
Concept explainers
(a)
Interpretation:
Chalk should be classified as chiral or achiral.
Concept Introduction:
Chirality is the presence of an asymmetric carbon center in a molecule and a molecule which contains a chiral center cannot superimpose on its mirror image. To consider as chiral, molecule or object and its mirror image should not superimpose. To consider as achiral, molecule or object and its mirror image should be superimposed with each other.
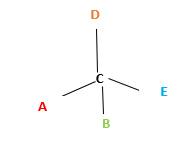
In the above diagram, where C is the chiral center/ asymmetric carbon center.
A, B, D, E are four different
Answer to Problem 21P
Chalk is achiral.
Explanation of Solution
To consider as chiral, molecule or object and its mirror image should not superimpose. To consider as achiral, molecule or object and its mirror image should be superimposed with each other. The mirror image of chalk is identical and is superimposable on their mirror images. Though they have not sharpened edges, the object and its mirror images are superimposable with each other. Thus, chalk is achiral.
(b)
Interpretation:
Pair of shoes should be classified as chiral or achiral.
Concept Introduction:
Chirality is the presence of an asymmetric carbon center in a molecule and a molecule which contains a chiral center cannot superimpose on its mirror image. To consider as chiral, molecule or object and its mirror image should not superimpose. To consider as achiral, molecule or object and its mirror image should be superimposed with each other.
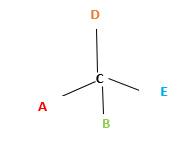
In the above diagram, where C is the chiral center/ asymmetric carbon center.
A, B, D, E are four different functional groups.
Answer to Problem 21P
Pair of shoes are chiral.
Explanation of Solution
To consider as chiral, molecule or object and its mirror image should not superimpose. To consider as achiral, molecule or object and its mirror image should be superimposed with each other. The mirror image of pair of shoes arenot identical and not superimposable. The object and its mirror images are not superimposable with each otherand not align all the parts of the objects with its mirror image. Thus, pair of shoes are chiral.
(c)
Interpretation:
A football should be classified as chiral or achiral.
Concept Introduction:
Chirality is the presence of an asymmetric carbon center in a molecule and a molecule which contains a chiral center cannot superimpose on its mirror image. To consider as chiral, molecule or object and its mirror image should not superimpose. To consider as achiral, molecule or object and its mirror image should be superimposed with each other.
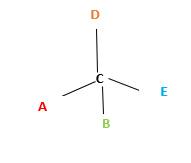
In the above diagram, where C is the chiral center/ asymmetric carbon center.
A, B, D, E are four different functional groups.
Answer to Problem 21P
Football is achiral.
Explanation of Solution
To be considered as chiral, molecule or object and its mirror image should not superimpose. To consider as achiral, molecule or object and its mirror image should be superimposed with each other. The mirror image of a football is identical and superimposable. The object and its mirror images are superimposable with each other. Thus, football is achiral.
Want to see more full solutions like this?
Chapter 15 Solutions
CONNECT IA GENERAL ORGANIC&BIO CHEMISTRY
- 1. Is it possible to distinguish between two congruent functional groups in a chiral environment? If so, what reactions or spectroscopic methods could be used? FG = functional group.arrow_forwardWhich of the following compounds has a plane of symmetry? & & I OI and II OII and III O II, III and IV O III and IV Question 16 O2, 3-dibromobutane 1,3-dibromobutane Which of the following compounds is never chiral? 1, 2-dichlorobutane || 1,4-dibromobutane 'CI 1-bromo-2-chlorobutane III & IVarrow_forwardDraw a structural formula of the SS configuration of the compound shown below. S OH S CH3 CH₂CH3 • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Include H atoms at chiral centers only. • If a group is achiral, do not use wedged or hashed bonds on it.arrow_forward
- Draw a structural formula of the SR configuration of the compound shown below. HO CH3 S -NHCH3 R H3C ephedrine • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Include H atoms at chiral centers only. • If a group is achiral, do not use wedged or hashed bonds on it. * #[ ] در ?arrow_forwardDraw a structural formula of the S configuration of the compound shown below. • Use the wedge /hash bond tools to indicate stereochemistry where it exists. Include H atoms at chiral centers only. • Ifa group is achiral, do not use wedged or hashed bonds on it. CH3 CH3 CH;CHCHCN CH,CH,CH,CHCH,CH, CH2NH2 Draw a structural formula of the RS configuration of the compound shown below. Use the wedge /hash bond tools to indicate stereochemistry where it exists. • Include H atoms at chiral centers only. If a group is achiral, do not use wedged or hashed bonds on it. ÇIarrow_forwardDraw a structural formula of the R configuration of the compound shown below. ÇNH₂ CN • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Include H atoms at chiral centers only. • If a group is achiral, do not use wedged or hashed bonds on it. #[ ] در ?arrow_forward
- Draw a structural formula of the RS configuration of the compound shown below. OH R U CH 8 _CH_ SI ephedrine [*** NH-CH3 CH3 • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Include H atoms at chiral centers only. • If a group is achiral, do not use wedged or hashed bonds on it. ▾ Sn [F ?arrow_forwardExamine the reaction below, and answer the question in the box. If the reaction is possible, draw the simplest possible mechanism for it, showing all lone pairs. You may need to expand parts of the structures to show some lone pairs. If the reaction is not possible, leave the drawing space blank. Is this reaction possible? You can assume that all reactants and products are shown. H OH + O yes O noarrow_forward4. Identify the chiral centers in the following molecule and label each as R or S. Ноarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
