
Concept explainers
a.
Interpretation:
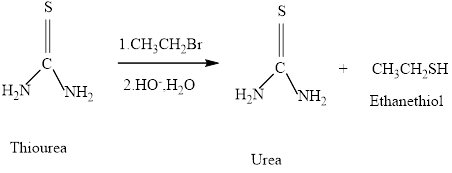
Thiols can be prepared from the reaction of thiourea with an
Concept introduction:
Organosulfur compound that contains a carbon –bonded sulfhydry group are called thiols. It has strong odour. The –SH froup is called a mercapto group. Named by adding the suffix –thiol to the
For example, the structure of 2-methyl propane 2-thiol.
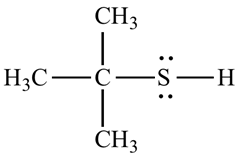
b.
Interpretation:
Thiols can be prepared from the reaction of thiourea with an alkyl halide , followed by hydroxide –io-promoted hydrolysis. What will be formed if the alkyl halide employed is pentyl bromide.
Concept introduction:
Organosulfur compound that contains a carbon –bonded sulfhydry group are called thiols. It has strong odour. The –SH froup is called a mercapto group. Named by adding the suffix –thiol to the alkane name. They are commonly prepared by an
For example, the structure of 2-methyl propane 2-thiol.
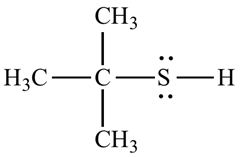
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Organic Chemistry (8th Edition)
- The reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, doesnot work with primary alcohols. a. Propose a mechanism for the Ritter reaction.b. Why does the Ritter reaction not work with primary alcohols?c. How does the Ritter reaction differ from the acid-catalyzed hydrolysis of a nitrile to form an amide?arrow_forwardThe reaction of a nitrile with an alcohol in the presence of a strong acid forms an N-substituted amide. This reaction, known as the Ritter reaction, does not work with primary alcohols. a. Propose a mechanism for the Ritter reaction. b. Why does the Ritter reaction not work with primary alcohols? c. How does the Ritter reaction differ from the acid-catalyzed hydrolysis of a nitrile to form an amide?arrow_forwardAcetals are often prepared during the synthetic sequence to protect the carbonyl group of an aldehyde or ketone. Such an acetal is not reactive to each of the following reagents except: a.) LiAlH4 b.) PCC(Collins) c.) H3O+ d.) RMgBr e.) NaOHarrow_forward
- Propose a mechanism for the reaction of acetic anhydride with water. a. How does this mechanism differ from the mechanism for the reaction of acetic anhydride with an alcohol?arrow_forwardPhosgene (COCl2) was used as a poison gas in World War I. What product would be formed from the reaction of phosgene with each of the following reagents? a. one equivalent of methanol b. excess methanol c. excess propylamine d. excess waterarrow_forwardHow many products are obtained when the phospholipid below is saponified? a. 4 b. 6 c. 2 d. 3 e. 5arrow_forward
- Which alkyl halide cannot be used effectively in a Gabriel synthesis of amines? Select one: a. 1-Bromo-2-methylpropane b. 2-Bromo-2-methylpropane c. Bromomethane d. Bromoethane O OOarrow_forwardDraw the product formed when pentanal (CH3 CH₂ CH₂ CH₂ CHO) is treated with each reagent. With some reagents, no reaction occurs. a. NaBH4, CH3OH b. [1] LiAiH4: [2] H₂O c. H₂, Pd-C d. PCC e. Na₂Cr₂O7, H₂SO4, H₂O f. Ag₂O, NH4OH g. [1] CH3 MgBr; [2] H₂O h. [1] C6H5 Li: [2] H₂O i. [1] (CH3)2 CuLi; [2] H₂O J. [1] HC=CNa; [2] H₂O k. [1] CH 3 C=CLI; [2] H₂O 1. The product in (a), then TBDMS-CI, imidazolearrow_forwardThe biosynthesis of lanosterol from squalene has intrigued chemists since its discovery. It is now possible, for example, to synthesize polycyclic compounds from acyclic or monocyclic precursors by reactions that form several C—C bonds in a single reaction mixture. a. Draw a stepwise mechanism for the following reaction.b. Show how X can be converted to 16,17-dehydroprogesterone.arrow_forward
- Answer the following questions about 2-acetylcyclopentanone.a. What starting materials are needed to form 2-acetylcyclopentanone by a Claisen reaction that forms bond (a)?b. What starting materials are needed to form 2-acetylcyclopentanone by a Claisen reaction that forms bond (b)?c. What product is formed when 2-acetylcyclopentanone is treated with NaOCH2CH3, followed by CH3I?d. Draw the Robinson annulation product(s) formed by reaction of 2- acetylcyclopentanone with methyl vinyl ketone (CH2=CHCOCH3).e. Draw the structure of the most stable enol tautomer(s).arrow_forwarda. Would you expect hemiacetals to be stable in basic solutions? Explain your answer.b. Acetal formation must be catalyzed by an acid. Explain why it cannot be catalyzed by CH3O-.c. Can the rate of hydrate formation be increased by hydroxide ion as well as by acid? Explain.arrow_forwarda. Propose a mechanism for the formation of succinic anhydride in the presence of acetic anhydride.b. How does acetic anhydride make it easier to form the anhydride?arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

