
The bromination of acetone is acid-catalyzed:
The rate of disappearance of bromine was measured for several different concentrations of acetone, bromine, and
|
|
|
Rate of Disappearance of
|
|
(1) |
0.30 |
0.050 |
0.050 |
|
(2) |
0.30 |
0.10 |
0.050 |
|
(3) |
0.30 |
0.050 |
0.10 |
|
(4) |
0.40 |
0.050 |
0.20 |
|
(5) |
0.40 |
0.050 |
0.050 |
|
(a) What is the rate law for the reaction? (b) Determine the rate constant. (c) The following mechanism has been proposed for the reaction:
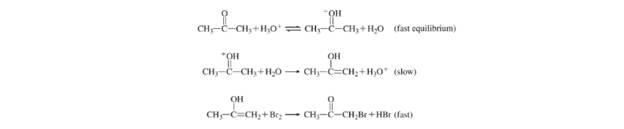
Show that the rate law deduced from the mechanism is consistent with that shown in part (a).
Interpretation:
The rate law and rate constant for the reaction are to be determined. The rate law is consistent with the mechanism shown in part (a), is to be determined.
Concept introduction:
The rate of reaction in terms of concentration is called the rate law.
The step-by-step reaction is called the reaction mechanism. The reaction that takes place in a single step is called an elementary reaction.
Answer to Problem 85AP
Solution:
(a)
(b)
(c) The rate law deduced from the mechanism is consistent.
Explanation of Solution
a)The rate law for the reaction
For the determination of rate law, calculate the exponents as follows:
Based on
Based on
Based on
Based on
Hence, the rate law for the reaction is:
b)The rate constant
According to rate law, the rate constant for the reaction is:
Rearrange the above equation for
Here,
From experiment 1,
The rate law is
The concentration of
Substitute,
The rate constant for the reaction is
c) The rate law deduced from the given following mechanism is consistent or not with
The proposed mechanism for the reaction is:
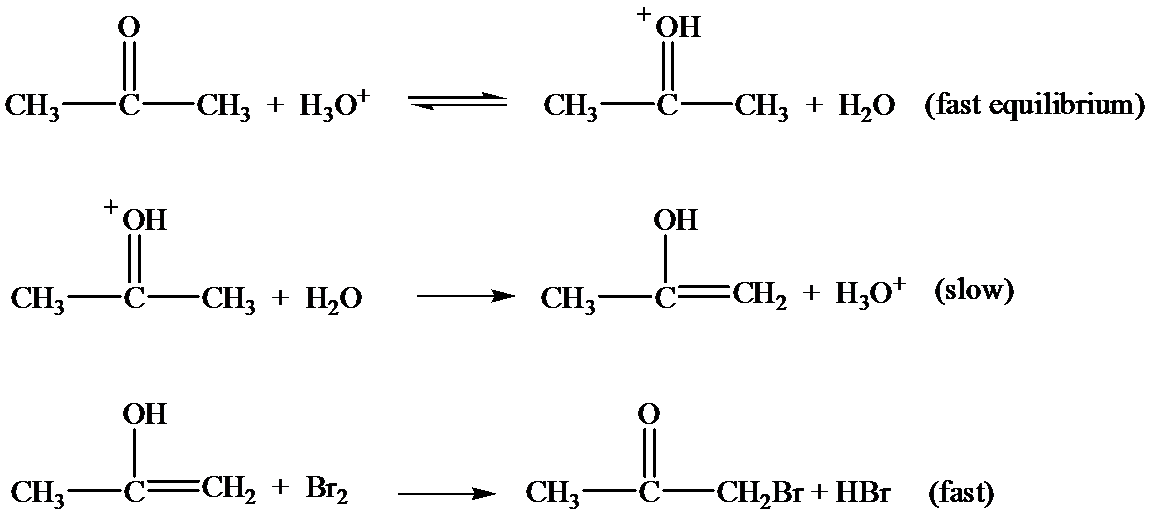
Let the rate constant for the slow step be
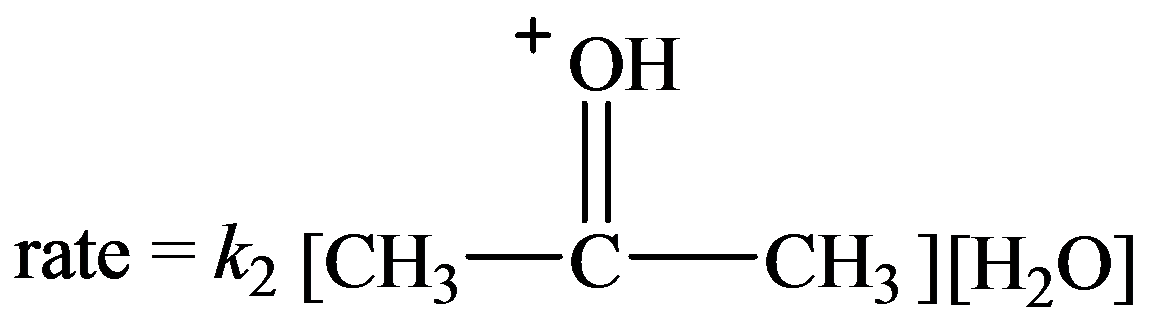 …… (1)
…… (1)
The rate constants for the forward and reverse steps in the fast equilibrium are
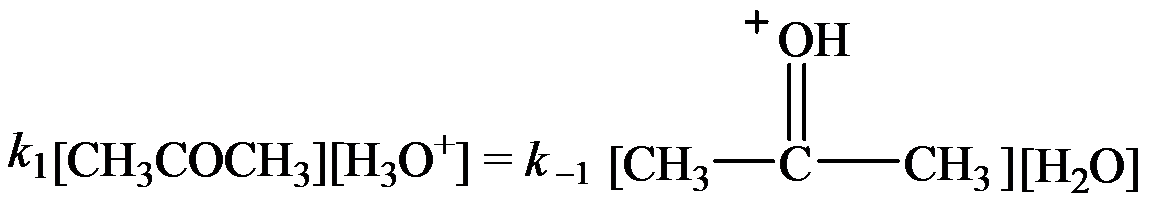 …… (2)
…… (2)
So, equation (1) becomes
The overall equation from part (a) is:
From equation (3) and (4),
So, the rate law is consistent with the mechanism shown in part (a).
Want to see more full solutions like this?
Chapter 14 Solutions
Chemistry
- 11.41 For a drug to be effective in treating an illness, its levels in the bloodstream must be maintained for a period of time. One way to measure the level of a drug in the body is to measure its rate of appearance in the urine. The rate of excretion of penicillin is first order, with a half-life of about 30 min. If a person receives an injection of 25 mg of penicillin at t = 0, how much penicillin remains in the body after 3 hours?arrow_forwardFor the reaction 4NO2(g)+O2(g)2N2O5(g) (a) express the rate of formation of N2O5 in terms of the rate of disappearance of O2. (b) suppose the rate of disappearance of O2 is 0.0037 mol L1 s1. Calculate the rate of disappearance of NO2.arrow_forwardCandle wax is a mixture of hydrocarbons. In the reaction of oxygen with candle w ax in Figure 11.2, the rate of consumption of oxygen decreased with time after the flask was covered, and eventually' the flame went out. From the perspective of the kinetic-molecular theory, describe what is happening in the flask. FIGURE 11.2 When a candle burns in a closed container, the flame will diminish and eventually go out. As the amount of oxygen present decreases, the rate of combustion will also decrease. Eventually, the rate of combustion is no longer sufficient to sustain the flame even though there is still some oxygen present in the vessel.arrow_forward
- Express the rate of the reaction 2N2O(g)2N2(g)+O2(g) in terms of (b) [ N2O ] (a) [ O2 ]arrow_forwardWhat are the relative rates of appearance or disappearance of each product and reactant in the decomposition of nitrosyl chloride, NOCI? 2 NOC1(g) 2 NO(g) + Cl2(g)arrow_forwardThe rate of the decomposition of hydrogen peroxide, H2O2, depends on the concentration of iodide ion present. The rate of decomposition was measured at constant temperature and pressure for various concentrations of H2O2and of KI. The data appear below. Determine the order of reaction for each substance, write the rate law, and evaluate the rate constant. Rate [H2OJ [Kll (mL min-’) (mol L ’) (mol L ’) 0.090 0.15 0.033 0.178 0.30 0.033 0.184 0.15 0.066arrow_forward
- A possible mechanism for a gas-phase reaction is given below. What is the rate law predicted by this mechanism? NO+Cl2krk2NOCl2(fastequilibrium)NOCl2+NOk22NOCl(slow)arrow_forwardThe isomerization of cyclopropane, C3H6, is believed to occur by the mechanism shown in the following equations: C3H6+C3H5k1C3H6+C3H6(Step1)C3H6k2C2=CHCH3(Step2) Here C3H6 is an excited cyclopropane molecule. At low pressure, Step 1 is much slower than Step 2. Derive the rate law for this mechanism at low pressure. Explain.arrow_forwardOne possible mechanism for the decomposition of nitryl chloride, NO2CI, is What is the overall reaction? What rate law would be derived from this mechanism? What effect does increasing the concentration of the product NO2 have on the reaction rate?arrow_forward
- The label on a bottle of 3% (by volume) hydrogen peroxide, H2O2, purchased at a grocery store, states that the solution should be stored in a cool, dark place. H2O2decomposes slowly over time, and the rate of decomposition increases with an increase in temperature and in the presence of light. However, the rate of decomposition increases dramatically if a small amount of powdered MnO- is added to the solution. The decomposition products are H2O and O2. MnO2 is not consumed in the reaction. Write the equation for the decomposition of H2O2. What role does MnO2 play? In the chemistry lab, a student substituted a chunk of MnO2 for the powdered compound. The reaction rate was not appreciably increased. WTiat is one possible explanation for this observation? Is MnO2 part of the stoichiometry of the decomposition of H2O2?arrow_forwardCan a reaction mechanism ever be proven correct? Can it be proven incorrect?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning





