
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 14.4, Problem 5P
How many alkyl chlorides can be obtained from monochlorination of the following


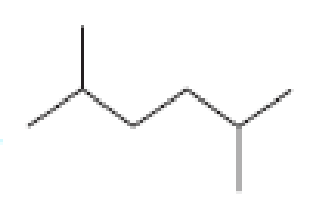
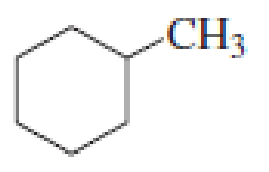
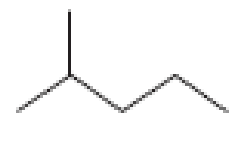
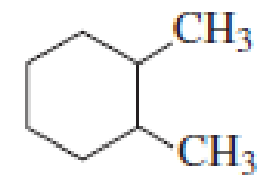
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
How many alkyl chlorides are obtained from monochlorination of the following alkanes? Disregard stereoisomers.
Explain the role strain plays on stability and reactivity of a cyclic alkane such as cyclopropane.
How many monochloro substitution products are produced when the alkanes below are chlorinated?
Chapter 14 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 14.2 - Write the mechanism for the monobromination of...Ch. 14.2 - Prob. 2PCh. 14.4 - Prob. 3PCh. 14.4 - Which of the hydrogens in the structure in the...Ch. 14.4 - How many alkyl chlorides can be obtained from...Ch. 14.4 - Prob. 7PCh. 14.5 - Prob. 9PCh. 14.6 - a. Which ether is most apt to form a peroxide? b....Ch. 14.7 - Prob. 11PCh. 14 - Prob. 12P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- How many monochloro substitution products are produced when the alkanes below are chlorinated? Consider constitutional isomers only, ignore stereoisomers. a) The number of monochloro substitution products isarrow_forwardHow many monochloro substitution products are produced when the alkanes below are chlorinated? Consider constitutional isomers only, ignore stereoisomers. a) b) The number of monochloro substitution products is 4 The number of monochloro substitution products is 3 X.arrow_forwardDraw all of the constitutional (structural) isomers that can be formed from the monochlorination of the hydrocarbon shown below. CH3 CH3 hv CH3 Cl2 + H3C- CH3 エーO-エarrow_forward
- Alkyl halides can be converted into alkanes by which reaction?arrow_forward2-chloropropane is a major product of the reaction of chlorine with propane under ultraviolet light. Write the mechanism for this reaction including the initiation step and the two propagation steps.arrow_forwardHow many monochloro substitution products are produced when the alkane below is chlorinated? Consider both constitutional isomers and stereoisomers. X The number of monochloro substitution products isarrow_forward
- 10. The relative rates of epoxidation reactions of the following four alkenes are 1, 22, 484, 6526. Which alkene does feature the relative epoxidation rate of 484? 11. Alkynes undergo addition reaction with HCl, forming corresponding alkenyl bromide. Please write the rate law of such a reaction between butyne and HCl.arrow_forwardThe reaction of 3-methylene-1-cyclohexene and HBr yields the four products shown in the attachment. Which two are formed at high temperatures and which two are formed at low temperatures? Why? Why is 1-bromo-3-methylenecyclohexane not formed?arrow_forwardWhat are the major and minor products formed when cyclohexene reacts with Br2 in the solvent CH2Cl2?arrow_forward
- How many monochlorination products are possible for the alkane shown below?arrow_forwardThe reaction of Hbr with 2-methylpropene produces 2-bromo-2-methylpropane. What is the structure of the carbocation formed during the reaction?arrow_forwardThe major product formed when methylenecyclohexane is treated with NBS in dichloromethane is 1-(bromomethyl)cyclohexene. Account for the formation of this product.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
How to Design a Total Synthesis; Author: Chemistry Unleashed;https://www.youtube.com/watch?v=9jRfAJJO7mM;License: Standard YouTube License, CC-BY