
CHEMISTRY:...,V2-W/MOD.ACCESS >CUSTOM<
17th Edition
ISBN: 9781323444498
Author: Brown
Publisher: PEARSON C
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 105AE
Benzoic acid (C6H5COOH) and aniline (C6H5NH2) are both derivatives of benzene. Benzonic acid is an acid with Ka = 6.3 × 10-5 and aniline is a base with Ka = 4.3 × 10-10.
.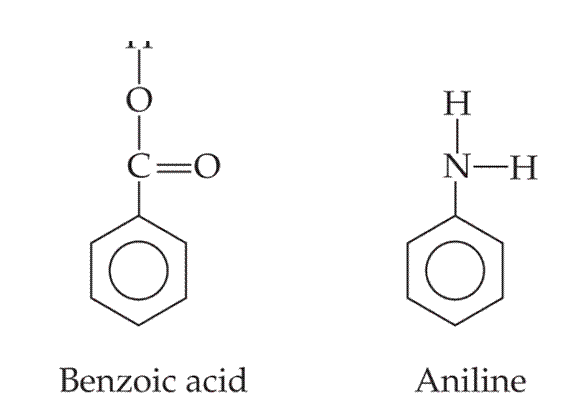
- What are the conjugate base of benzoic acid and the conjugate acid of aniline?
- Anilinium chloride (C6H5NH3CI) is a strong electrolyte that dissociates into anilinium ions (C6H5NH3+) and chloride ions. Which will be more acidic, a 0.10 M solution of benzoic acid or a 0.10 M solution of anilinium chloride?
- What is the value of the equilibrium constant for the following equilibrium? C 6H 5COOH(aq) + C 6H 5NH 2(aq) ⇌
C6H5COO -(aq) + C6H5NH3 +(aq)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
CHEMISTRY:...,V2-W/MOD.ACCESS >CUSTOM<
Ch. 16.2 - Practice Exercise 1 Consider the following...Ch. 16.2 - Prob. 16.1.2PECh. 16.2 - Prob. 16.2.1PECh. 16.2 - Practice Exercise 2 When lithium oxide (Li2O) is...Ch. 16.2 - Based on information in Figure 16.4, place the...Ch. 16.2 - Practice Exercise 2 For each reaction, use Figure...Ch. 16.3 - Practice Exercise 1 In a certain acidic solution...Ch. 16.3 - Practice Exercise 2 Indicate whether solutions...Ch. 16.3 - Prob. 16.5.1PECh. 16.3 - Prob. 16.5.2PE
Ch. 16.4 - Practice Exercise 1 A solution at 250C has [OH-] =...Ch. 16.4 - Practice Exercise 2 In a sample of lemon juice,...Ch. 16.4 - Practice Exercise 1 A solution at 25° C has pOH =...Ch. 16.4 - Prob. 16.7.2PECh. 16.5 - Practice Exercise 1 Order the following three...Ch. 16.5 - Prob. 16.8.2PECh. 16.5 - Practice Exercise 1 Order the following three...Ch. 16.5 - Prob. 16.9.2PECh. 16.6 - Prob. 16.10.1PECh. 16.6 - Practice Exercise 2 Niacin, one of the B vitamins,...Ch. 16.6 - Prob. 16.11.1PECh. 16.6 - Practice Exercise 2 A 0.020 M solution of niacin...Ch. 16.6 - Practice Exercise 1 What is the pH of a 0.40 M...Ch. 16.6 - Practice Exercise 2 The Ka for niacin (Sample...Ch. 16.6 - Prob. 16.13.1PECh. 16.6 - Prob. 16.13.2PECh. 16.6 - Practice Exercise 1 What is the pH of a 0.28 M...Ch. 16.6 - Practice Exercise 2 Calculate the pH of a 0.020 M...Ch. 16.7 - Prob. 16.15.1PECh. 16.7 - Practice Exercise 2 Which of the following...Ch. 16.7 - Prob. 16.16.1PECh. 16.7 - Practice Exercise 2 What is the morality of an...Ch. 16.8 - Practice Exercise 1 By using information from...Ch. 16.8 - Practice Exercise 2 Based on information in...Ch. 16.9 - Prob. 16.18.1PECh. 16.9 - Prob. 16.18.2PECh. 16.9 - Practice Exercise 1 How many of the following...Ch. 16.9 - Practice Exercise 2 Predict whether the...Ch. 16.10 - Prob. 16.20.1PECh. 16.10 - In each pair, choose the compound that gives the...Ch. 16 - Prob. 1DECh. 16 - a. Identify the Br ted-Lowry acid and base in the...Ch. 16 - Prob. 2ECh. 16 - Prob. 3ECh. 16 - Prob. 4ECh. 16 - 16.5 The following diagrams represent aqueous...Ch. 16 - Prob. 6ECh. 16 - Which of these statements about how the percent...Ch. 16 - 16.8 Each of the three molecules shown here...Ch. 16 - Prob. 9ECh. 16 - Which of the following diagrams best represent an...Ch. 16 - Prob. 11ECh. 16 - Prob. 12ECh. 16 - Prob. 13ECh. 16 - 16.14 Which of the following statements is...Ch. 16 - Prob. 15ECh. 16 - Prob. 16ECh. 16 - Identify the Bronsted-Lowry acid and the...Ch. 16 - Prob. 18ECh. 16 - Prob. 19ECh. 16 - Prob. 20ECh. 16 - Prob. 21ECh. 16 - Prob. 22ECh. 16 - Prob. 23ECh. 16 - Prob. 24ECh. 16 - Prob. 25ECh. 16 - Prob. 26ECh. 16 - Prob. 27ECh. 16 - Prob. 28ECh. 16 - 16.29 Calcualte [H +] for each of the following...Ch. 16 - Prob. 30ECh. 16 - 16.31 At the freezing point of water (0 o C), K10...Ch. 16 - Prob. 32ECh. 16 - Prob. 33ECh. 16 - Prob. 34ECh. 16 - 16.35 Complete the following table by calculating...Ch. 16 - Prob. 36ECh. 16 - Prob. 37ECh. 16 - 16.38 Carbon dioxide in the atmosphere dissolves...Ch. 16 - Prob. 39ECh. 16 - Prob. 40ECh. 16 - Prob. 41ECh. 16 - Prob. 42ECh. 16 - Prob. 43ECh. 16 - Prob. 44ECh. 16 - Prob. 45ECh. 16 - Prob. 46ECh. 16 - Prob. 47ECh. 16 - Prob. 48ECh. 16 - Prob. 49ECh. 16 - write the chemical equation and the Ka expression...Ch. 16 - Prob. 51ECh. 16 - Prob. 52ECh. 16 - Prob. 53ECh. 16 - Prob. 54ECh. 16 - Prob. 55ECh. 16 - Prob. 56ECh. 16 - Prob. 57ECh. 16 - Prob. 58ECh. 16 - Calculate the pH of each of the following solution...Ch. 16 - Prob. 60ECh. 16 - Prob. 61ECh. 16 - Prob. 62ECh. 16 - Calculate the percent ionization of hydrazoic acid...Ch. 16 - 16.64 Calculate the percent ionization of...Ch. 16 - Prob. 65ECh. 16 - Prob. 66ECh. 16 - Prob. 67ECh. 16 - 16.68 The hypochlorite ion, CIO- , acts as a weak...Ch. 16 - Prob. 69ECh. 16 - Prob. 70ECh. 16 - Calculate the molar concentration of OH- in a...Ch. 16 - 16.72 Calculate the molar concentration of OH- in...Ch. 16 - Prob. 73ECh. 16 - Prob. 74ECh. 16 - Prob. 75ECh. 16 - Prob. 76ECh. 16 - a. Given that Ka for acetic acid is 1.8 10-5 and...Ch. 16 - 16.78
a. Given that Kb for ammonia is 1.8 X 10 -5...Ch. 16 - Prob. 79ECh. 16 - Prob. 80ECh. 16 - Prob. 81ECh. 16 - Pyridinium bromide (C5H5NHBr) is a strong...Ch. 16 - Prob. 83ECh. 16 - Prob. 84ECh. 16 - Prob. 85ECh. 16 - 16.86 An unknown salt is either KBr, NH4 C1, KCN,...Ch. 16 - Prob. 87ECh. 16 - Prob. 88ECh. 16 - 16.89 Based on their compositions and structures...Ch. 16 - Prob. 90ECh. 16 - 16.91 Indicate whether each of the following...Ch. 16 - Prob. 92ECh. 16 - Prob. 93ECh. 16 - Prob. 94ECh. 16 - Prob. 95ECh. 16 - Prob. 96ECh. 16 - Prob. 97ECh. 16 - Prob. 98ECh. 16 - Prob. 99AECh. 16 - Prob. 100AECh. 16 - Prob. 101AECh. 16 - Prob. 102AECh. 16 - Prob. 103AECh. 16 - Prob. 104AECh. 16 - Benzoic acid (C6H5COOH) and aniline (C6H5NH2) are...Ch. 16 - Prob. 106AECh. 16 - Prob. 107AECh. 16 - Prob. 108AECh. 16 - Butyric acid is responsible for the foul smell of...Ch. 16 - Prob. 110AECh. 16 - Prob. 111AECh. 16 - Prob. 112AECh. 16 - 1S.113 Many moderately large organic molecules...Ch. 16 - Prob. 114AECh. 16 - Prob. 115AECh. 16 - Prob. 116IECh. 16 - Prob. 117IECh. 16 - Prob. 118IECh. 16 - Prob. 119IECh. 16 - 16.120 At 50 oC, the ion-product constant for H2...Ch. 16 - Prob. 121IECh. 16 - Prob. 122IECh. 16 - Prob. 123IECh. 16 - Prob. 124IECh. 16 - Prob. 125IECh. 16 - Prob. 126IE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 8-21 Write the formula for the conjugate base of each acid. (a) H2PO4- (b) H2S (c) HCO3- (d) CH3CH2OH (e) H2Oarrow_forwardHow is acid strength related to the value of Ka? What is the difference between strong acids and weak acids (see Table 13-1)? As the strength of an acid increases, what happens to the strength of the conjugate base? How is base strength related to the value of Kb? As the strength of a base increases, what happens to the strength of the conjugate acid?arrow_forwardUsing the diagrams shown in Problem 10-37, which of the four acids is the weakest acid?arrow_forward
- How do the components of a conjugate acid—base pair differ from one another4? Give an example of a conjugate acid—base pair to illustrate your answer.arrow_forwardFor oxyacids, how does acid strength depend on a. the strength of the bond to the acidic hydrogen atom? b. the electronegativity of the element bonded to the oxygen atom that bears the acidic hydrogen? c. the number of oxygen atoms? How does the strength of a conjugate base depend on these factors? What type of solution forms when a nonmetal oxide dissolves in water? Give an example of such an oxide. What type of solution forms when a metal oxide dissolves in water? Give an example of such an oxide.arrow_forwardWhich acid has the strongest conjugate base? (a) HNO2 (b) C6H5CO2H (c) HCN (d) HClarrow_forward
- Classify each of the acids in Problem 10-20 as a strong acid or a weak acid. a. H2SO4 (sulfuric acid) b. HC2H3O2 (acetic acid) c. H2C5H6O4 (glutaric acid) d. HCN (cyanic acid)arrow_forward8-17 For each of the following, tell whether the base is strong or weak. (a) NaOH (b) Sodium acetate (c) KOH (d) Ammonia (e) Waterarrow_forwardFind the value of Kb for the conjugate base of the following organic acids. (a) picric acid used in the manufacture of explosives; Ka = 0.16 (b) trichloroacetic acid used in the treatment of warts; Ka = 0.20arrow_forward
- Find the value of Kb for the conjugate base of the following organic acids. (a) picric acid used in the manufacture of explosives; K a =0.16 (b) trichloroacetic acid used in the treatment of warts; K a =0.20arrow_forwardUse Table 13-3 to help answer the following questions. a. Which is the stronger base, ClO4 or C6H5NH2? b. Which is the stronger base, H2O or C6H5NH2? c. Which is the stronger base, OH or C6H5NH2? d. Which is the stronger base, C6H5NH2 or CH3NH2?arrow_forwardFor conjugate acidbase pairs, how are Ka and Kb related? Consider the reaction of acetic acid in water CH3CO2H(aq)+H2O(l)CH3CO2(aq)+H3O+(aq) where Ka = 1.8 105 a. Which two bases are competing for the proton? b. Which is the stronger base? c. In light of your answer to part b. why do we classify the acetate ion (CH3CO2) as a weak base? Use an appropriate reaction to justify your answer. In general, as base strength increases, conjugate acid strength decreases. Explain why the conjugate acid of the weak base NH3 is a weak acid. To summarize, the conjugate base of a weak acid is a weak base and the conjugate acid of a weak base is a weak acid (weak gives you weak). Assuming Ka for a monoprotic strong acid is 1 106, calculate Kb for the conjugate base of this strong acid. Why do conjugate bases of strong acids have no basic properties in water? List the conjugate bases of the six common strong acids. To tie it all together, some instructors have students think of Li+, K+, Rb+, Cs+, Ca2+, Sr2+, and Ba2+ as the conjugate acids of the strong bases LiOH, KOH. RbOH, CsOH, Ca(OH)2, Sr(OH)2, and Ba(OH)2. Although not technically correct, the conjugate acid strength of these cations is similar to the conjugate base strength of the strong acids. That is, these cations have no acidic properties in water; similarly, the conjugate bases of strong acids have no basic properties (strong gives you worthless). Fill in the blanks with the correct response. The conjugate base of a weak acid is a_____base. The conjugate acid of a weak base is a_____acid. The conjugate base of a strong acid is a_____base. The conjugate acid of a strong base is a_____ acid. (Hint: Weak gives you weak and strong gives you worthless.)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY