
CHEMISTRY: AN INTRODUCTION TO GENERAL, O
13th Edition
ISBN: 9780137444298
Author: Timberlake
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14.6, Problem 14.48PP
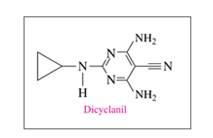
a. Identify the
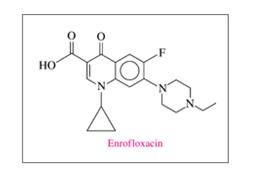
b. The recommended dose for enrofloxacin for sheep is 30. mg/kg of body
mass for 5 days. Enrofloxacin is supplied in 50.-mL vials with a
concentration of 100. mg/mL. How many vials are needed to treat a 64-
kg sheep for 5 days?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1. The Ability of a substance to exist in different crystalline form
a. Lattice
b. Polymorphism
c. Crystallization
d. Amphoterism
2. A drug can exert its pharmacological effect only if it is
a. Protein bound
b. Protein unbound
c. Free drug
d. Both B & C
e. Both A & C
3. In order for the drug to be ready and available for absorption, it must be release first from its dosage form with the exception of:
a. Capsule
b. Tablet
c. Solution
d. Suspension
4. All of the following are true, except
a. Solubility increase with decrease particle size
b. Solubility increase with increase surface area
c. Solubility increase with increase particle size
d. Solubility decrease with decrease surface area
5. The rate in which the drug appears in the bloodstream is also known as
a. Half-life
b. Potency
c. Bioavailability
d. Area under the curve
1. An inactive or much less active form of drug which transformed to active drug in the body is
a. Coated drug
b. Specialized dosage form
c. Prodrug
d. Reservoir
2. Sodium pump is an example of
a. G-protein receptors
b. Ion channels
c. Gene Transcription linked receptor
d. Enzyme-linked tyrosine kinase3. As the length of a non-polar chain of aliphatic alcohol increases, its solubility in water
a. Increases
b. Decreases
c. No effect
4. More common force of attraction for drugs
a. Covalent
b. Electrostatic
c. Hydrogen bonding
d. Ionic5. In organs and tissues that are well-perfuse, drug distribution is
a. Faster
b. Slower
c. Negligible
d. The same
3. Lipids
a. What are saturated fats?
b. What is the main origin of triacylglycerides or
oils?
O
c. Why do unsaturated oils discolor bromine in
solution?
d. What functional groups are present in
triacylglycerides?
and. Why are lipids not soluble in water when they
have a polar part?
F. What positive function does cholesterol have in
the body?
pility: Good to go
D
(Ctrl)
Chapter 14 Solutions
CHEMISTRY: AN INTRODUCTION TO GENERAL, O
Ch. 14.1 - What carboxylic acid is responsible for the pain...Ch. 14.1 - What carboxylic acid is found in vinegar?Ch. 14.1 - Prob. 14.3PPCh. 14.1 - Prob. 14.4PPCh. 14.1 - Draw the condensed structural formulas for a and b...Ch. 14.1 - Draw the condensed structural formulas for a and b...Ch. 14.2 - Identify the compound in each group that is most...Ch. 14.2 - Prob. 14.8PPCh. 14.2 - Prob. 14.9PPCh. 14.2 - Prob. 14.10PP
Ch. 14.2 - Prob. 14.11PPCh. 14.2 - Prob. 14.12PPCh. 14.2 - Prob. 14.13PPCh. 14.2 - Prob. 14.14PPCh. 14.3 - Prob. 14.15PPCh. 14.3 - Prob. 14.16PPCh. 14.3 - Prob. 14.17PPCh. 14.3 - Prob. 14.18PPCh. 14.3 - Prob. 14.19PPCh. 14.3 - Prob. 14.20PPCh. 14.3 - Prob. 14.21PPCh. 14.3 - Prob. 14.22PPCh. 14.3 - Prob. 14.23PPCh. 14.3 - Prob. 14.24PPCh. 14.4 - What are the products of the acid hydrolysis of an...Ch. 14.4 - Prob. 14.26PPCh. 14.4 - Prob. 14.27PPCh. 14.4 - Prob. 14.28PPCh. 14.5 - Prob. 14.29PPCh. 14.5 - Prob. 14.30PPCh. 14.5 - Prob. 14.31PPCh. 14.5 - Prob. 14.32PPCh. 14.5 - Prob. 14.33PPCh. 14.5 - Prob. 14.34PPCh. 14.5 - Prob. 14.35PPCh. 14.5 - Prob. 14.36PPCh. 14.5 - Prob. 14.37PPCh. 14.5 - Prob. 14.38PPCh. 14.6 - Prob. 14.39PPCh. 14.6 - Prob. 14.40PPCh. 14.6 - Prob. 14.41PPCh. 14.6 - Prob. 14.42PPCh. 14.6 - Prob. 14.43PPCh. 14.6 - Prob. 14.44PPCh. 14.6 - Draw the condensed structural or line-angle...Ch. 14.6 - Draw the condensed structural or line-angle...Ch. 14.6 - a. Identify the functional groups in dicyclanil....Ch. 14.6 - a. Identify the functional groups in enrofloxacin....Ch. 14 - Prob. 14.49UTCCh. 14 - Prob. 14.50UTCCh. 14 - The ester methyl butanoate has the odor and flavor...Ch. 14 - Prob. 14.52UTCCh. 14 - Phenylephrine is the active ingredient in some...Ch. 14 - Melatonin is a naturally occurring compound in...Ch. 14 - Prob. 14.55UTCCh. 14 - Prob. 14.56UTCCh. 14 - Prob. 14.57APPCh. 14 - 14.58 Write the IUPAC and common names, if any,...Ch. 14 - Prob. 14.59APPCh. 14 - Prob. 14.60APPCh. 14 - Draw the condensed structural or line-angle...Ch. 14 - Prob. 14.62APPCh. 14 - Prob. 14.63APPCh. 14 - 14.64 Draw the condensed structural or line-angle...Ch. 14 - Prob. 14.65APPCh. 14 - 14.66 Write the common name and classify each of...Ch. 14 - Prob. 14.67APPCh. 14 - Draw the condensed structural or line-angle...Ch. 14 - Prob. 14.69APPCh. 14 - Prob. 14.70APPCh. 14 - Write the IUPAC name for each of the following:...Ch. 14 - Prob. 14.72APPCh. 14 - Prob. 14.73APPCh. 14 - Prob. 14.74APPCh. 14 - Prob. 14.75APPCh. 14 - Toradol is used in dentistry to relieve pain....Ch. 14 - Prob. 14.77CPCh. 14 - Draw the line-angle formula and write the IUPAC...Ch. 14 - Prob. 14.79CPCh. 14 - Prob. 14.80CPCh. 14 - Prob. 14.81CPCh. 14 - Prob. 14.82CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Biological waxes have a head and two tails structure. Give the chemical identity of the head and of the two tails.arrow_forward7. Why the need for a homogenizer? 8. Why the use of a water bath instead of a stove? 9. Why should heating be at approximately 70 to 75 deg. C.? 10. Aside from its skin moisturizing ability, for what other purposes are creams and lotions used?arrow_forward1. Describe the uses of astringents in a. Medicine b. Food 2. Name a. at least 5 herbal extracts that are often the main ingredients in astringents and toners b. the benefits or functions of each extract c. the commercial astringent or toner that uses the extractarrow_forward
- Which compounds are water soluble? a. LiCI water soluble b. C-Hs (select) c. NazPO4 (select)arrow_forward1. What impurity is most likely to be present in the sample of aspirin you prepared? What effect would this impurity have on the body if the product were intended for human consumption?arrow_forwardI. Name 5 potentially toxic pigments or dyes and explain why each substance is considered harmful when used in either food or cosmetics. II. Name 5 potentially toxic ingredients in hair dyes and explain why each substance is considered harmful to human scalp/skin.arrow_forward
- 2. What properties of waxes permit them to be effective coatings to prevent ducklings from getting their feathers wet?arrow_forwardThe LD50 (“Lethal Dose, 50 %”) is a value used in toxicology which gives the amount of a substance required to kill 50 % of a test population. It is usually reported based on body weight. For racemic carvone LD50 is reported as 1640 mg/kg. 1. Would it consider carvone to be toxic to an average person? Explain. 2. Would it be expected this the LD50 value to be the same for each enantiomer? Explain at the molecular level in stereochemical terms.arrow_forward2. Soap can be made using a variety of different fats, oils and bases. Suppose a student wanted to make a soft, liquid soap that lathers easily. What type(s) of fat/oil and base would you recommend? How would this differ from making a solid bar of soap? 3. Vegetable oils are typically liquid at room temperature, yet Crisco (which is made of vegetable oil) is solid at room temperature. Explain why/how this is the case. 4. Explain, with the use of a diagram how soap molecules work to clean “dirt” particles. 5. Amylase is a natural enzyme found in saliva. Describe this enzyme’s function. 6. Lactose is a sugar found in milk and other dairy products. Explain how it is broken down in most individuals. Explain lactose “intolerance”.arrow_forward
- 271. Indicate the characteristic for Ononin. I. biflavonoid II. isoflavonoid III. dimer structure IV. glucose monosaccharide is coupled by C7 atom V. taken from Ononis arvensis VI. aglycone VII. flavanon derivate VIII. taken from Viola arvensis IX. aglycone part is formonetin X. aglycone part is onogenin A) II; V; VII; VII B) II; IV; V; IX C) I; III; VI; X D) I; IV; V; VIII; IX E) III; IV; VI; VII; Xarrow_forward28. A structural feature with in a molecule which is responsible for chirality is called c. chirality d. Chiral C e. Chiral center d. all 29. Optically active compounds have isomer a Two C. four b. Three d. five 30. Propanone and propenol are a. structural isomers C. tautomer b. metamers d position isomerarrow_forward67. This compound is called Steroid Glycerin Waxarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY