
Concept explainers
Interpretation:
The structures of all possible adducts obtainable when the diene given reacts with one equivalent of HCl are to be given.
Concept introduction:
Conjugated dienes undergo electrophilic addition reactions through the formation of an allyl carbocation. The allyl cation is resonance stabilized and the attack of chloride ion on each of these forms leads to the formation of a mixture of 1, 2- and 1, 4-addition products.
To give:
The structures of all possible adducts obtainable when the diene given reacts with one equivalent of HCl.
Answer to Problem 16VC
The compound given is 1-vinylcyclopentene.The structures of all the possible adducts obtained when it reacts with one equivalent of HCl are given below.
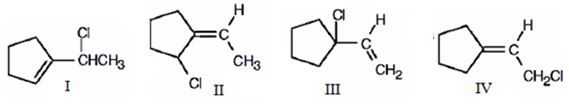
Explanation of Solution
1-Vinylcyclopentene is an unsymmetrical conjugated diene. Protonation of the CH2 in double bond and that in the ring lead to the formation of two different allyl carbocations, each of which is in resonance with another form. Hence four carbocations as shown are possible. Attack of the chloride ion on each of these carbocation leads to the formation of four different products.
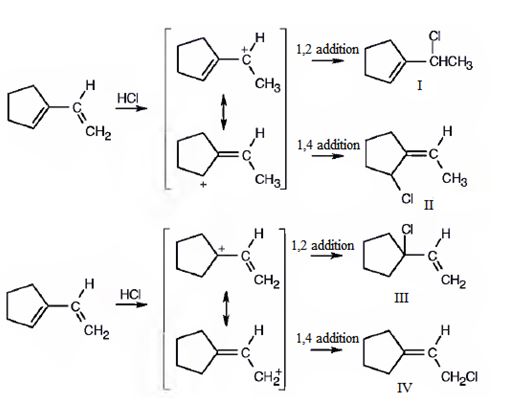
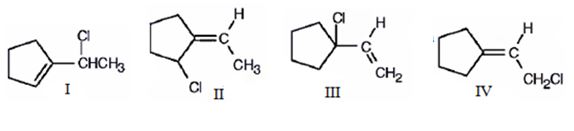
The compound given is 1-vinylcyclopentene.The structures of all the possible adducts obtained, when it reacts with one equivalent of HCl, are given below.
Want to see more full solutions like this?
Chapter 14 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- Show the product of the Diels–Alder reaction of the following diene with 3-buten-2-one, H2C = CHCOCH3. Make sure you show the full stereochemistry of the reaction product.arrow_forwardUnder the reaction of 2-methyl-1,3-pentadiene with a single equivalent of HBr, as shown below:arrow_forwardPlease answer both parts to the questions completely. I will rate the answer afterwards. For a diels-alder reaction between anthracene and maleic anhydride, are the exo and endo forms of product 9,10-dihydroanthracene-9,10-ɑ,β-succinic acid anhydride different stereoisomers or are they the same molecule? Explain your answer by drawing the molecule. Would it be favorable to get a 1,4-adduct of anthracene and maleic anhydride? Why or why not? If the 1,4-adduct of anthracene and maleic anhydride had formed, would it have different exo and endo isomers? Yes or no and why?arrow_forward
- If you were asked to convert 2-iodohexane to 1-hexene, which reagent would you choose? Select one: O a. (CH3);COK, (CH3);COH O b. CH3ONA, CH3OH ос. кон, СНзОн O d. NaOH, H20 Which of these is a correct representation of (S)-3-bromo-1,1-dichloropropan-2-ol? Select one: O a. Cl OH -Br Ob. он H+CHC, H Cl „CI HO CH,Br CH,Br HO-H ČHCI, d.arrow_forwardWhen 2-bromobutane is reacted with CH3O-, two alkene products, namely 2-butene and 1-butene are obtained. Explain why the E2 reaction produces 2-butene as the major product (80%) and 1-butene as the minor product (20%)arrow_forwardConsider the following compound. Part 1 of 2 H Draw the structure of the diene used to prepare the given compound. Show relevant stereochemistry when applicable.arrow_forward
- Draw structural formulas for the diene and dienophile that combine in a Diels-Alder reaction to form the product shown. CN Diene + Dienophile CH₂O • Consider E/Z stereochemistry of alkenes. CNarrow_forwardFor a diels-alder reaction between anthracene and maleic anhydride, are the exo and endo forms of product 9,10-dihydroanthracene-9,10-ɑ,β-succinic acid anhydride different stereoisomers or are they the same molecule?arrow_forwardDraw structural formulas for the diene and dienophile that combine in a Diels-Alder reaction to form the product shown. Diene + Dienophile OHarrow_forward
- What represents the structure of the diene to carry out the following Diels-Alder reaction?arrow_forwardShow the structure of the product from the Diels-Alder reaction between 2 moles of isoprene (2-methyl-1,3-butadiene) and one mole of quinone.arrow_forwardDraw the reactants that would be used to form this cyclohexene derivative in a Diels- Alder reaction. Include any relevant stereochemical configurations. Draw Diene + ·00 aarrow_forward
