
Figure 15.32 show a pV diagram for an ideal gas in which its pressure tripled from a to b when 534 J of heat was put into the gas. (a) How much work was done by the gas between a and b? (b) Without doing any calculations, decide whether the temperature of this gas increased, decreased, or remained the same between a and b. Explain your reasoning. (c) By how much did the internal energy of the gas change between a and b? Did it increase or decrease? (d) What is the temperature of the gas at point b in terms of its temperature at a, Ta?
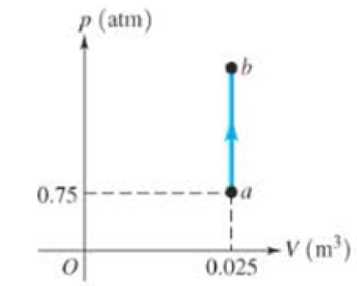
Figure 15.32
Problem 49.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
College Physics (10th Edition)
Additional Science Textbook Solutions
Lecture- Tutorials for Introductory Astronomy
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Life in the Universe (4th Edition)
Physics for Scientists and Engineers with Modern Physics
- A 2.00-mol sample of a diatomic ideal gas expands slowly and adiabatically from a pressure of 5.00 atm and a volume of 12.0 L to a final volume of 30.0 L. (a) What is the final pressure of the gas? (b) What are the initial and final temperatures? Find (c) Q, (d) Eint, and (e) W for the gas during this process.arrow_forwardIf a gas is compressed isothermally, which of the following statements is true? (a) Energy is transferred into the gas by heat. (b) No work is done on the gas. (c) The temperature of the gas increases. (d) The internal energy of the gas remains constant. (e) None of those statements is true.arrow_forwardFor a temperature increase of 10 at constant volume, what is the heat absorbed by (a) 3.0 mol of a dilute monatomic gas; (b) 0.50 mol of a dilute diatomic gas; and (c) 15 mol of a dilute polyatomic gas?arrow_forward
- A 1.00-mol sample of hydrogen gas is heated at constant pressure from 300 K to 420 K. Calculate (a) the energy transferred to the gas by heat, (b) the increase in its internal energy, and (c) the work done on the gas.arrow_forwardA gas expands from I to F in the figure below. The energy added to the gas by heat is 252 J when the gas goes from I to F along the diagonal path. IN P (atm) 4 3 1 V (liters) 4 1 3 (a) What is the change in internal energy of the gas? Use the relations between various features of the graph and the work done on the gas to find the energy added by work and then use your result to find the change in internal energy of the gas. J (b) How much energy must be added to the gas by heat along the indirect path IAF?arrow_forwardA gas expands from I to F in the figure below. The energy added to the gas by heat is 422 J when the gas goes from I to F along the diagonal path. Three paths are plotted on a PV diagram, which has a horizontal axis labeled V (liters), and a vertical axis labeled P (atm). The green path starts at point I (2,4), extends vertically down to point B (2,1), then extends horizontally to point F (4,1). The blue path starts at point I (2,4), and extends down and to the right to end at point F (4,1). The orange path starts at point I (2,4), extends horizontally to the right to point A (4,4), then extends vertically down to end at point F (4,1). (a) What is the change in internal energy of the gas? J(b) How much energy must be added to the gas by heat for the indirect path IAF to give the same change in internal energy? Jarrow_forward
- Consider the thermodynamic process, A->B->C->A shown above. The heat absorbed during A->B is 591J. If the change in internal energy during B->C is 4146J, What is the change in internal energy in SI units during C->A? Express only the number of your answer with 4 significant figures.arrow_forwardThe temperature at state A is 20.0°C, that is 293 K. During the last test, you have found the temperature at state D is 73.0 K and n = 164 moles for this monatomic ideal gas. What is the work done by the gas for process A to D, in MJ (MegaJoules)? 1.0 atm = 1.00 x 10° Pa. Your answer needs to have 2 significant figures, including the negative sign in your answer if needed. Do not include the positive sign if the answer is positive. No unit is needed in your answer, it is already given in the question statement. P (atm) A 4 3 2 B V (m) 4.arrow_forwardAn ideal gas is taken from a to b on the pV diagram shown in the figure. During this process, 700 J of heat is added and the pressure doubles. (a) How much work is done by or on the gas? Explain. (b) How does the temperature of the gas at a compare to its temperature at b? Be specific. (c) How does the internal energy of the gas at a compare to the internal energy at b? Be specific and explain.arrow_forward
- A gas expands from I to F in the figure below. The energy added to the gas by heat is 302 J when the gas goes from I to Falong the diagonal path. Three paths are plotted on a PV diagram, which has a horizontal axis labeled V(liters), and a vertical axis labeled P (atm). The green path starts at point I (2,4), extends vertically down to point B(2,1), then extends horizontally to point F (4,1). The blue path starts at point I (2,4), and extends down and to the right to end at point F (4,1). The orange path starts at point I(2,4), extends horizontally to the right to point A (4,4), then extends vertically down to end at point F(4,1). (a) What is the change in internal energy of the gas?J(b) How much energy must be added to the gas by heat for the indirect path IAF to give the same change in internal energy?Jarrow_forwardA gas expands from I to F in the figure below. The energy added to the gas by heat is 212 J when the gas goes from I to F along the diagonal path. Three paths are plotted on a PV diagram, which has a horizontal axis labeled V (liters), and a vertical axis labeled P (atm). The green path starts at point I (2,4), extends vertically down to point B (2,1), then extends horizontally to point F (4,1). The blue path starts at point I (2,4), and extends down and to the right to end at point F (4,1). The orange path starts at point I (2,4), extends horizontally to the right to point A (4,4), then extends vertically down to end at point F (4,1). (a) What is the change in internal energy of the gas? Use the relations between various features of the graph and the work done on the gas to find the energy added by work and then use your result to find the change in internal energy of the gas. J(b) How much energy must be added to the gas by heat for the indirect path IAF to give the same change in…arrow_forwardAn ideal gas in a container with a sliding piston is held at atmospheric pressure. Heat is added and the volume increases from 1 m^3 to 3 m^3. The initial temp of the gas is 0 degrees celcius. Question A: How many moles of gas are in this container? Question B: How much work does the gas perform on the piston? Please answer in J Question C: What is the final temperature of the gas? Please answer in degrees celcius Question D: what is the change in internal energy of the gas? Please answer in J Question E: How much heat was added to the gas? Please answer in Jarrow_forward
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning


