
Concept explainers
The chapter sections to review are shown in parentheses at the end of each problem.
14.87 Determine if each of the following diagrams represents a strong acid or a weak acid. The acid has the formula HX. (14.3)
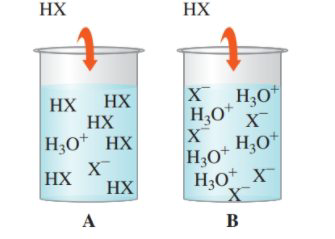
Interpretation:
To identify the strong and weak acids, given the diagrams with the ion species.


Concept introduction:
- Weak acids will not dissociate completely
- Strong acid will dissociate 100% in solution.
To determine: The weak and strong acids
Answer to Problem 87UTC
Solution: weak acid is A; strong acid is B
Explanation of Solution
An acid is said to be a strong acid if it will be 100% dissociated into ions in the solution. The acid will donate all of its acidic protons, and therefore, will have a higher hydronium ion concentration in solution after dissolution. The pH is likely to be lower.
An acid is said to be a weak acid if it will NOT be 100% dissociated into ions in the solution. The acid will not donate all its acidic protons, and therefore, will form equilibrium between ions and acid. The pH is likely to be higher.
Since, the number of ions (
Weak acids and strong acids can be identified if the dissociation factor is given.
Want to see more full solutions like this?
Chapter 14 Solutions
EP BASIC CHEMISTRY-STANDALONE ACCESS
- There is a spillage of concentrated hydrochloric acid (HC ) into the only water storage tank at the School. The acidity of a sample of water is tested and the pH is found to be 3, 5. 7.2.1 State which ions (OH or H,O") were in excess in the sample 7.2.2 Calculate the concentration of H,O' ions in the samplearrow_forwardWhat is the pH of a solution of a 0.045 M of a strong acid? (1.35)arrow_forward(4.1 201 6. In each of the following chemical equations, identify the conjugate acid-base pairs. a. CH3NH2 + H₂O CH3NH3+ + OH- b. CH3COOH + NH3 CH3COO + NH4+ of vroud marrow_forward
- 9.16 Complete and balance the following neutralization reactions: a. HNO3(aq) + Mg(OH)2 (s) b. HCl(aq) + NaHCO3(s) →→→→ -arrow_forwardWrite the equilibrium constant expression, Kc, for the following reactions. Indicate also if the equilibrium is homogeneous or heterogeneous. (ONLY 1 AND 2) COMPLETE SOLUTIONarrow_forwardwi What is the pH of abinffor which has been prepared y mi ing 25 , l sinel 200 ml, oiM N Solution? by 250 M Váz solution ? what is the pH of this after 10 mmul Hl has been udded ? Aas. (4.88, 9.09)arrow_forward
- 3 6. 7 10 11 12 13 14 For each chemical reaction in the table below, decide whether the highlighted reactant is a Brønsted-Lowry acid, a Brønsted-Lowry base, or neither. highlighted reactant reaction Bronsted-Lowry Bronsted-Lowry neither acid base NO3(aq) + H,O (aq) → HNO3(aq) + H,0(1) HNO3(aq) + H,O) NO3(aq) + H,0 (aq) HNO3(aq) + H,O() → NO3(aq) + H,o (aq) NO3(aq) + H,O (aq) → HNO3(aq) + H,O(1) Submit Assignment Continue MacBook Air F12 FI F9 FIC D00 FB F7 吕0 F3 D00 F4 F5 escarrow_forward1- What is the p H of a 2.31 M solution of HA that has a K a of 1.81E-5? Answer: (2.19) 2- Calculate the percentage that allows for the deliberation of whether or not 0.05 - [H+] ~ 0.05 in accordance with 5% rule. 0.05 M solution of HA with a K a = 7.70E-7 Answer to 2 decimal places (even though this might violate significant figure rules). Answer: (0.39)arrow_forward9.48 Calculate the [H3O+] for each of the following mea- surements of pH: a. hand soap, pH 9.5 b. sea water, pH 8.2 c. apple juice, pH 3.5 d. beer, pH 4.5 volentes indileparrow_forward
- 5.4 Write the formulas of the conjugate bases of the following acids: (a) HNO2, (b) H2SO4, (c) H,S, (d) HCN, (e) HCOOH (formic acid). 15.3arrow_forwardDr. Pepper has an [H+] = 0.000014 M. What is its pH? (4.85)arrow_forward5. 2.65 gram potassium hydroxide was dissolved in water to make a 750.0 ml stock solution. 20.0 ml of this stock solution was diluted with water to make a 1.000 L working solution. i) What is the concentration of OH in the final working solution after dilution. ii) What is the concentration of H3O+ in the final working solution? iii) What is the pH of the final working solution? Jtryarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





