
Consider the gas-phase reaction between A2 (green) and B2 (red) to form AB at 298 K:
(a) Which of the following reaction mixtures is at equilibrium?
(b) Which of the following reaction mixtures has a negative ΔG value?
(c) Which of the following reaction mixtures has a positive ΔG value?
The partial pressures of the gases in each frame are equal to the number of A2, B2, and AB molecules times 0.10 atm. Round your results to two significant figures.
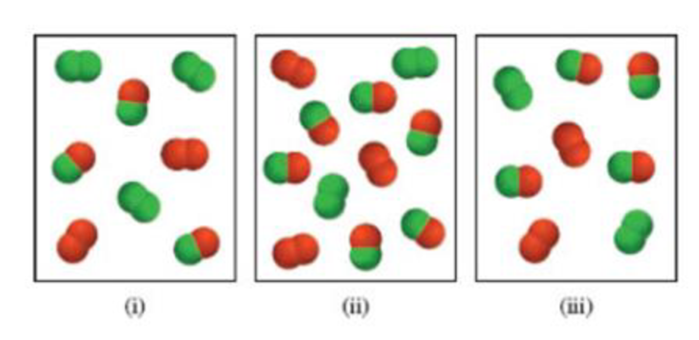
(a)
Interpretation:
Calculate the free energy values
Concept Introduction:
Chemical equilibrium: The term applied to reversible chemical reactions. It is the point at which the rate of the forward reaction is equal to the rate of the reverse reaction. The equilibrium is achieved; the concentrations of reactant and products become constant.
Homogeneous equilibrium: A homogeneous equilibrium involved has a everything present in the same phase and same conditions, for example reactions where everything is a gas, or everything is present in the same solution.
Entropy
Gibbs free energy (G): The thermodynamic quantity to the (
To find: Calculate the pressure values
Answer to Problem 15.104QP
The system equilibrium reaction on given respective images (a-c) are shown below.
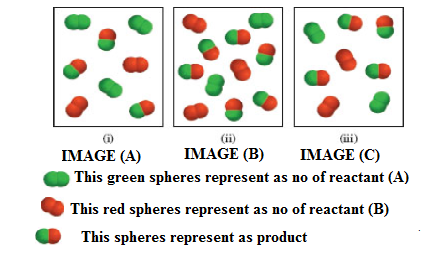
The given equilibrium reaction (Image (A) as positive and Images (ii and iii) has negative
Explanation of Solution
Calculate the chemical equilibrium process
Consider the following equation (1)
The equilibrium constant is related to the to the standard free energy change by the followed above equation (1). Than the given statement of values (KP, R and T) are substituted in same equation.
The standard free energy values and partial pressure values are derived given the equilibrium reactions.
(b)
Interpretation:
Calculate the free energy values
Concept Introduction:
Chemical equilibrium: The term applied to reversible chemical reactions. It is the point at which the rate of the forward reaction is equal to the rate of the reverse reaction. The equilibrium is achieved; the concentrations of reactant and products become constant.
Homogeneous equilibrium: A homogeneous equilibrium involved has a everything present in the same phase and same conditions, for example reactions where everything is a gas, or everything is present in the same solution.
Entropy
Gibbs free energy (G): The thermodynamic quantity to the (
To find: Calculate the reaction quotient (Qr) values for given the equilibrium reaction.
Answer to Problem 15.104QP
The system equilibrium reaction on given respective images (a-c) are shown below.

The given equilibrium reaction (Image (A) as positive and Images (ii and iii) has negative
Explanation of Solution
Calculate and analyze the (Qr) values for equilibrium reactions.
Analysis for image (1): The equilibrium process (a) also smallest equilibrium constant, here three products are present in the diagrams.
Analyzing image (2): Further we consider the equilibrium images (2), this process has the smallest equilibrium constant (Kc), because low amount of reactant present in this equilibrium.
Analyzing image (3): Given the equilibrium reaction (3) has largest equilibrium constant, because the three diagrams, there is the most products present at equilibrium process, and this products indicated the respective image.
(c)
Interpretation:
Calculate the free energy values
Concept Introduction:
Chemical equilibrium: The term applied to reversible chemical reactions. It is the point at which the rate of the forward reaction is equal to the rate of the reverse reaction. The equilibrium is achieved; the concentrations of reactant and products become constant.
Homogeneous equilibrium: A homogeneous equilibrium involved has a everything present in the same phase and same conditions, for example reactions where everything is a gas, or everything is present in the same solution.
Entropy
Gibbs free energy (G): The thermodynamic quantity to the (
To find: Calculate the entropy values
Calculate the chemical equilibrium process
Answer to Problem 15.104QP
The system equilibrium reaction on given respective images (a-c) are shown below.

The given equilibrium reaction (Image (A) as positive and Images (ii and iii) has negative
Explanation of Solution
Let us consider the following equation (1)
The entropy values of given equilibrium reaction (1) has negative
Want to see more full solutions like this?
Chapter 15 Solutions
CHEMISTRY:ATOMS FIRST-2 YEAR CONNECT
- Consider a metal ion A2+ and its nitrate salt, In an experiment, 35.00 mL of a 0.217 M solution of A(NO3)2 is made to react with 25.00 mL of 0.195 M NaOH. A precipitate, A(OH)2, forms. Along with the precipitation, the temperature increases from 24.8C to 28.2C. What is H for the precipitation of A(OH)2? The following assumptions can be made. • The density of the solution is 1.00 g/mL. • Volumes are additive. • The specific heat of the solution is 4.18 J/g C.arrow_forwardUse the data in Appendix J to calculate rG andKPat 25 C for the reaction 2HBr(g)+Cl2(g)2HCl(g)+Br2() Comment on the connection between the sign of rG and the magnitude ofKP.arrow_forwardA process that is reactant-favored at equilibrium can never be spontaneous. This statement is (a) true (b) falsearrow_forward
- Elemental boron, in the form of thin fibers, can be made by reducing a boron halide with H2. BCl3(g) + 3/2 H2(g) B(s) + 3HCl(g) Calculate H, S, and G at 25 C for this reaction. Is the reaction predicted to be product favored at equilibrium at 25 C? If so, is it enthalpy driven or entropy driven?arrow_forwardAdenosine triphosphate, ATP, is used as a free-energy source by biological cells. (See the essay on page 624.) ATP hydrolyzes in the presence of enzymes to give ADP: ATP(aq)+H2O(l)ADP(aq)+H2PO4(aq);G=30.5kJ/molat25C Consider a hypothetical biochemical reaction of molecule A to give molecule B: A(aq)B(aq);G=+15.0kJ/molat25C Calculate the ratio [B]/[A] at 25C at equilibrium. Now consider this reaction coupled to the reaction for the hydrolysis of ATP: A(aq)+ATP(aq)+H2O(l)B(aq)+ADP(aq)+H2PO4(aq) If a cell maintains a high ratio of ATP to ADP and H2PO4 by continuously making ATP, the conversion of A to B can be made highly spontaneous. A characteristic value of this ratio is [ATP][ADP][H2PO4]=500 Calculate the ratio [B][A] in this case and compare it with the uncoupled reaction. Compared with the uncoupled reaction, how much larger is this ratio when coupled to the hydrolysis of ATP?arrow_forwardHydrogenation, the addition of hydrogen to an organic compound, is an industrially important reaction. Calculate rH, rS, and rG for the hydrogenation of octene, C8H16, to give octane, C8H19 at 25 C. Is the reaction product- or reactant-favored at equilibrium? C8H16(g) + H2(g) C8H18(g) Along with data in Appendix L, the following information is needed for this calculation.arrow_forward
- Predict whether each of the following processes results in an increase in entropy in the system. (Define reactants and products as the system.) (a) Water vapor condenses to liquid water at 90 C and 1 atm pressure. (b) The exothermic reaction of Na(s) and Cl2(g) forms NaCl(s). (c) The endothermic reaction of H2 and I2 produces an equilibrium mixture of H2(g), I2(g), and HI(g). (d) Solid NaCl dissolves in water forming a saturated solution.arrow_forwardA crucial reaction for the production of synthetic fuels is the production of H2 by the reaction of coal with steam. The chemical reaction is C(s) + H2O(g) CO(g) + H2(g) (a) Calculate rG for this reaction at 25 C, assuming C(s) is graphite. (b) Calculate Kp for the reaction at 25 C. (c) Is the reaction predicted to be product-favored at equilibrium at 25 C? If not, at what temperature will it become so?arrow_forwardConsider the reaction 2SO2(g)+O2(g)2SO3(g) (a) Calculate G at 25C. (b) If the partial pressures of SO2 and SO3 are kept at 0.400 atm, what partial pressure should O2 have so that the reaction just becomes nonspontaneous (i.e., G=+1.0 k J)?arrow_forward
- Describe a nonchemical system that is not in equilibrium, and explain why equilibrium has not been achieved.arrow_forwardFor each reaction, an equilibrium constant at 298 K is given. Calculate G for each reaction. (a) Br2()+ H2(g)2HBr(g) KP = 4.4 1018 (b) H2O()H2O(g) KP = 3.17 102 (c) N2(g) +3H2(g)2NH3(g) Kc = 3.5 108arrow_forward12.108 A nuclear engineer is considering the effect of discharging waste heat from a power plant into a lake and estimates that this may warm the water locally to 25 °C. One question to be considered is the effect of this temperature change on the uptake of CO2 by the water. The equilibrium constant for the reaction CO2+H2OH2CO3 ; is K=1.7103 at 25 °C. Because bonds form, the reaction is exothermic. (a) Will this reaction progress further toward products at higher temperatures near the water discharge with its warmer water than it would in the cooler lake water? Explain your reasoning. (b) Carbonic acid has a Kaof 2.5104 at 25 °C. What is the equilibrium constant for the CO2+2H2OHCO3+H3O+? (c) What additional factor should the engineer be considering about CO2 gas, probably before considering this reaction chemistry?arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





