
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 15.15, Problem 33P
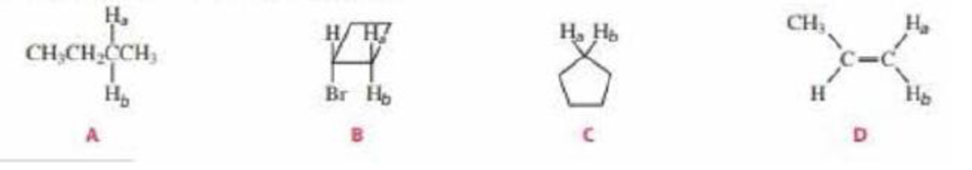
- a. For the following compounds, which pairs of hydrogens (Ha and Hb) are enantiotopic hydrogens?
- b. Which pairs are diastereotopic hydrogens?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
a. For the following compounds, which pairs of hydrogens (Ha and Hb) are enantiotopic hydrogens?b. Which pairs are diastereotopic hydrogens?
For the following compounds, which pairs of hydrogens (Ha and Hb) are enantiotopic hydrogens? b. Which pairs are diastereotopic hydrogens?
(30) Take a look at the two multi-substituted benzene compounds below. Trinitrotoluene (TNT) has its NO, groups on carbons 2, 4, and 6.
Which of the two fits this numerical scheme? Hint: Toluene is a special name for benzene whose primary functional group is a methyl group
substituted in place of one of its hydrogens.
CH3
CH3
O,N.
ZON
O,N
NO2
NO2
NO2
Chapter 15 Solutions
Organic Chemistry
Ch. 15.1 - Prob. 1PCh. 15.1 - Prob. 2PCh. 15.4 - How many signals would you expect to see in the 1H...Ch. 15.4 - Prob. 4PCh. 15.4 - How could you distinguish the 1H NMR spectra of...Ch. 15.4 - Draw an isomer of dichlorocyclopropane that gives...Ch. 15.5 - Prob. 7PCh. 15.5 - Prob. 8PCh. 15.5 - Prob. 9PCh. 15.5 - Where would you expect to find the 1H NMR signal...
Ch. 15.6 - Prob. 11PCh. 15.7 - Prob. 12PCh. 15.7 - Prob. 13PCh. 15.7 - Without referring to Table 14.1, label the proton...Ch. 15.8 - [18]-Annulene shows two signals in its 1H NMR...Ch. 15.9 - How would integration distinguish the 1H NMR...Ch. 15.9 - Which of the following compounds is responsible...Ch. 15.10 - Prob. 19PCh. 15.10 - Explain how the following compounds, each with the...Ch. 15.10 - The 1H NMR spectra of two carboxylic acids with...Ch. 15.11 - Draw a diagram like the one shown in Figure 14.12...Ch. 15.12 - Indicate the number of signals and the...Ch. 15.12 - How can their 1H NMR spectra distinguish the...Ch. 15.12 - Identify each compound from its molecular formula...Ch. 15.12 - Prob. 27PCh. 15.12 - Propose structures that are consistent with the...Ch. 15.12 - Describe the 1H NMR spectrum you would expect for...Ch. 15.13 - Prob. 30PCh. 15.13 - Identify the compound with molecular formula...Ch. 15.14 - Prob. 32PCh. 15.15 - a. For the following compounds, which pairs of...Ch. 15.17 - Explain why the chemical shift of the OH proton of...Ch. 15.17 - Prob. 37PCh. 15.17 - Prob. 38PCh. 15.17 - Prob. 39PCh. 15.20 - Answer the following questions for each compound:...Ch. 15.20 - Prob. 41PCh. 15.20 - How can 1,2-, 1,3-, and 1,4-dinitrobenzene be...Ch. 15.20 - Identify each compound below from its molecular...Ch. 15.22 - Prob. 44PCh. 15.22 - What does cross peak X in Figure 14.34 tell you?Ch. 15 - Prob. 46PCh. 15 - Draw a spitting diagram for the Hb proton and give...Ch. 15 - Label each set of chemically equivalent protons,...Ch. 15 - Match each of the 1H NMR spectra with one of the...Ch. 15 - Determine the ratios of the chemically...Ch. 15 - How can 1H NMR distinguish between the compounds...Ch. 15 - Prob. 52PCh. 15 - The 1H NMR spectra of three isomers with molecular...Ch. 15 - Prob. 54PCh. 15 - Prob. 55PCh. 15 - Prob. 56PCh. 15 - Compound A, with molecular formula C4H9Cl, shows...Ch. 15 - The 1H NMR spectra of three isomers with molecular...Ch. 15 - Would it be better to use 1H NMR or 13C NMR...Ch. 15 - There are four esters with molecular formula...Ch. 15 - An alkyl halide reacts with an alkoxide ion to...Ch. 15 - Identity each of the following compounds from its...Ch. 15 - Identity each of the following compounds from its...Ch. 15 - Prob. 64PCh. 15 - How can the signals in the 6.5 to 8.1 ppm region...Ch. 15 - The 1H NMR spectra of two compounds, each with...Ch. 15 - Draw a splitting diagram for the Hb proton if Jbc...Ch. 15 - Sketch the following spectra that would be...Ch. 15 - How can 1H NMR be used to prove that the addition...Ch. 15 - Identity each of the following compounds from its...Ch. 15 - Dr. N. M. Arr was called in to help analyze the 1H...Ch. 15 - Calculate the amount of energy (in calories)...Ch. 15 - The following 1H NMR spectra are four compounds,...Ch. 15 - When compound A (C5H12O) is treated with HBr, it...Ch. 15 - Identity each of the following compounds from its...Ch. 15 - Identify each of the following compounds from its...Ch. 15 - Identity the compound with molecular formula...Ch. 15 - Identify the compound with molecular formula C6H14...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the structure of the product of the Diels-Alder reaction below. CH3 a CH3 CH3OC-C=C-COCH3 • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Include H atoms at chiral centers only. / • If a group is achiral, do not use wedged or hashed bonds on it. • If the reaction produces a racemic mixture, just draw one stereoisomer. O. Sn » [ ]# ?arrow_forwardPlease help with 4c, 4d, and 4e. We havent learned kaw of equivalents so please do 4c a different way. thank youarrow_forward4. Explain why the protons on the sp³-hybridized carbon of propene (CH3-CH=CH₂) are more acidic than the protons on the sp²-hybridized carbons of propene, even though sp2-hybridized carbons are more electronegative than sp³-hybridized carbons. Use both structures and sentences in your answer.arrow_forward
- 3. Determine the relationship for the following pairs of structures. Use "E" for enantiomers, "D" for diastereomers, "C" for constitutional isomers, "S" for same, and "NS" (not same) for different compounds. a. b. с. d. е. f. Н н iPr "Де Br Br OH CH3 CH3 Ή OH OH OH Br Br CH3 CH3 Br -Н НО -Н H3C -ОН COOH Br Br Br Н H3C- Br OH CH3 Н CH3 iPr CH3 Н- -Br НО _H H OH OH CH3 ОН -OH COOHarrow_forwardAnswer the following questions for the MOs of 1,3-butadiene: a. Which are pie bonding MOs, and which are p* antibonding MOs? b. Which MOs are symmetric, and which are antisymmetric? c. Which MO is the HOMO and which is the LUMO in the ground state? d. Which MO is the HOMO and which is the LUMO in the excited state? e. What is the relationship between the HOMO and the LUMO and symmetric and antisymmetric orbitals?arrow_forwardA B C D E 1. NaBH4 2. H3O+ 1. CH3MgBr 2. H3O+ 1. (CH3)2CuLi 2. H3O+ 1. CH3Li 2. H3O+ 1. (CH3CH2)2CuLi 2. H3O+arrow_forward
- A. B. C. Me CH3 ClC H ¿ OH NH₂ H Br-C-C H3C H Br CH3 Comparing structure pair in B OH Comparing structure pair in C [Select] Me HC CI Comparing structure pair in A [Select] [Select] Same structure drawn differently Enantiomers Diastereomers NH₂ Br H3C CH3 Br HC-CCH3 H For the following two structures compared in parts A and B, determine whether they are enantiomers OR diastereomers OR the same structure drawn differently (non-bonding electrons not included for clarity).arrow_forwardII. Indicate the correct correlation between the following pairs of molecules. Write the appropriate letter in the box. S = Same Compound, E = Enantiomers, D = Diastereomers, and C = Constitutional Isomers. (? Br CI CI Br 7. and 8. 9. and and Br "Br CI CH3 CI CH3 HBr and CH H- Br and 11. 10. Br Br -H CH,CH3 CH,CH3arrow_forwardDraw structural formulas for the diene and dienophile that combine in a Diels-Alder reaction to form the product shown CH3CH2O. Diene + Dienophile Co,CH3 •Consider E Z stereochemistry of alkenes. •Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate multiple products using the sign from the drop-down menu. C. P. opy aste %3Darrow_forward
- 2.) Give structures for the following compounds that show two PMR sin- glets each. a. C,H,O, b. C,H10 c. CH, CI d. C,H,O, e. C,H,CI f. C,H,CI,arrow_forwardIdentify the meso compound or compounds. H. H. H. Br-C-CI Cl -C-Br Br -C-CI C-Br Br-C-CI Br-C-CI CI-C-Br CI-C-Br H. H. H. A D a) A d) D b) e) B and C C f) A and D c)arrow_forward1. An aliphatic ketone absorbs at 1,715 cm-1. What is the frequency of this vibration in hertz, which is cycles per second or just per second, reciprocal seconds? 2. What is the energy equivalent of this stretching vibration in kcal/mole? 3. Why does 3,4-diethyl-3-hexene not have a carbon to carbon double bond stretching absorption band? 4. Why does a carbon to oxygen double bond absorption band have a greater intensity than a carbon to carbon double bond absorption band? 5. Using only IR, explain in detail how one could most easily differentiate between oct-1-ene and oct-1-yne if all carbon to carbon bonds are ignored. 6. Using only IR, explain in detail how one could most easily differentiate between butan-1-ol and butanoic acid.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY