
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 15, Problem 47P
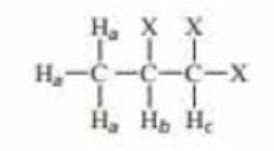
Draw a spitting diagram for the Hb proton and give its multiplicity if
- a. Jba = Jbc.
- b. Jba = 2Jbc.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the Ka value for each of the compounds? Express the answers in proper scientific notation.
A. Determine the Ka value for the compound with
pKa = 10.26.
H3C-
OH
B. Determine the K₂ value for the compound with
pK₂ = 16.50.
OH
C. Determine the K₂ value for the compound with
pKa
= 5.21.
K₁ =
K₂ =
K₁ =
Which of the following is R configuration?
...
OH
i
Br
(1)
a. (1)
b. (11)
C. (III)
€
(11)
O
d. (IV)
(111)
(IV)
Draw a splitting diagram for the Hb proton if Jbc = 10 and Jba = 5.
Chapter 15 Solutions
Organic Chemistry
Ch. 15.1 - Prob. 1PCh. 15.1 - Prob. 2PCh. 15.4 - How many signals would you expect to see in the 1H...Ch. 15.4 - Prob. 4PCh. 15.4 - How could you distinguish the 1H NMR spectra of...Ch. 15.4 - Draw an isomer of dichlorocyclopropane that gives...Ch. 15.5 - Prob. 7PCh. 15.5 - Prob. 8PCh. 15.5 - Prob. 9PCh. 15.5 - Where would you expect to find the 1H NMR signal...
Ch. 15.6 - Prob. 11PCh. 15.7 - Prob. 12PCh. 15.7 - Prob. 13PCh. 15.7 - Without referring to Table 14.1, label the proton...Ch. 15.8 - [18]-Annulene shows two signals in its 1H NMR...Ch. 15.9 - How would integration distinguish the 1H NMR...Ch. 15.9 - Which of the following compounds is responsible...Ch. 15.10 - Prob. 19PCh. 15.10 - Explain how the following compounds, each with the...Ch. 15.10 - The 1H NMR spectra of two carboxylic acids with...Ch. 15.11 - Draw a diagram like the one shown in Figure 14.12...Ch. 15.12 - Indicate the number of signals and the...Ch. 15.12 - How can their 1H NMR spectra distinguish the...Ch. 15.12 - Identify each compound from its molecular formula...Ch. 15.12 - Prob. 27PCh. 15.12 - Propose structures that are consistent with the...Ch. 15.12 - Describe the 1H NMR spectrum you would expect for...Ch. 15.13 - Prob. 30PCh. 15.13 - Identify the compound with molecular formula...Ch. 15.14 - Prob. 32PCh. 15.15 - a. For the following compounds, which pairs of...Ch. 15.17 - Explain why the chemical shift of the OH proton of...Ch. 15.17 - Prob. 37PCh. 15.17 - Prob. 38PCh. 15.17 - Prob. 39PCh. 15.20 - Answer the following questions for each compound:...Ch. 15.20 - Prob. 41PCh. 15.20 - How can 1,2-, 1,3-, and 1,4-dinitrobenzene be...Ch. 15.20 - Identify each compound below from its molecular...Ch. 15.22 - Prob. 44PCh. 15.22 - What does cross peak X in Figure 14.34 tell you?Ch. 15 - Prob. 46PCh. 15 - Draw a spitting diagram for the Hb proton and give...Ch. 15 - Label each set of chemically equivalent protons,...Ch. 15 - Match each of the 1H NMR spectra with one of the...Ch. 15 - Determine the ratios of the chemically...Ch. 15 - How can 1H NMR distinguish between the compounds...Ch. 15 - Prob. 52PCh. 15 - The 1H NMR spectra of three isomers with molecular...Ch. 15 - Prob. 54PCh. 15 - Prob. 55PCh. 15 - Prob. 56PCh. 15 - Compound A, with molecular formula C4H9Cl, shows...Ch. 15 - The 1H NMR spectra of three isomers with molecular...Ch. 15 - Would it be better to use 1H NMR or 13C NMR...Ch. 15 - There are four esters with molecular formula...Ch. 15 - An alkyl halide reacts with an alkoxide ion to...Ch. 15 - Identity each of the following compounds from its...Ch. 15 - Identity each of the following compounds from its...Ch. 15 - Prob. 64PCh. 15 - How can the signals in the 6.5 to 8.1 ppm region...Ch. 15 - The 1H NMR spectra of two compounds, each with...Ch. 15 - Draw a splitting diagram for the Hb proton if Jbc...Ch. 15 - Sketch the following spectra that would be...Ch. 15 - How can 1H NMR be used to prove that the addition...Ch. 15 - Identity each of the following compounds from its...Ch. 15 - Dr. N. M. Arr was called in to help analyze the 1H...Ch. 15 - Calculate the amount of energy (in calories)...Ch. 15 - The following 1H NMR spectra are four compounds,...Ch. 15 - When compound A (C5H12O) is treated with HBr, it...Ch. 15 - Identity each of the following compounds from its...Ch. 15 - Identify each of the following compounds from its...Ch. 15 - Identity the compound with molecular formula...Ch. 15 - Identify the compound with molecular formula C6H14...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- a. Can a low-resolution mass spectrometer distinguish between C2H5+ and CHO+ ?b. Can a high-resolution mass spectrometer distinguish between them?arrow_forward(iii) H,O2 + Н-Br (iv) (1) O3 + (ii) Zn/H,Oarrow_forwardWhy is it important for solutions to be diluted before they are run through spectroscopy?arrow_forward
- In mass spectrometry, the molecular ion peak occurs at O A. the largest m/z value. O B. the smallest m/z value. O C. the m/z value of the most abundant ion. O D. an m/z value equal to the molecular weight of the sample molecule.arrow_forwardWhich of the indicated protons would have the smallest pKa value? C. H. H CH3 EC H H. d. b. b. C A Moving to another question will save this response. O O O 0arrow_forward12. Which absorption spectrum corresponds to a red solution? a. (a) b. (b) C. (c) d. (a) and (c) e. (b) and (c) 400 nm e 700 nmarrow_forward
- The leaves of the Brazilian Tree Senna multijunga contain a number of pryidine alkaloids that inhibit acetylcholinterinase. Two recentyl isolated isomeric compounds have the strcture have the strcture shown below. (NOTE: M=293) Use the mass spectral data provided to determine the precise location of the hydroxyl group in each isomer. Isomer A: EI-MS, m/z(rel. int): 222(20), 150(10), 136(25), 123(100) Isomer B:EI-MS, m/z(re;. int): 236(20), 150(10), 136(25), 123(100)arrow_forwardIf you were performing following reaction (5) in the lab, how could you use IR spectroscopy to determine if the reaction worked? SELECT THE ANSWER FROM THE FOLLOWING THREE OPTIONS: A) Formation of C-Br bond and loss of C=C bond B) Loss of C-Br bond and appearance of C-OMe bond C) Loss of C-Br bond and appearance of C=C bondarrow_forwardPredict the ¹H shifts of the protons on the labeled carbons of the following chemical structure: OH NH₂ 1 2 a. H(1)=6.44, H(2)=6.40 b. H(1)=6.64, H(2)=6.60 c. H(1)-5.46, H(2)=5.40arrow_forward
- 1. Which one of the given compounds is consistent with the mass spectrum below? 100 40 20 10 20 25 30 35 40 5o 55 45 60 65 70 75 m/z Courtesy of SDBS: National Institute of Advanced Industrial Science and Technology A. CH,CH,CH(CH,), В. CH,CHOHCH,CH, C. CH,CH,OCH,CH, D. CH,CH,NHCH,CH, E. CH,CH,CH,CH, Relative htensityarrow_forward7. Given the pK, information for different acids below, place the following acids in order of increasing acid strength (from weakest to strongest). Anilinium ion, CSH6N*, pKa = 4.60 Benzilic acid, pka = 3.09 %3D Chloroacetic acid, pKa = 2.98 %3D Dibromophenol, pka = 8.06 %3D a) Anilinium ion → Benzilic acid → Chloroacetic acid → Chloroacetic acid b) Dibromophenol → Chloroacetic acid → Benzilic acid → Anilinium ion c) Chloroacetic acid → Benzilic acid → Anilinium ion → Dibromophenol d) Dibromophenol → Anilinium ion → Benzilic acid → Chloroacetic acid e) Chloroacetic acid → Anilinium ion → Dibromophenol→ Benzilic acid f) none of the abovearrow_forwardConsider the mass spectrum below. 100 80- 60- 20 0.0- 0.0 60 100 miz SDBS, National Institute of Advanced Industrial Science and Technology Which peak is the molecular ion? Choose one OA OB OD. OGarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY