
Concept explainers
(a)
Interpretation:
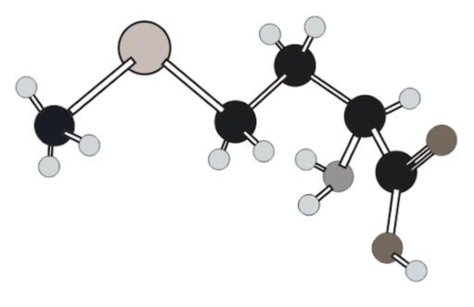
Amino acid in ball and stick model below has to be determined.
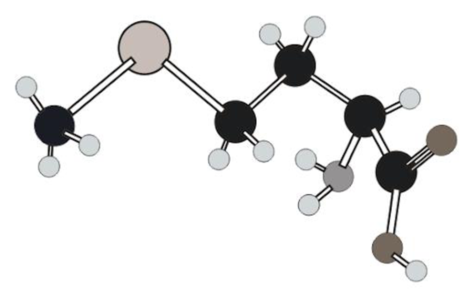
Concept Introduction:
Amino acids are the
(1) Neutral amino acids exist in their zwitterionic form at
(2) Amino acids exist with net
(3) Amino acids exist with net
(4) At physiological
(5) Value of
(6) Isoelectric point of amino acids with additional carboxylic group is around 3.
(7) Isoelectric point of amino acids with additional basic nitrogen atom is around
(a)
Explanation of Solution
In ball and stick model different atoms are as follows:
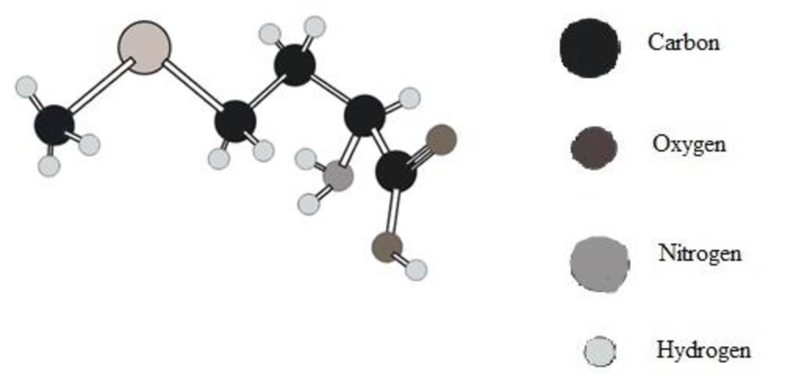
Amino acid can be drawn as follows:
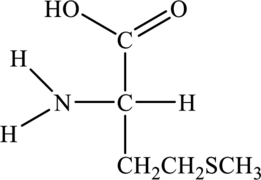
(b)
Interpretation:
Three-letter and one-letter abbreviations for amino acid in ball and stick model below have to be determined.
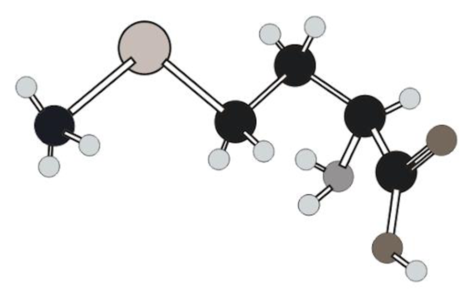
Concept Introduction:
Refer to part (a).
(b)
Explanation of Solution
Structure of given amino acid as follows:
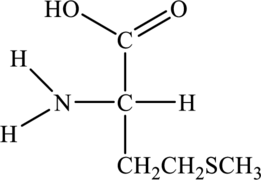
Since amino acid has
(c)
Interpretation:
The form that presents at isoelectric point for amino acid represents below has to be determined.
Concept Introduction:
Refer to part (a).
(c)
Explanation of Solution
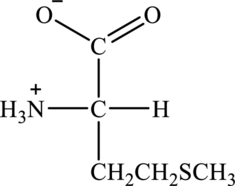
Structure of given amino acid as follows:
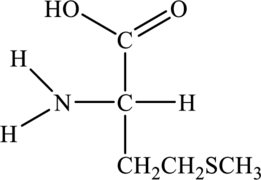
Since amino acid has
Want to see more full solutions like this?
Chapter 16 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- Identify the R group of the side chain in the following amino acids that results in the side-chain classification indicated in parentheses see Table 19.1: a. tyrosine neutral, polar b. glutamate acidic, polar c. methionine neutral, nonpolar d. histidine basic, polar e. cysteine neutral, polar f. valine neutral, nonpolararrow_forwardAt room temperature, amino acids are solids with relatively high decomposition points. Explain why.arrow_forwardWhich is NOT a characteristic of proteins? a. They contain genetic information. b. They can act as hormones. c. They can catalyze chemical reactions. d. They act in cell membrane trafficking.arrow_forward
- With reference to the following peptide: (a) Identify the N-terminal and Cterminal amino acids. (b) Name the peptide using one-letter abbreviations. (c) Label all the amide bonds in the peptide backbone.arrow_forwardIdentify each of the amino acids in the structure.arrow_forwardAre negatively charged amino acids acidic or basic and why? Are positively charged amino acids acidic or basic and why? Thank you.arrow_forward
- Please explain thoroughly each factors with supporting references. Thank you 2. What other factors influence the basic properties of amino compounds? Explain how they influence basic properties.arrow_forwardThis amino acid belongs to a "basic amino acid" (at neutral pH). Molecule "A" COO™ I H₂N-C-H CH₂ Molecule "D" COO H₂N-C-H CH₂ H₂N O structure A O O structure B structure C structure D structure E structure F Molecule "B" COO™ + H₂N-C-H H-C-OH CH 3 Molecule "E" COO H₂N-C-H CH₂ C NH C-N CH Molecule "C" COO™ H₂N-C-H CH₂ OH Molecule "F" COO™ H₂N-C-H CH₂ | COO™arrow_forwardHow to identify this amino acid?arrow_forward
- 5. For each of the following modified amino acid side chains, iden- tify the amino acid from which it was derived and the type of chemical modification that has occurred. (a) –CH,OPO, (b) –CH,CH1CO0©2, (c) – 1CH2,-NH-C102CH3arrow_forwardName each peptide using both the three-letter and one-letter abbreviations of the component amino acids.arrow_forwardWrite only the three letter abbreviation of the amino acid pictured. SH CH, O H,N--C-C-OH H.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning




