
EBK A SMALL SCALE APPROACH TO ORGANIC L
4th Edition
ISBN: 9781305446021
Author: Lampman
Publisher: CENGAGE LEARNING - CONSIGNMENT
expand_more
expand_more
format_list_bulleted
Question
Chapter 14, Problem 3Q
Interpretation Introduction
Interpretation:
The fact according to which carvone fits the isoprene rule needs to be explained.
Concept Introduction:
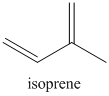
In the isoprene rule, it is explained how isoprene units are joined in a head to tail configuration. The isoprene unit is represented as follows:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Organic chemistry:Discuss the Grignard synthesis of triphenyl methanol and benzoic acid .
3. If both water and your alkene products distill into your collection flask, why might you get two layers?
because there is a temperature gradient in the graduated cyclinder
because methylcyclohexenes and water are immiscible
because methylcyclohexenes and water are miscible
Draw a structural formula of an alkene or alkenes (if more than one) that undergo acid-catalyzed hydration and without rearrangement give 1-methylcyclohexanol as the
major product.
• You do not have to consider stereochemistry.
• You do not have to explicitly draw H atoms.
• If more than one structure fits the description, draw them all.
• Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.
Separate structures with + signs from the drop-down menu.
●
AAVII
-85
?
ChemDoodleⓇ
Y
[F
Chapter 14 Solutions
EBK A SMALL SCALE APPROACH TO ORGANIC L
Knowledge Booster
Similar questions
- Propose a structure for an aromatic hydrocarbon, C3H10, that can form three C3H,Br products on substitution of a hydrogen on the aromatic ring with bromine. • You do not have to consider stereochemistry. In cases where there is more than one answer, just give one. • Ignore the ortho, para-directing effects of the alkyl groups in answering this question. Consider only the number of nonequivalent hydrogens on the aromatic ring. .... ChemDoodlearrow_forwardDraw a structural formula of an alkene or alkenes (if more than one) that undergo acid-catalyzed hydration and without rearrangement give 1-methylcyclohexanol as the major product. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • If more than one structure fits the description, draw them all. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate structures with + signs from the drop-down menu. ● VIL ? ChemDoodle Ⓡ Qt Sn [Farrow_forwardGive the IUPAC name of the product that would form when the following cycloalkene undergoes catalytic hydrogenation. CH3 Name of product Submit Answer = CH3 Retry Entire Group 9 more group attempts remaining Previous Next Save anarrow_forward
- 1) Based on the Material Safety Data Sheets (MSDS) for the compounds, which of the compounds in this experiment is the most hazardous, and why (you have been given a representative aryl halide rather than all of the alkenes – as the products are all new, there’s not a simple answer to gauge their potential hazard)? 2,3-dihydrofuran,2-Mercaptobenzimidazole,Selectfluor, Dimethylformamide,dichloromethane-methylene chloride.arrow_forwardDraw the structural formula of the major product of the reaction of (S)-2,2,3-trimethyloxirane with Me,NH. • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • Include H atoms at chiral centers only. • If a group is achiral, do not use wedged or hashed bonds on it.arrow_forwardDraw structural formulas for all of the enol forms of the carbonyl compound below. You do not have to consider stereochemistry. • Consider stereoisomeric enols as a single enol form. • In the case of dicarbonyl compounds, do not include stuctures in which both carbonyl groups are present as enols. Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate structures with + signs from the drop-down menu. -85 0 n ChemDoodle > laarrow_forward
- Table given in P14 of Lec5, Lecture note provides the reactivity ratio for free-radical copolymerization of styrene with (a) butadiene, (b) methyl methacrylate, (c) methyl acrylate, (d) acrylonitrile, (e) maleic anhydride, (f) vinyl chloride, and (e) vinyl acetate. For each of these copolymerizations, use the terminal model copolymer composition equation to calculatethe following. (a) The composition of the copolymer formed at low conversions of an equimolar mixture of the two monomers(b) The comonomer composition required to form a copolymer consisting of 50 mol% styrene repeat units.For the azeotropic copolymerization, calculate the azeotropic compositionarrow_forwardDraw the structure(s) of the major organic product(s) of the following reaction. bul H₂C • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • If the reaction produces a racemic mixture, draw both stereoisomers. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. • Include anionic counter-ions, e.g., I, in your answer, but draw them in their own sketcher. B CH₂ PPh ChemDoodle THFarrow_forwardAn important class of substances in organic chemistry are the aliphatic and aromatic hydrocarbons and theirssubstituted derivatives. In this assignment: Remember to take any isomerism and the VSEPR principles into account A) Study the series of ring structures a – d in the figure above. Fill in the IUPAC names for the rings a - d.Are any of these rings aromatic? Can you say something about what aromaticity means chemically? B) Suggest probable IUPAC names of the following substituted hydrocarbon compounds which areshown in skeletal structure: See image barrow_forward
- 1. The following synthetic schemes all have at least one flaw in them. What is wrong with each? (a) Br CO2H CH3CH2CHCH2CH3 1. Mg CH3CH2CHCH,CH3 2. NaCN 3. H30* (b) .CH2CO2H „CH2CH3 1. LIAIH 2. H30* (c) он он CH3CCH2CH2CI 1. 1. NaCN 2. H30* CH3CCH2CH2COH CH3 CH3arrow_forwardThe conversion of alcohols into alkyl halides by reaction with hydrogen halides is an example of a Nucleophilic Substitution Reaction. This kind of reaction can proceed by two different mechanisms depending on the structure of alcohol substrates used. Generally, primary alcohols are substituted via SN2 mechanism, while secondary and tertiary alcohols undergo SN1 mechanism. Consider the following reaction given in the picture below and the questions in the picture too.arrow_forwarda) Free radical bromination is more selective than free radical chlorination. Draw a reaction coordinate diagram for the specific step in the radical chain mechanism that illustrates the source of this selectivity, and explain your reasoning. b) Explain why the bond dissociate energy (BDE) of tert-butane is 95 kcal/mol while the BDE for propane is 99 kcal/mol.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning