
- Draw a Fischer projection formula for the enantiomer of each of the following monosaccharides. (a to d)arrow_forwardFor A, B, C, D, E, F, identify the circled functional groups and linkages in the compound in the picture.arrow_forwardAre the following molecules chiral or achiral? If they are chiral, identify the chiral carbon atom(s).(a) Pentan-3-ol (b) 2-Bromobutane(c) 2-Methylcyclohexanolarrow_forward
- IDENTIFY THE FUNCTIONAL GROUP PRESENT IN THESE COMPOUNDS. A = ? B = ? C = ?arrow_forwardFor the first part, draw a Fischer projection formula for the enantiomer of each of the following monosaccharides. For the second part, identify whether the images are in D- or L-configuration. Write your answers first on a piece of bond paper.arrow_forwardFollowing are Fischer projections for a groupof five-carbon sugars, all of which are aldopentoses. Identify thepairs that are enantiomers and the pairs that are epimers. (Thesugars shown here are not all of the possible five-carbon sugars.)arrow_forward
- Dimethyl ether has the same molecular formula as ethanol (Problem 4.57) but very different properties. Propose a structure for dimethyl ether in which the oxygen is bonded to two carbonsarrow_forwardFollowing are Fischer projections for a group of five-carbon sugars, all of which are aldopentoses. Identify the pairs that are enantiomers and the pairs that are epimers. (The sugars shown here are not all of the possible five-carbon sugars.) 1 СНО 2 СНО СНО Н-С—ОН Н-С—ОН Н—С—ОН Н-С—ОН НО —С—Н Н-С—ОН Н- С—ОН НО—С— Н НО—С—Н CH̟OH CH̟OH ČH̟OH в iоchemistry |61 STATE ATION Republic of the Philippines Romblon State University Romblan, Philippines 4 СНО 5 СНО 6 СНО НО—С—Н Н-С—ОН НО -С—Н Н—С—ОН НО -С—Н НО -С—Н H-C–OH Н-С—ОН НО—С—Н CH,OH ČH,OH CH,OHarrow_forwardWhich of the following define the stereochemistry of alanine (as per the structure shown)? Note: Functional groups arranged horizontally are facing towards the front, and the functional groups arranged vertically are facing towards the back. СООН + H₂N to OS- Od- CH, OR-arrow_forward
- Draw the structures of the following compounds. (Includes both new and old names.) 3-cyclopentylhexan-3-olarrow_forwardWhat is unusual about glutathione’s structure? (If you cannot answer this question, draw the structure you would expect for the tripeptide and compare your structure with the actual structure.)arrow_forwardThe reaction of methoxy benzene with hydrogen iodide will yield a phenol and an alkyl halide. Which of following choices is the correct combination of the products?arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





